Three new Penicillium species isolated from the tidal flats of China
- Published
- Accepted
- Received
- Academic Editor
- Hector Mora-Montes
- Subject Areas
- Biodiversity, Molecular Biology, Mycology, Taxonomy
- Keywords
- New taxa, Penicillia, Mudflats, Mycobiota, Polyphasic taxonomy
- Copyright
- © 2022 Xu et al.
- Licence
- This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, reproduction and adaptation in any medium and for any purpose provided that it is properly attributed. For attribution, the original author(s), title, publication source (PeerJ) and either DOI or URL of the article must be cited.
- Cite this article
- 2022. Three new Penicillium species isolated from the tidal flats of China. PeerJ 10:e13224 https://doi.org/10.7717/peerj.13224
Abstract
During a survey of culturable fungi in the coastal areas of China, three new species of Penicillium sect. Lanata-Divaricata were discovered and studied with a polyphasic taxonomic approach, and then named as P. donggangicum sp. nov. (ex-type AS3.15900T = LN5H1-4), P. hepuense sp. nov. (ex-type AS3.16039T = TT2-4X3, AS3.16040 = TT2-6X3) and P. jiaozhouwanicum sp. nov. (ex-type AS3.16038T = 0801H2-2, AS3.16207 = ZZ2-9-3). In morphology, P. donggangicum is unique in showing light yellow sclerotia and mycelium, sparse sporulation, restricted growth at 37 °C, irregular conidiophores, intercalary phialides and metulae, and pyriform to subspherical conidia. P. hepuense is distinguished by the fast growth on CYA and YES and slow growth on MEA at 25 °C, weak or absence of growth at 37 °C, biverticillate and monoverticillate penicilli, and ellipsoidal conidia. P. jiaozhouwanicum is characterized by abundant grayish-green conidia en masse and moderate growth at 37 °C, the appressed biverticillate penicilli and fusiform, smooth-walled conidia. These three novelties were further confirmed by the phylogenetic analyses based on either the combined BenA-CaM-Rpb2 or the individual BenA, CaM, Rpb2 and internal transcribed spacer (ITS) sequences.
Introduction
With the application of molecular approaches in fungal taxonomy and environmental studies, the number of the estimated fungal species on the earth may be more than doubled, i.e., from about 1.5 million to 2.2–3.8 million. However, until now, the number of discovered species accounts for only 3–8%, or approximately 120,000–150,000 (Hawksworth & Lücking, 2017; Hawksworth & Lücking, 2018; Lücking et al., 2020; Lücking et al., 2021). The less-studied environments, for example, the coastal tidal flats, are thought to be the treasure house of new fungal species. China has a coastline of ca. 32,000 km and the tidal flat areas cover about 20,000 sq. km., which are potential valuable land resources either economically or ecologically (e.g., Long et al., 2016; Nicoletti & Trincone, 2016).
Numerous studies on the fungal communities of coastal environments have been carried out based on culture-dependent and/or culture-independent approaches (e.g., rDNA-based NGS) (e.g., Singh et al., 2012; Zhang et al., 2012; Heo et al., 2019). For example, Li et al. (2016) conducted an extensive study on the mycobiota of the intertidal regions along the coastline of China using an environmental metabarcoding approach with ITS2 as the barcode, which revealed the diverse fungal communities in mudflats, in which the facultative marine fungal taxa were in the majority, particularly, the members of some ubiquitous genera, such as Penicillium, Aspergillus, Cladosporium, Fusarium (as Gibberella) were abundant in aquaculture areas. However, the ITS-based metabarcoding approach alone cannot perform the accurate species identification for most of the fungal genera, especially Penicillium, Aspergillus and Talaromyces (e.g., Visagie et al., 2014a; Visagie et al., 2014b; Lücking et al., 2020; Houbraken, Visagie & Frisvad, 2021). Thus, the integration of morphological, multi-locus phylogenetic and metabarcoding approaches is considered as a proper practice in studying environmental mycobiota (e.g., Lücking et al., 2020). For instance, using morphological and multi-locus phylogenetic approaches based on ITS, BenA and RPB2 sequences, Park et al. (2014) isolated 36 Penicillium taxa along the southern and eastern coastlines of Korea, among which nine were undetermined species. Employing the culture-dependent and culture-independent approaches with ITS-based and BenA-based metabarcoding, Park et al. (2019) discovered 96 taxa of Penicillium along the western and southern intertidal zones of Korea, among which 65 were isolated with a culture-dependent method, and eight might be new species.
The number of reported Penicillium species has more than doubled since 2000, namely, about 483 until now (Pitt, Samson & Frisvad, 2000; Visagie et al., 2014a; Visagie et al., 2014b; Houbraken et al., 2020). The understudied geographic areas and ecological habitats are considered as one major source for undiscovered species (Hawksworth & Lücking, 2017; Hawksworth & Lücking, 2018). With the extensive employment of molecular approaches in the exploration of these new ecological habitats or geographical areas, especially the environmental metabarcoding technique which has shown the powerful capability to uncover new taxa, many more members of this ubiquitous genus would be expected to be discovered, though a portion of OTUs may not be true natural taxa (Lücking et al., 2020; Lücking et al., 2021).
