Polymeric Micelles for Breast Cancer Therapy: Recent Updates, Clinical Translation and Regulatory Considerations
Abstract
:1. Introduction
2. Properties and Recent Updates of PMs
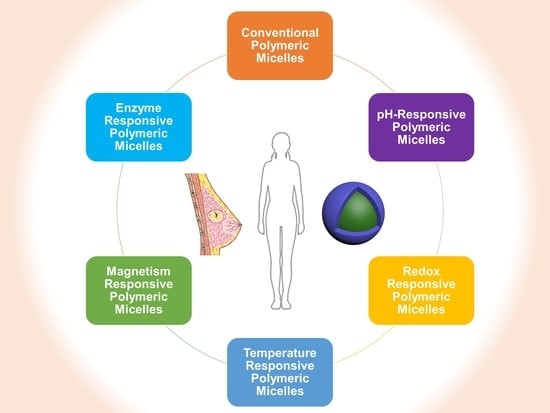
3. PMs for Breast Cancer
3.1. Conventional PMs
3.2. pH-Responsive PMs
3.3. Redox-Responsive PMs
3.4. Temperature-Responsive PMs
3.5. Magnetism-Responsive PMs
3.6. Enzyme-Responsive PMs
4. Clinical Studies
5. Regulatory Aspects
5.1. Regulatory Guidelines, eCTD Submission Format in Various Countries
5.2. Guidelines Specific to PMs and Injectable Nanomedicines:
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wilkinson, L.; Gathani, T. Understanding breast cancer as a global health concern. Br. J. Radiol. 2022, 95, 1130. [Google Scholar] [CrossRef] [PubMed]
- Lei, S.; Zheng, R.; Zhang, S.; Wang, S.; Chen, R.; Sun, K.; Zeng, H.; Zhou, J.; Wei, W. Global patterns of breast cancer incidence and mortality: A population-based cancer registry data analysis from 2000 to 2020. Cancer Commun. 2021, 41, 1183–1194. [Google Scholar] [CrossRef] [PubMed]
- Ring, A.; Harper-Wynne, C.; Smith, I.; Breast Cancer. Cancer Chemotherapy and Biological Response Modifiers. 2005. Available online: https://www.who.int/news-room/fact-sheets/detail/breast-cancer (accessed on 10 August 2022).
- Medina, M.A.; Oza, G.; Sharma, A.; Arriaga, L.; Hernández, J.M.H.; Rotello, V.M.; Ramirez, J.T. Triple-Negative Breast Cancer: A Review of Conventional and Advanced Therapeutic Strategies. Int. J. Environ. Res. Public Health. 2020, 17, 2078. [Google Scholar] [CrossRef]
- Jain, V.; Kumar, H.; Anod, H.V.; Chand, P.; Gupta, N.V.; Dey, S.; Kesharwani, S.S. A review of nanotechnology-based approaches for breast cancer and triple-negative breast cancer. J. Control. Release 2020, 326, 628–647. [Google Scholar] [CrossRef] [PubMed]
- Alven, S.; Aderibigbe, B.A. The Therapeutic Efficacy of Dendrimer and Micelle Formulations for Breast Cancer Treatment. Pharmaceutics 2020, 12, 1212. [Google Scholar] [CrossRef]
- Saloustros, E.; Mavroudis, D.; Georgoulias, V. Paclitaxel and docetaxel in the treatment of breast cancer. Expert Opin. Pharmacother. 2008, 9, 2603–2616. [Google Scholar] [CrossRef]
- Khasraw, M.; Bell, R.; Dang, C. Epirubicin: Is it like doxorubicin in breast cancer? A clinical review. Breast 2012, 21, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Shamseddine, A.I.; Farhat, F.S. Platinum-Based Compounds for the Treatment of Metastatic Breast Cancer. Chemotherapy 2011, 57, 468–487. [Google Scholar] [CrossRef]
- Li, Z.-Y.; Zhang, Z.; Cao, X.-Z.; Feng, Y.; Ren, S.-S. Platinum-based neoadjuvant chemotherapy for triple-negative breast cancer: A systematic review and meta-analysis. J. Int. Med Res. 2020, 48, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Tsume, Y.; Mudie, D.M.; Langguth, P.; Amidon, G.E.; Amidon, G.L. The Biopharmaceutics Classification System: Subclasses for in vivo predictive dissolution (IPD) methodology and IVIVC. Eur. J. Pharm. Sci. 2014, 57, 152–163. [Google Scholar] [CrossRef] [Green Version]