In a survey of culturable fungi in the tidal flats of China with a culture-dependent method, we isolated numerous strains of Penicilium and here propose three new species of sect. Lanata-Divaricata in line with the taxonomic scheme of Houbraken et al. (2020), namely, P. donggangicum sp. nov., P. hepuense sp. nov., and P. jiaozhouwanicum sp. nov.
Materials & Methods
Isolation of strains
Soil samples were collected from the coastal mudflats of Yalu River Estuary Wetland Park, Donggang City, Liaoning (39°29′24″N, 124°19′12″E, 39 m); Dangjiang Town, Hepu County, Beihai City, Guangxi (21°18′13.68″N, 109°23′44.88″E, 10 m); Shatian Town, Babu District, Hezhou City, Guangxi (21°30′38″N, 109°39′58″E, 200 m); Jiaozhouwan, Qingdao City, Shandong (37°10′13″N, 120°7′36″E, 3 m); Jiulongkou, Zhangzhou City, Fujian (24°25′57N, 117°47′28E, 32 m). The dilution method of Malloch (1981) was used to isolate the culturable fungi with 0.1% agar water solution instead of water. Five distinctive strains were isolated and deposited in China General Microbiological Culture Collection Center (CGMCC) as AS3.15900 = LN5H1-4 (P. donggangicum sp. nov.); AS3.16039 = TT2-4X3, AS3.16040 = TT2-6X3 (P. hepuense sp. nov.); AS3.16038 = 0801H2-2, AS3.16207 = ZZ2-3-9 (P. jiaozhouwanicum sp. nov.).
Morphological studies
Colony characters were examined according to the methods and culturing media proposed by Raper & Thom (1949; Czapek agar, Cz), Pitt (1979; Czapek yeast autolysate agar, CYA, yeast extract; Oxoid, Hants, UK), Samson et al. (2010; 5% malt extract agar, MEA, malt extract; Oxoid, Hants, UK), and Frisvad (1981; yeast extract sucrose agar, YES, yeast extract; Oxoid, Hants, UK; creatine sucrose agar, CREA). The names of colors were referenced to Kornerup & Wanscher (1978). Wet mounts were made by picking fungal materials with reproductive structures from a colony on MEA and teasing them apart with two needles in a small drop of 85% lactic acid water solution without dye. Microscopic observation and photographs were made with an Axioplan2 imaging and Axiophot2 universal microscope (Carl Zeiss, Oberkochen, Germany).
Molecular studies
Genomic DNA was extracted with a Plant Genomic DNA Kit (Cat. No. TSP101) (TsingKe Biotech. Co., Ltd., Beijing, China). The primers bt2a and bt2b were used for PCR amplification of the partial β-tubulin gene (BenA) (Glass & Donaldson, 1995), primers AD1 and Q1 for the partial calmodulin gene (CaM) (Wang, 2012), T2 and E2 for the partial DNA-dependent RNA polymerase II second largest subunit gene (Rpb2) (Jiang et al., 2018), and ITS5 and ITS4 for the nuclear rDNA ITS1-5.8S-ITS2 (ITS) (White et al., 1990). PCR reaction was employed in the 20 µL reaction mixture containing 0.5 µL of each primer (10 pM/µL), 1.0 µL of genomic DNA (10 ng/µL), 10 µL of 2 × PCR MasterMix buffer (0.05 u/µL Taq polymerase, 4 mM MgCl2, 0.4 mM dNTPs), and 8 µL of double-distilled water (Tsingke Biotech. Co. Ltd, Beijng, China). Amplification was performed in an AB 2720 thermal cycler (Applied Biosystems, Foster City, California, USA), with the program consisting of 94 °C for 3 min, 34 cycles of 94 °C for 30 s, 50 °C for 30 s, and 72 °C for 30 s, and a last elongation at 72 °C for 5 min. The amplicons were purified and sequenced in two directions with an ABI 3730 DNA analyzer (Applied Biosystems, Foster City, California, USA). Raw sequences were proofread and edited manually with BioEdit 7.0.9 (Hall, 1999). The sequences of the five strains generated in this study were deposited in GenBank (AS3.15900: ITS = MW946996, BenA = MZ004914, CaM = MZ004918, Rpb2 = MW979253; AS3.16038: ITS = MW946993, BenA = MZ004911; CaM = MZ004915; Rpb2 = MW979252; AS3.16039: ITS = MW946994; BenA = MZ004912; CaM = MZ004916; Rpb2 = MW979254; AS3.16040: ITS = MW946995, BenA = MZ004913, CaM = MZ004917, Rpb2 = MW979255; AS3.16207: ITS = OM203537, BenA = OM220087, CaM = OM220088, Rpb2 = OM220089).