- Basak, D.; Arrighi, S.; Darwiche, Y.; Deb, S. Comparison of Anticancer Drug Toxicities: Paradigm Shift in Adverse Effect Profile. Life 2021, 12, 48. [Google Scholar] [CrossRef] [PubMed]
- Akbarzadeh, A.; Rezaei-Sadabady, R.; Davaran, S.; Joo, S.W.; Zarghami, N.; Hanifehpour, Y.; Samiei, M.; Kouhi, M.; Nejati-Koshki, K. Liposome: Classification, preparation, and applications. Nanoscale Res. Lett. 2013, 8, 102. [Google Scholar] [CrossRef] [PubMed]
- Ridolfo, R.; Tavakoli, S.; Junnuthula, V.; Williams, D.S.; Urtti, A.; Van Hest, J.C.M. Exploring the Impact of Morphology on the Properties of Biodegradable Nanoparticles and Their Diffusion in Complex Biological Medium. Biomacromolecules 2020, 22, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Junnuthula, V.; Boroujeni, A.S.; Cao, S.; Tavakoli, S.; Ridolfo, R.; Toropainen, E.; Ruponen, M.; van Hest, J.; Urtti, A. Intravitreal Polymeric Nanocarriers with Long Ocular Retention and Targeted Delivery to the Retina and Optic Nerve Head Region. Pharmaceutics 2021, 13, 445. [Google Scholar] [CrossRef]
- Ramachandran, R.; Junnuthula, V.R.; Gowd, G.S.; Ashokan, A.; Thomas, J.; Peethambaran, R.; Thomas, A.; Unni, A.K.K.; Panikar, D.; Nair, S.V.; et al. Theranostic 3-Dimensional nano brain-implant for prolonged and localized treatment of recurrent glioma. Sci. Rep. 2017, 7, srep43271. [Google Scholar] [CrossRef]
- Devassy, G.; Ramachandran, R.; Jeena, K.; Junnuthula, V.R.; Gopinatha, V.K.; Manju, C.A.; Manohar, M.; Nair, S.V.; Raghavan, S.C.; Koyakutty, M.; et al. Simultaneous release of two drugs from polymer nano-implant inhibits recurrence in glioblastoma spheroids. Precis. Nanomed. 2018, 2, 218–229. [Google Scholar] [CrossRef]
- Khatik, R.; Dwivedi, P.; Junnuthula, V.R.; Sharma, K.; Chuttani, K.; Mishra, A.K.; Dwivedi, A.K. Potential in vitro and in vivo colon specific anticancer activity in a HCT-116 xenograft nude mice model: Targeted delivery using enteric coated folate modified nanoparticles. RSC Adv. 2015, 5, 16507–16520. [Google Scholar] [CrossRef]
- Gera, S.; Sampathi, S.; Maddukuri, S.; Dodoala, S.; Junnuthula, V.; Dyawanapelly, S. Therapeutic Potential of Naringenin Nanosuspension: In Vitro and In Vivo Anti-Osteoporotic Studies. Pharmaceutics 2022, 14, 1449. [Google Scholar] [CrossRef]
- Pailla, S.R.; Sampathi, S.; Junnuthula, V.; Maddukuri, S.; Dodoala, S.; Dyawanapelly, S. Brain-Targeted Intranasal Delivery of Zotepine Microemulsion: Pharmacokinetics and Pharmacodynamics. Pharmaceutics 2022, 14, 978. [Google Scholar] [CrossRef]
- Dyawanapelly, S.; Junnuthula, V.R.; Singh, A. The Holy Grail of Polymer Therapeutics for Cancer Therapy: An Overview on the Pharmacokinetics and Bio Distribution. Curr. Drug Metab. 2015, 16, 522–537. [Google Scholar] [CrossRef]