A pilot phylogenetic analysis based on BenA sequences of all the members in sect. Lanata-Divaricata was launched first, then only the species of sers. Janthinella and Oxalica were included in the final analyses to identify these five strains. Besides our five strains, there were 29 species of the two series in the analyses, among which 34 strains were included in the combined BenA-CaM-Rpb2 mactrix, 36 strains in CaM and Rpb2 mactrices, and 38 strains in BenA and ITS mactrices. In addition, three species of the closely-related sections, namely, P. stolkiae CBS 315.67 of sect. Stolkia, P. macrosclerotiorum AS3.5681 of sect. Gracilenta and P. citrinum CBS 139.45 of sect. Citrina were included in the analyses, among which P. citrinum was selected as the outgroup. The edited sequences were aligned with MUSCLE implemented in MEGA 6 with the parameters set as default (Tamura et al., 2013), then the aligned sequences were trimmed to generate sequence matrices. The concatenated BenA-CaM-Rpb2 and the individual BenA, CaM, Rpb2, and ITS matrices were analyzed with the Maximum Likelihood (ML) method and subjected to 1000 bootstrap replications, with substitution model and rates among sites set as K2+G+I for the combined BenA-CaM-Rpb2, BenA, CaM and Rpb2 sequences, and GTR+G for ITS, which were determined by the “find best DNA model” tool of MEGA 6, with the default parameters used for the best model search. Gaps were treated as partial deletion according to Hall (2013). In BenA-CaM-Rpb2 analysis, each gene region was not treated as separate partitions.
Nomenclature
The electronic version of this article in Portable Document Format (PDF) will represent a published work according to the International Code of Nomenclature for algae, fungi, and plants, and hence the new names contained in the electronic version are effectively published under that Code from the electronic edition alone. In addition, new names contained in this work have been submitted to MycoBank from where they will be made available to the Global Names Index. The unique MycoBank number can be resolved and the associated information viewed through any standard web browser by appending the MycoBank number contained in this publication to the prefix “http://www.mycobank.org/MycoTaxo.aspx?Link=T&Rec=”. The online version of this work is archived and available from the following digital repositories: PeerJ, PubMed Central, and CLOCKSS.
Results
Phylogenetic analyses
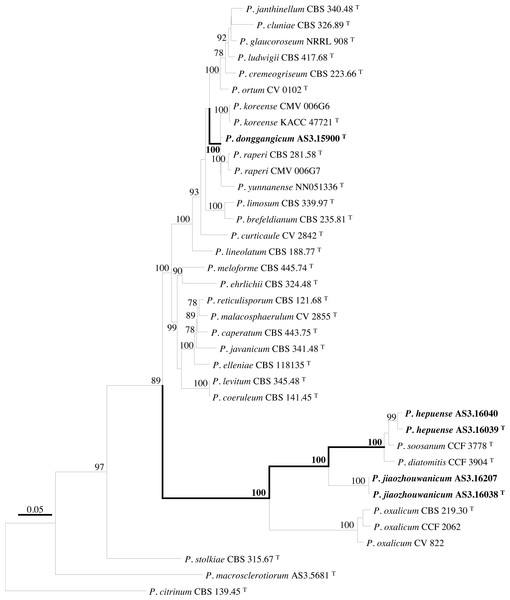
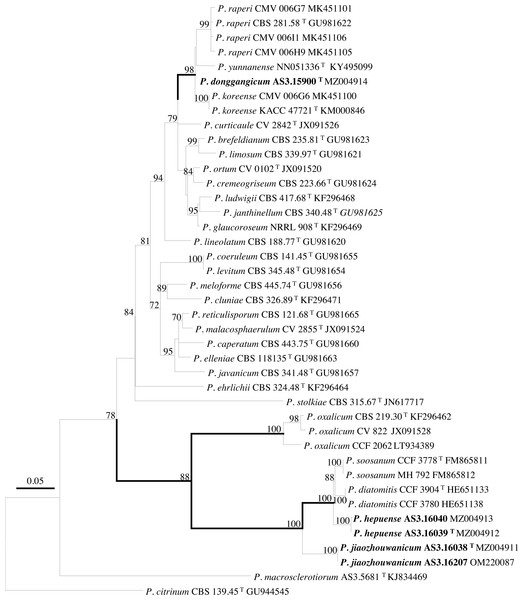
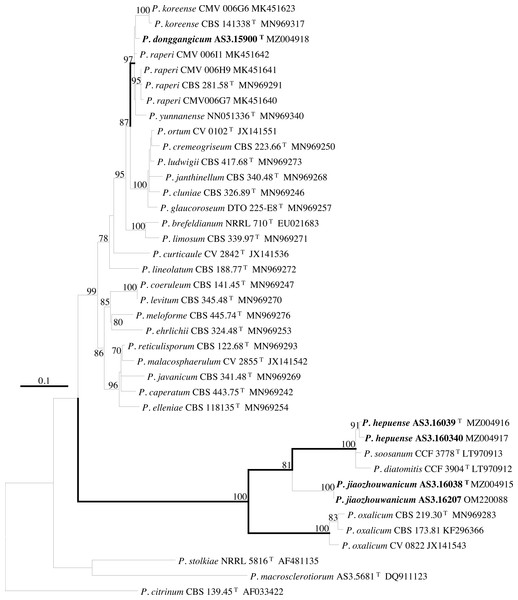
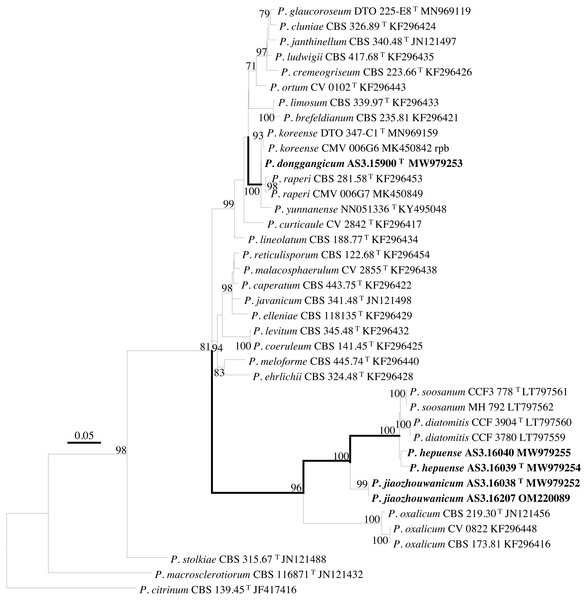
PCR amplification of BenA, CaM, Rpb2 and ITS generated ca. 410 bp, 660 bp, 800 bp, and 600 bp amplicons, respectively. The sequence matrices of BenA, CaM, Rpb2, ITS, and the concatenated BenA-CaM-Rpb2 had 441, 536, 719, 509 and 1693 characters with gaps, respectively. The phylograms inferred from either the combined BenA-CaM-Rpb2 or the four individual loci readily confirmed the three novelties (Figs. 1–4, Fig. S1).
Figure 1: ML phylogram inferred from partial BenA-CaM-Rpb2 sequences.
Bootstrap percentages over 70% derived from 1,000 replicates are indicated at the nodes, T indicates ex-type strains, strains belonging to new species are indicated in boldface. Bar = 0.05 substitutions per nucleotide position.Figure 2: ML phylogram inferred from partial BenA sequences.
Bootstrap percentages over 70% derived from 1,000 replicates are indicated at the nodes, T indicates ex-type strains, strains belonging to new species are indicated in boldface. Bar = 0.05 substitutions per nucleotide position.Figure 3: ML phylogram inferred from partial CaM sequences.
Bootstrap percentages over 70% derived from 1,000 replicates are indicated at the nodes, T indicates ex-type strains, strains belonging to new species are indicated in boldface. Bar = 0.1 substitutions per nucleotide position.Figure 4: ML phylogram inferred from partial Rpb2 sequences.
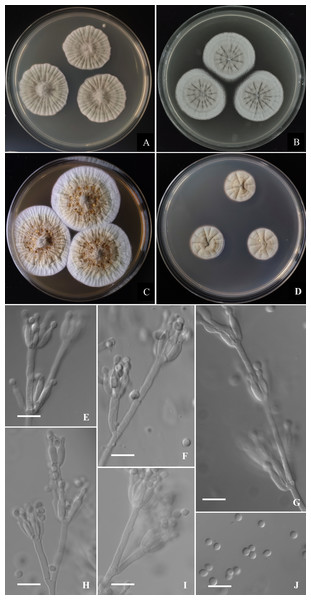
Bootstrap percentages over 70% derived from 1,000 replicates are indicated at the nodes, T indicates ex-type strains, strains belonging to new species are indicated in boldface. Bar = 0.05 substitutions per nucleotide position.Figure 5: Morphological characters of Penicillium donggangicum AS3.15900 T incubated at 25 °C for 7 days.
(A) CYA. (B) MEA. (C) YES. (D) Cz. (E)–(I) Conidiophores. (J) Conidia. Bar = 10 µm.According to the phylograms, one new species, namely, P. donggangicum is in the clade with P. koreense, P. raperi and P. yunnansense of ser. Janthinella in BenA-CaM-Rpb2, BenA, CaM, Rpb2 phylograms with 100%, 98%, 97%, and 100% bootstrap support, respectively, but ITS phylogram does not show any clades in which the new species belongs.
The other two new species, i.e., P. hepuense and P. jiaozhouwanicum are in the clade with P. soosanum, P. diatomitis and P. oxalicum of ser. Oxalica in BenA-CaM-Rpb2, BenA, CaM, Rpb2 and ITS phylograms with 100%, 88%, 100%, 96% and 98% bootstrap support, respectively. Among these five species, P. hepuense, P. soosanum and P. diatomitis consistently form an innermost subclade in BenA-CaM-Rpb2, BenA, CaM, Rpb2 and ITS phylograms with 100%, 100%, 100%, 100% and 79% bootstrap support, respectively, and P. jiaozhouwanicum is close-related to them and as an outgroup to this clade with 81%–100% bootstrap support; P. oxalicum is as the outermost taxon to these four species. Although ITS phylogram shows that these five species are in one clade with 98% bootstrap support, the two strains of P. hepuense AS3.16039 and AS3.16040 are well separated in different subclades and many clades presented in the protein gene phylograms are absent, thus only the three protein genes has been concatenated for analysis.
Taxonomy
Etymology. The specific epithet is derived from the locale where the ex-type strain was isolated, Donggang, Dandong, Liaoning, China.
Holotype. CHINA. LIAONING: Dandong, Donggang, from soil of tidal flats, 39°29′24″N, 124°19′12″E, 39 m, Mar. 29, 2020 (holotype: HMAS350265 from dried culture of ex-type AS3.15900 on CYA), ex-type culture AS3.15900 = LN5H1-4. GenBank: BenA = MZ004914, CaM = MZ004918, ITS = MW946996, Rpb2 = MW979253.
Diagnosis. This new taxon is characterized by the restricted growth at 37 °C, light yellow sclerotia, sparse sporulation, irregular conidiophores with concurrent phialides and metulae, pyriform to subspherical smooth-walled conidia.