- Jain, A.S.; Pawar, P.S.; Sarkar, A.; Junnuthula, V.; Dyawanapelly, S. Bionanofactories for Green Synthesis of Silver Nanoparticles: Toward Antimicrobial Applications. Int. J. Mol. Sci. 2021, 22, 11993. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Sodha, S.J.; Junnuthula, V.; Kolimi, P.; Dyawanapelly, S. Novel and investigational therapies for wet and dry age-related macular degeneration. Drug Discov. Today 2022, 27, 2322–2332. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Junnuthula, V.; Dyawanapelly, S. Ocular Therapeutics and Molecular Delivery Strategies for Neovascular Age-Related Macular Degeneration (nAMD). Int. J. Mol. Sci. 2021, 22, 10594. [Google Scholar] [CrossRef] [PubMed]
- Shete, M.B.; Patil, T.S.; Deshpande, A.S.; Saraogi, G.; Vasdev, N.; Deshpande, M.; Rajpoot, K.; Tekade, R.K. Current trends in theranostic nanomedicines. J. Drug Deliv. Sci. Technol. 2022, 71, 103280. [Google Scholar] [CrossRef]
- Su, H.; Wang, F.; Ran, W.; Zhang, W.; Dai, W.; Wang, H.; Anderson, C.F.; Wang, Z.; Zheng, C.; Zhang, P.; et al. The role of critical micellization concentration in efficacy and toxicity of supramolecular polymers. Proc. Natl. Acad. Sci. USA 2020, 117, 4518–4526. [Google Scholar] [CrossRef]
- SBandopadhyay, S.; Manchanda, S.; Chandra, A.; Ali, J.; Deb, P.K. Overview of different carrier systems for advanced drug delivery. In Drug Delivery Systems; Academic Press: Cambridge, MA, USA, 2019; pp. 179–233. [Google Scholar]
- Yadav, D.; Sandeep, K.; Pandey, D.; Dutta, R.K. Liposomes for Drug Delivery. J. Biotechnol. Biomater. 2017, 7, 1–8. [Google Scholar] [CrossRef]
- Oerlemans, C.; Bult, W.; Bos, M.; Storm, G.; Nijsen, J.F.W.; Hennink, W.E. Polymeric Micelles in Anticancer Therapy: Targeting, Imaging and Triggered Release. Pharm. Res. 2010, 27, 2569–2589. [Google Scholar] [CrossRef]
- Ghosh, B.; Biswas, S. Polymeric micelles in cancer therapy: State of the art. J. Control. Release 2021, 332, 127–147. [Google Scholar] [CrossRef] [PubMed]
- Hari, S.K.; Gauba, A.; Shrivastava, N.; Tripathi, R.M.; Jain, S.K.; Pandey, A.K. Polymeric micelles and cancer therapy: An ingenious multimodal tumor-targeted drug delivery system. Drug Deliv. Transl. Res. 2022, 1–29. [Google Scholar] [CrossRef]
- Majumder, N.; Das, N.G.; Das, S.K. Polymeric micelles for anticancer drug delivery. Ther. Deliv. 2020, 11, 613–635. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, E.; Yang, J.; Cao, Z. Strategies to improve micelle stability for drug delivery. Nano Res. 2018, 11, 4985–4998. [Google Scholar] [CrossRef] [PubMed]
- Miskolczy, Z.; Sebők-Nagy, K.; Biczók, L.; Göktürk, S. Aggregation and micelle formation of ionic liquids in aqueous solution. Chem. Phys. Lett. 2004, 400, 296–300. [Google Scholar] [CrossRef]
- Gaucher, G.; Dufresne, M.-H.; Sant, V.P.; Kang, N.; Maysinger, D.; Leroux, J.-C. Block copolymer micelles: Preparation, characterization and application in drug delivery. J. Control. Release 2005, 109, 169–188. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, M.; Fukushima, S.; Uehara, R.; Okamoto, K.; Kataoka, K.; Sakurai, Y.; Okano, T. Characterization of physical entrapment and chemical conjugation of adriamycin in polymeric micelles and their design for in vivo delivery to a solid tumor. J. Control. Release 1998, 50, 79–92. [Google Scholar] [CrossRef]