Description
Colonies 32–34 mm diam on CYA at 25 °C after 7 d, thin, radially sulcate densely and centrally raised, margins on agar surface, sinuous, texture velutinous and overlaid with sparse aerial mycelium in central areas; sporulation sparse to moderate, conidia en masse near greyish green (27B3–C3); mycelium greyish yellow (1B2–2B2); exudate absent or limited, light yellow; limited sclerotia in central areas, light yellow; soluble pigment absent; moldy odor strong; colony underside near Reddish Blond (5C3). Colonies 30–33 mm diam on MEA at 25 °C after 7 d, thin, radially sulcate, margins on agar surface, regular; velutinous and overlaid with sparse mycelium in central areas; sporulation absent to sparse, conidia en masse light greyish green; mycelium white at colony margins and pale orange (5A3) in central areas; sclerotia abundant in central areas, light yellow to pale orange (5A3); exudate and soluble pigment absent; moldy odor strong; colony underside near grayish brown (7D3–E4). Colonies 43–45 mm diam on YES at 25 °C after 7 d, slightly thick, radially and annularly sulcate densely, centrally raised, margins on agar surface, regular; velutinous; sporulation sparse at the centers, conidia en masse grey (3C1); mycelium white at marginal areas and greyish yellow (1B2–2B2) in central areas; light yellow sclerotia abundant in central areas; exudate moderate, light brownish-yellow; soluble pigment absent; moldy odor strong; colony underside near light brown (6D7–D8). Colonies 18–21 mm diam on CA at 25 °C after 7 d, thin, radially and annularly plicate, margins submerged, regular; velutinous; sporulation absent or sparse in central areas, light grey; mycelium pale orange (5A2–A3) and white at colony margins; clear exudate absent or limited, light yellow; soluble pigment absent; moldy odor strong; colony underside near grayish brown (7D3–E4). Colonies 12–14 mm diam on CYA at 37 °C after 7 d, thick, chapped, irregularly sulcate, centrally depressed; velutinous; sporulation sparse, light gray; mycelium dirty-white; exudate and soluble pigment absent; moldy odor strong; colony underside near light yellow. On CYA at 5 °C after 7 d, no growth. On CREA at 25 °C after 7 d, colonies 16–18 mm diam, acid production absent.
Conidiophores borne from aerial hyphae or surface mycelium, stipes indeterminate, with the diameters about 2.5–3 µm, walls smooth; penicilli irregular, with intercalary phialides and metulae along the conidiophores; metulae 1–3 per verticil, 15–30 × 2–3; phialides, ampulliform, 3–7 per verticil, 9–12 × 2.5–3; conidia pyriform to subspherical, 2.5–3.5 × 2.5–3 µm, smooth-walled.
Etymology. The specific epithet is derived from the locale where the ex-type strain was isolated, Hepu, Beihai, Guangxi, China.
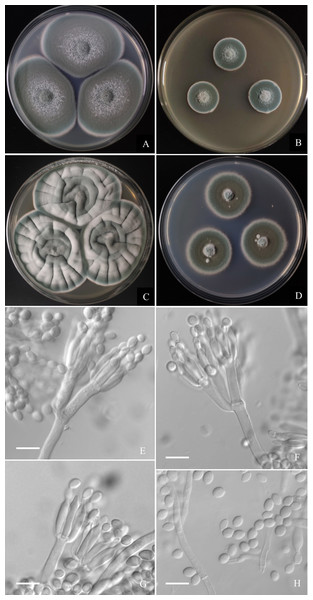
Figure 6: Morphological characters of Penicillium hepuense AS3.16039T incubated at 25 °C for 7 days.
(A) CYA. (B) MEA. (C) YES. (D) Cz. (E)–(G) Conidiophores. (H) Conidia. Bar = 10 µm.Holotype. CHINA. GUANGXI: Beihai, Hepu County, from soil of tidal flats, 21°18′13.68″N, 109°23′44.88″E, 10 m, December 30, 2020 (holotype: HMAS350263 from dried culture of ex-type AS3.16039 on CYA), ex-type culture AS3.16039 = TT2-4X3. GenBank: BenA = MZ004912, CaM = MZ004916, ITS = MW946994, Rpb2 = MW979254.
Diagnosis. This new taxon is characterized by fast growth on CYA and YES, slow growth on MEA, biverticillate and monoverticillate penicilli, ellipsoidal conidia with smooth to finely rough walls.
Description
Colonies 45–48 mm diam on CYA at 25 °C after 7 d, thin, plane, margins on agar surface, regular; velutinous; sporulation heavy, conidia en masse near greyish green (25E4); clear exudate limited; soluble pigment absent; colony underside grey, while orange grey (6B2) at the centers. Colonies 26–29 mm diam on MEA at 25 °C after 7 d, thin, plane, margins on agar surface, irregular; velutinous, and overlaid with sparse mycelium; sporulation heavy, conidia en masse near greyish green to dull green (26E3–E4); exudate and soluble pigment absent; colony underside yellowish grey (M3B2). Colonies 55–59 mm diam on YES at 25 °C after 7 d, slightly thick, radially and annularly sulcate, centrally convolute or depressed; velutinous; sporulation moderate to heavy, conidia en masse near greyish green (25D3); exudate and soluble pigment absent; colony underside brownish grey (M7C4), and darker at centers. Colonies 27–29 mm diam on CA at 25 °C after 7 d, thin, plane, margins submerged, fimbriate; velutinous; sporulation heavy, conidia en masse greyish green (25E4); mycelium white at colony margins; exudate and soluble pigment absent; colony underside orange grey (M5B2). On CYA at 37 °C after 7 d, colonies absent (AS3.16040) or 5–7 mm (AS3.16039), thick, centrally depressed, irregularly plicate; velutinous; sporulation moderate, light gray; exudate and soluble pigment absent; colony underside dark brown. On CYA at 5 °C after 7 d, no growth. On CREA at 25 °C after 7 d, colonies 20–25 mm diam, acid production absent.
Conidiophores borne from substatum or surface hyphae; stipes (60–) 150–180 (–250) × 3.5–4 µm, smooth-walled; penicilli usually biverticillate, less commonly monoverticillate, metulae 2–4 per verticil, appressed, 15–20 × 3–4 µm; phialides ampuliform, 4–8 per verticil, 12–15 × 3–3.5 µm; conidia ellipsoidal, (4–) 5–6 × 3–4 µm, walls smooth to finely rough.
Additional strains: CHINA. GUANGXI: Hezhou, Babu District, from soil of the river side, 21°30′38″N, 109°39′58″E, 200 m, Jul 12, 2020, AS3.16040 = TT2-6X3. GenBank: BenA = MZ004913, CaM = MZ004917, ITS = MW946995, Rpb2 = MW979255.
Etymology. The specific epithet is derived from the locale where the ex-type strain was isolated, Jiaozhouwan, Qingdao, Shandong, China.
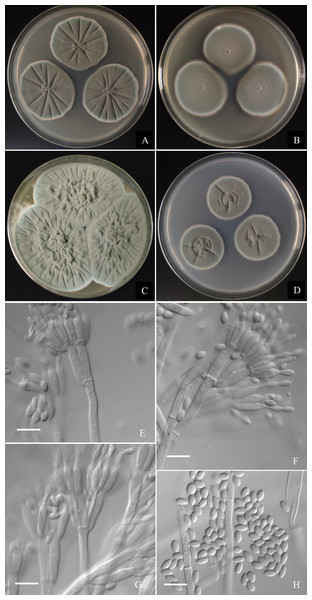
Figure 7: Morphological characters of Penicillium jiaozhouwanicum AS3.16038T incubated at 25 °C for 7 days.
(A) CYA. (B) MEA. (C) YES. (D) Cz. (E)–(G) Conidiophores. (H) Conidia. Bar = 10 µm.Holotype. CHINA. SHANDONG: Qingdao, Jiaozhouwan, from soil of tidal flats, 37°10′13″N, 120°7′36″E, 3 m, January 29, 2020 (holotype: HMAS350262 from dried culture of ex-type AS3.16038 on CYA), ex-type culture AS3.16038 = 0801H2-2. GenBank: BenA = MZ004911, CaM = MZ004915, ITS = MW946993, Rpb2 = MW979252.
Diagnosis. This new taxon is characterized by veltutinous colonies and abundant conidia, normal growth at 37 °C, appressed biverticillate penicilli and fusiform smooth-walled conidia.
Description
Colonies 37–40 mm diam on CYA at 25 °C after 7 d, thin, radially sulcate, colony margins on agar surface, regular; velutinous; sporulation abundant, conidia en masse greyish green to dull green (27D4–E4), easily falling off; mycelium white at colony margins; exudate and soluble pigment absent; colony underside near greyish green (30B3–B4). Colonies 34–37 mm diam on MEA at 25 °C after 7 d, thin, plane, margins on agar surface, regular; velutinous; sporulation abundant, conidia en masse near greyish green to dull green (27D4–E4), easily falling off; mycelium white at colony margins; exudate and soluble pigment absent; colony underside near brownish grey (6C2). Colonies 57–60 mm diam on YES at 25 °C after 7 d, densely radially to irregularly sulcate with convoluted centers, margins submerged, regular; velutinous; sporulation abundant, conidia en masse near greyish green (27C3), easily falling off; mycelium white at colony margins; exudate and soluble pigment absent; colony underside commonly near greyish yellow (3C3) at margins, while dull yellow (3B3) at centers. Colonies 26–29 mm diam on CA at 25 °C after 7 d, thin, radially sulcate slightly; margins on agar surface, regular; velutinous; sporulation abundant, conidia en masse near greyish green to dull green (27C3–D3), easily falling off; mycelium white at colony margins; exudate and soluble pigment absent; colony underside near greyish green (30C5). Colonies 23–25 mm diam on CYA at 37 °C after 7 d, thin, radially sulcate, centrally convolute, margins on surface, lightly sinuous; sporulation abundant, conidia en masse near greyish green (27C3), easily falling off; mycelium white at colony margins; exudate and soluble pigment absent; colony underside near light brown at margins, while dark brown at centers. On CYA at 5 °C after 7 d, no growth. On CREA at 25 °C after 7 d, colonies 28–30 mm diam, acid production weak, just underneath the colony.
Conidiophores borne from substratum; stipes from substratum 140–240 × 3–3.5 µm, smooth-walled; penicilli biverticillate, occasionally with one subterminal branch bearing biverticillate or monoverticillate penicilli; metulae 2–4 per verticil, appressed, 13–15 × 3–3.5; phialides, ampuliform with long collula, 4–8 per verticil, 10–13 × 2.5–3.5; conidia fusiform, seldom ellipsoidal, (3.5–) 4–5.5 × 2–2.5 (–3) µm, smooth-walled.