- Ghezzi, M.; Pescina, S.; Padula, C.; Santi, P.; Del Favero, E.; Cantù, L.; Nicoli, S. Polymeric micelles in drug delivery: An insight of the techniques for their characterization and assessment in biorelevant conditions. J. Control. Release 2021, 332, 312–336. [Google Scholar] [CrossRef]
- Adak, T.; Kumar, J.; Shakil, N.A.; Walia, S.; Kumar, A.; Watterson, A.C. Synthesis and Characterization of Novel Surfactant Molecules Based on Amphiphilic Polymers. J. Macromol. Sci. Part A 2011, 48, 767–775. [Google Scholar] [CrossRef]
- Sarkar, D.J.; Kumar, J.; Shakil, N.A.; Adak, T.; Watterson, A.C. Synthesis and Characterization of Amphiphilic PEG Based Aliphatic and Aromatic Polymers and their Self-Assembling Behavior. J. Macromol. Sci. Part A 2012, 49, 455–465. [Google Scholar] [CrossRef]
- Pang, N.; Li, J.; Sun, A.; Yang, Z.; Cheng, S.; Qi, X.-R. Prior anti-CAFs break down the CAFs barrier and improve accumulation of docetaxel micelles in tumor. Int. J. Nanomed. 2018, 13, 5971–5990. [Google Scholar] [CrossRef]
- Liang, Y.; Huo, Q.; Lu, W.; Jiang, L.; Gao, W.; Xu, L.; Han, S.; Cao, J.; Zhang, T.; Sun, Y.; et al. Fluorescence Resonance Energy Transfer Visualization of Molecular Delivery from Polymeric Micelles. J. Biomed. Nanotechnol. 2018, 14, 1308–1316. [Google Scholar] [CrossRef]
- Söderman, O.; Stilbs, P.; Price, W.S. NMR studies of surfactants. Concepts Magn. Reson. Part A Bridg. Educ. Res. 2004, 23, 121–135. [Google Scholar] [CrossRef]
- Furó, I. NMR spectroscopy of micelles and related systems. J. Mol. Liq. 2005, 117, 117–137. [Google Scholar] [CrossRef]
- Rahmani, A.; Mousavi, H.Z.; Salehi, R.; Bagheri, A. Novel pH-sensitive and biodegradable micelles for the combined delivery of doxorubicin and conferone to induce apoptosis in MDA-MB-231 breast cancer cell line. RSC Adv. 2020, 10, 29228–29246. [Google Scholar] [CrossRef] [PubMed]
- Topel, Ö.; Çakır, B.A.; Budama, L.; Hoda, N. Determination of critical micelle concentration of polybutadiene-block-poly(ethyleneoxide) diblock copolymer by fluorescence spectroscopy and dynamic light scattering. J. Mol. Liq. 2013, 177, 40–43. [Google Scholar] [CrossRef]
- Owen, S.C.; Chan, D.P.; Shoichet, M.S. Polymeric micelle stability. Nano Today 2012, 7, 53–65. [Google Scholar] [CrossRef]
- Minatti, E.; Viville, P.; Borsali, R.; Schappacher, M.; Deffieux, A.A.; Lazzaroni, R. Micellar Morphological Changes Promoted by Cyclization of PS-b-PI Copolymer: DLS and AFM Experiments. Macromolecules 2003, 36, 4125–4133. [Google Scholar] [CrossRef]
- Kuntsche, J.; Horst, J.C.; Bunjes, H. Cryogenic transmission electron microscopy (cryo-TEM) for studying the morphology of colloidal drug delivery systems. Int. J. Pharm. 2011, 417, 120–137. [Google Scholar] [CrossRef]
- Trujillo, M.; Schramm, M.P. Measuring Critical Micelle Concentration as a Function of Cavitand Additives Using Surface Tension and Dye Micellization. Ronald E. McNair Postbaccalaureate Achiev. Progr. 2010, 14, 155–168. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30627055%0Ahttp://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC6322906 (accessed on 13 August 2022).