Additional strains: CHINA. FUJIAN: Zhangzhou, Jiulongkou, from soil of tidal flats, 24°25′57N, 117°47′28E, 32 m, Apr 22, 2021, AS3.16027 = ZZ2-9-3. GenBank: BenA = OM220087, CaM = OM220088, ITS = OM203537, Rpb2 = OM220089.
Discussion
According to the taxonomic scheme of Houbraken et al. (2020), P. donggangicum belongs to ser. Jathinella; P. hepuense and P. jiaozhouwanicum belong to ser. Oxalica of sect. Lanata-Divaricata in subgen. Aspergilloides. In the phylogenies inferred from the three protein genes, P. donggangicum, P. koreense, P. raperi and P. yunnanense consistently form a clade with strong bootstrap support, and P. jiaozhouwanicum, P. hepuense, P. soosanum, P. diatomitis and P. oxalicum are closely related with moderate to strong bootstrap support (Figs. 1–4, Fig. S1).
The morphological characters, such as sparse sporulation, light yellow mycelium and the intercalary vericils of phialides and metulae of P. donggangicum quite resemble those of P. raperi and the phylogenies based on the protein-coding genes also showed the close relationship between them. But physiologically, P. donggangicum grows faster than P. raperi at 25 °C (CYA: 32–34 mm vs. 21–23 mm, MEA: 30–33 mm vs. 23–24 mm, YES: 43–45 mm vs. 27–28 mm) and the colony reverse on CYA of P. donggangicum shows orange-brown while that of P. raperi shows olive color. Moreover, P. donggangicum produces abundant sclerotia, while P. raperi does not produce sclerotia. Though the conidiophores of the two species are both from aerial hyphae with intercalary metulae and phialdes, and their metulae are unequal in length and divergently positioned, the diameters of the conidiophore elements of the new species are relatively larger than those of P. raperi (2–3 µm vs. 1.3–2.2 µm) (Smith, 1957), and the conidia of P. donggangicum are smooth-walled but those of P. raperi are slightly rough (Smith, 1957; Visagie et al., 2015). P. donggangicum shows faster growth than P. yunnanense at 25 °C on CYA (32–34 mm vs. 23–25 mm) and YES (43–45 mm vs. 22–23 mm), in addition, P. donggangicum produces abundant sclerotia, light yellow mycelium and grows at 37 °C, but P. yunnanense does not produce sclerotia, bears white mycelium and cannot grow at 37 °C. Microscopically, P. donggangicum bears concurrent verticils of phialides and metulae along the trailing conidiophores, whereas P. yunnanense does not have this character (Diao et al., 2018). The sparse sporulation, abundant sclerotia, light yellow mycelium and concurrent phialides and metulae also distinguish P. donggangicum from P. koreense, which has moderate to heavy sporulation, white mycelium and predominantly monoverticillate penicilli, and lacks sclerotia (You et al., 2014). A synopsis of these morphological comparisons among the four species is given in Table 1.
| P. donggangicum | P. raperia | P. yunnanenseb | P. koreensec | |
|---|---|---|---|---|
| On CYA at 25 C, 7 d diam (mm) | 32–34 | 21–23 | 23–25 | 32–34 |
| On MEA at 25 C, 7 d diam (mm) | 30–33 | 23–24 | 33–34 | 42–48 |
| On YES at 25 C, 7 d diam (mm) | 43–45 | 27–28 | 22–23 | N/A |
| On CYA at 37 C, 7 d diam (mm) | 12–14 | 15–16 | Absent | 15–19 |
| Sclerotia | Abundant | Absent | Absent | Absent |
| Mycelium color | Yellow | Pale yellow to mauve | White | White |
| Sporulation | Sparse | Sparse | Sparse to moderate | Moderate to heavy |
| Penicilli | Irregular, with intercalary metulae and phialides | Irregular, with intercalary metulae and phialides | Monoverticillate to irregular | Predominantly monoverticillate |
| Conidiophore elements diam (µm) | 2–3 | 1.3–2.2 | 2–4 | 2–3 |
| Conidia | Pyriform to subspherical, smooth | Irregularly ovate, slightly rough | Broadly ellipsoidal to subglobose, smooth | Globose to broadly ellipsoidal, smooth or finely rough |
Notes:
| P. hepuense | P. jiaozhouwanicum | P. oxalicuma | P. diatomitisa | P. soosanua | |
|---|---|---|---|---|---|
| On CYA at 25 C, 7 d diam (mm) | 45–48 | 37–40 | 36–62 | 45–65 | 45–65 |
| On MEA at 25 C, 7d diam (mm) | 26–29 | 34–37 | 25–62 | 48–62 | 55–65 |
| On YES at 25 C, 7d diam (mm) | 55–59 | 57–60 | 37–63 | 59–67 | 60–69 |
| On CYA at 37 C, 7 d diam (mm) | 0–7 | 23–25 | 10–38 | 5–25 | 5–20 |
| On CREA at 25 C, 7 d diam (mm); acid production | 20–25; absent | 28–30; weak | 12–19; poor | 7–13; absent | 13–17; absent |
| Penicilli | Biverticillate, rarely monoverticillate | Biverticillate, rarely with a subterminal metula | Biverticillate and monoverticillate, rarely terverticillate | Biverticillate | Biverticillate, rarely with a subterminal metula |
| Stipe length (µm) | 60–250 | 140–240 | 75–550 | 75–300 | 60–250 |
| Conidia | Ellipsoidal, smooth to finely rough | Fusiform, seldom ellipsoidal, smooth | Ellipsoidal, finely rough | Ellipsoidal, finely rough | Broadly ellipsoidal, subglobose, rough |