- Ray, G.B.; Chakraborty, I.; Moulik, S.P. Pyrene absorption can be a convenient method for probing critical micellar concentration (cmc) and indexing micellar polarity. J. Colloid Interface Sci. 2005, 294, 248–254. [Google Scholar] [CrossRef]
- Fluksman, A.; Benny, O.; Ofra, B. A robust method for critical micelle concentration determination using coumarin-6 as a fluorescent probe. Anal. Methods 2019, 11, 3810–3818. [Google Scholar] [CrossRef]
- Sabra, S.A.; Sheweita, S.A.; Haroun, M.; Ragab, D.; Eldemellawy, M.A.; Xia, Y.; Goodale, D.; Allan, A.L.; Elzoghby, A.O.; Rohani, S. Magnetically Guided Self-Assembled Protein Micelles for Enhanced Delivery of Dasatinib to Human Triple-Negative Breast Cancer Cells. J. Pharm. Sci. 2018, 108, 1713–1725. [Google Scholar] [CrossRef]
- Hanafy, N.A.N.; El-Kemary, M.; Leporatti, S. Micelles structure development as a strategy to improve smart cancer therapy. Cancers 2018, 10, 238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, G.; Ning, Q.; Mo, Z.; Tang, S. Intelligent polymeric micelles for multidrug co-delivery and cancer therapy. Artif. Cells Nanomed. Biotechnol. 2019, 47, 1476–1487. [Google Scholar] [CrossRef] [PubMed]
- Ahamed, M.; Akhtar, M.J.; Khan, M.M.; A Alhadlaq, H. SnO2-Doped ZnO/Reduced Graphene Oxide Nanocomposites: Synthesis, Characterization, and Improved Anticancer Activity via Oxidative Stress Pathway. Int. J. Nanomed. 2021, 16, 89–104. [Google Scholar] [CrossRef] [PubMed]
- Ahamed, M.; Akhtar, M.J.; Khan, M.A.M.; Alhadlaq, H.A. Enhanced Anticancer Performance of Eco-Friendly-Prepared Mo-ZnO/RGO Nanocomposites: Role of Oxidative Stress and Apoptosis. ACS Omega 2022, 7, 7103–7115. [Google Scholar] [CrossRef]
- Ahamed, M.; Akhtar, M.J.; Khan, M.M.; Alhadlaq, H.A. Facile green synthesis of ZnO-RGO nanocomposites with enhanced anticancer efficacy. Methods 2021, 199, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Jhaveri, A.; Torchilin, V.P. Multifunctional polymeric micelles for delivery of drugs and siRNA. Front. Pharmacol. 2014, 5, 77. [Google Scholar] [CrossRef] [PubMed]
- Nasongkla, N.; Bey, E.; Ren, J.; Ai, H.; Khemtong, C.; Guthi, J.S.; Chin, S.-F.; Sherry, A.D.; Boothman, D.A.; Gao, J. Multifunctional Polymeric Micelles as Cancer-Targeted, MRI-Ultrasensitive Drug Delivery Systems. Nano Lett. 2006, 6, 2427–2430. [Google Scholar] [CrossRef]
- Carrillo-Castillo, T.D.; Castro-Carmona, J.S.; Luna-Velasco, A.; Zaragoza-Contreras, E.A. pH-responsive polymer micelles for methotrexate delivery at tumor microenvironments. e-Polymers 2020, 20, 624–635. [Google Scholar] [CrossRef]
- Ku, T.-H.; Chien, M.-P.; Thompson, M.P.; Sinkovits, R.S.; Olson, N.H.; Baker, T.S.; Gianneschi, N.C. Controlling and Switching the Morphology of Micellar Nanoparticles with Enzymes. J. Am. Chem. Soc. 2011, 133, 8392–8395. [Google Scholar] [CrossRef]
- Wang, X.; Xuan, Z.; Zhu, X.; Sun, H.; Li, J.; Xie, Z. Near-infrared photoresponsive drug delivery nanosystems for cancer photo-chemotherapy. J. Nanobiotechnol. 2020, 18, 108. [Google Scholar] [CrossRef]
- Kennedy, L.; Sandhu, J.K.; Harper, M.-E.; Cuperlovic-Culf, M. Role of Glutathione in Cancer: From Mechanisms to Therapies. Biomolecules 2020, 10, 1429. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.-C.; Wang, J.-X.; Tao, W.; Sun, C.-Y.; Wang, Y.-C.; Li, D.-D.; Fan, F.; Qian, H.-S.; Yang, X.-Z. Redox-Responsive Polyphosphoester-Based Micellar Nanomedicines for Overriding Chemoresistance in Breast Cancer Cells. ACS Appl. Mater. Interfaces 2015, 7, 26315–26325. [Google Scholar] [CrossRef] [PubMed]