Notes:
Only three species were included in ser. Oxalica before this study (Houbraken et al., 2020). There are many characters shared by the taxa in this series, such as good growth at 25 °C and 37 °C, typical velutinous colonies with abundant dark green conidia en masse, predominantly biverticillate penicilli with 2–4 appressed metulae, and ellipsoidal conidia. One new member introduced to this series here, namely, P. hepuense possesses most of these common characters but its maximal growth rate at 25 °C is much less than those of the other members (29 mm vs. 62 mm for P. oxalicum, 62 mm for P. diatomitis, 57 mm for P. soosanum), and it does not grow at 37 °C, or only forms colonies about 5–7 mm diam, while the other members all grow well at 37 °C. It can be further distinguished from P. oxalicum by the absence of acid production on CREA, from P. diatomitis by longer conidia (4–6 µm vs. 3.5–4.5 µm) and producing a portion of monoverticillate penicilli, from P. soosanum by the smooth to finely rough ellipsoidal conidia and the production of a portion of monoverticillate penicilli. Another new species, i.e., P. jiaozhouwanicum is different from the above four species by its distinctive smooth-walled fusiform conidia, and metulae of nearly equal length. In pairwise comparison, P. jiaozhouwanicum differs from P. oxalicum by the weaker acid production on CREA (“underneath the colony” vs. “in colony periphery”), and much shorter stipes in maximal length (240 µm vs. 550 µm) (Kubátová et al., 2019). Though the isolate IJFM 2062 (ex-type of “P. asturianum”) also produces conidia in fusiform, their walls are roughened and the roughening is arranged in spiral bands (Ramírez & Martínez, 1981). The lower growth rate on CYA and MEA at 25 °C and faster growth at 37 °C of P. jiaozhouwanicum allow its distinction from P. diatomitis (CYA: 37–40 mm vs. 45–65 mm, and MEA: 34–37 mm vs. 48–62 mm at 25 °C; 20–23 mm vs. 7–14 mm on CYA at 37 °C). P. jiaozhouwanicum can be further distinguished from P. soosanum by producing smooth-walled, fusiform conidia, while the latter has roughened, subglobose to broadly ellipsoidal conida. In addition, P. jiaozhouwanicum shows slower growth at 25 °C (CYA: 37–40 mm vs. 45–65 mm, MEA: 34–37 mm vs. 55–65 mm) but faster growth at 37 °C (20–23 mm vs. 5–20 mm) than P. soosanum (Kubátová et al., 2018). Comparing to P. hepuense, P. jiaozhouwanicum grows faster on MEA at 25 °C (34–37 mm vs. 26–29 mm) and grows moderately at 37 °C (20–23 mm vs. 0–7 mm), P. hepuense bears a portion of monoverticillate penicilli, but P. jiaozhouwanicum commonly has biverticillate ones. A synopsis of these morphological comparisons among these five species is given in Table 2.
In this study, we employed a culture-dependent approach to isolate the culturable fungi and identified them using a polyphasic taxonomic method aiming to enrich the culture collection from various environmemts. Though the NGS using Illumina sequencing platform is widely used in the metabarcoding studies on environmental mycobiota, flaws exist in each step of the workflow (e.g., Hugerth & Andersson, 2017). Particularly, the genetic markers commonly used are the sub-regions of ITS1-5.8S-ITS2, namely, ITS1 and ITS2, which are as short as about only 360 bp and 300 bp, respectively (e.g., Francioli et al., 2021). According to literatures and our experience, ITS1-5.8S-ITS2 alone cannot provide the accurate identification of species of certain widely distributed genera, such as Aspergillus, Cladosporium, Fusarium, Penicillium, Talaromyces, etc. Some species in Aspergillus and Penicillium have identical ITS sequences, for instance, A. flavus and A. minisclerotigenes; P. echinulatum and P. solitum, etc. (e.g., Houbraken, Visagie & Frisvad, 2021). Although certain protein genes can be used as the alternative barcodes in NGS, they are usually genus-specific and not easy to be PCR-amplified, and their sequence databases are incomplete. Therefore, the ployphasic method including phenotypic, phylogenetic and metabarcoding approaches in studying environmental mycobiota is recommended, which would present both the outlines of species richness and the accuracy and precision of species delimitation if properly used (e.g., Lücking et al., 2020).
Supplemental Information
ML phylogram inferred from partial ITS sequences
Bootstrap percentages over 70% derived from 1000 replicates are indicated at the nodes, T indicates ex-type strains, strains belonging to new species are indicated in boldface. Bar = 0.02 substitutions per nucleotide position.
The alignment of BenA-CaM-Rpb2 sequences
View using MEGA software.
The alignment of ITS1-5.8S-ITS2 sequences
View using MEGA software.