- Varshosaz, J.; Sadeghi-Aliabadi, H.; Ghasemi, S.; Behdadfar, B. Use of Magnetic Folate-Dextran-Retinoic Acid Micelles for Dual Targeting of Doxorubicin in Breast Cancer. BioMed Res. Int. 2013, 2013, 680712. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Li, D.; Lu, Y.; Jiang, K.; Yang, Y.; Xu, Y.; Dong, L.; Yan, X.; Ling, D.; Yang, X.; et al. Ferrimagnetic mPEG-b-PHEP copolymer micelles loaded with iron oxide nanocubes and emodin for enhanced magnetic hyperthermia–chemotherapy. Natl. Sci. Rev. 2020, 7, 723–736. [Google Scholar] [CrossRef] [PubMed]
- Zajdel, A.; Wilczok, A.; Jelonek, K.; Musiał-Kulik, M.; Foryś, A.; Li, S.; Kasperczyk, J. Cytotoxic Effect of Paclitaxel and Lapatinib Co-Delivered in Polylactide-co-Poly(ethylene glycol) Micelles on HER-2-Negative Breast Cancer Cells. Pharmaceutics 2019, 11, 169. [Google Scholar] [CrossRef]
- Wan, X.; Beaudoin, J.J.; Vinod, N.; Min, Y.; Makita, N.; Bludau, H.; Jordan, R.; Wang, A.; Sokolsky, M.; Kabanov, A.V. Co-delivery of paclitaxel and cisplatin in poly(2-oxazoline) polymeric micelles: Implications for drug loading, release, pharmacokinetics and outcome of ovarian and breast cancer treatments. Biomaterials 2018, 192, 1–14. [Google Scholar] [CrossRef]
- Xiang, J.; Wu, B.; Zhou, Z.; Hu, S.; Piao, Y.; Zhou, Q.; Wang, G.; Tang, J.; Liu, X.; Shen, Y. Synthesis and evaluation of a paclitaxel-binding polymeric micelle for efficient breast cancer therapy. Sci. China Life Sci. 2018, 61, 436–447. [Google Scholar] [CrossRef]
- Zhao, Y.; Alakhova, D.Y.; Zhao, X.; Band, V.; Batrakova, E.V.; Kabanov, A.V. Eradication of cancer stem cells in triple negative breast cancer using doxorubicin/pluronic polymeric micelles. Nanomed. Nanotechnol. Biol. Med. 2019, 24, 102124. [Google Scholar] [CrossRef]
- Liu, T.; Romanova, S.; Wang, S.; Hyun, M.A.; Zhang, C.; Cohen, S.M.; Singh, R.K.; Bronich, T.K. Alendronate-Modified Polymeric Micelles for the Treatment of Breast Cancer Bone Metastasis. Mol. Pharm. 2019, 16, 2872–2883. [Google Scholar] [CrossRef]
- Sabra, S.A.; Elzoghby, A.O.; Sheweita, S.; Haroun, M.; Helmy, M.W.; Eldemellawy, M.A.; Xia, Y.; Goodale, D.; Allan, A.L.; Rohani, S. Self-assembled amphiphilic zein-lactoferrin micelles for tumor targeted co-delivery of rapamycin and wogonin to breast cancer. Eur. J. Pharm. Biopharm. 2018, 128, 156–169. [Google Scholar] [CrossRef]
- Wang, Y.; Liang, X.; Tong, R.; Yang, J.; Yang, Y.; Zhong, J.; Wu, Q.; Gong, C.; Yu, J.; Cai, L. Gambogic Acid-Loaded Polymeric Micelles for Improved Therapeutic Effect in Breast Cancer. J. Biomed. Nanotechnol. 2018, 14, 1695–1704. [Google Scholar] [CrossRef] [PubMed]
- Gregoriou, Y.; Gregoriou, G.; Yilmaz, V.; Kapnisis, K.; Prokopi, M.; Anayiotos, A.; Strati, K.; Dietis, N.; Constantinou, A.I.; Andreou, C. Resveratrol loaded polymeric micelles for theranostic targeting of breast cancer cells. Nanotheranostics 2021, 5, 113–124. [Google Scholar] [CrossRef]
- Gener, P.; Montero, S.; Monje, H.X.; Díaz-Riascos, Z.V.; Rafael, D.; Andrade, F.; Martínez-Trucharte, F.; González, P.; Seras-Franzoso, J.; Manzano-Muñoz, A.; et al. Zileuton™ loaded in polymer micelles effectively reduce breast cancer circulating tumor cells and intratumoral cancer stem cells. Nanomed. Nanotechnol. Biol. Med. 2019, 24, 102106. [Google Scholar] [CrossRef] [PubMed]
- Raveendran, R.; Chen, F.; Kent, B.; Stenzel, M.H. Estrone-Decorated Polyion Complex Micelles for Targeted Melittin Delivery to Hormone-Responsive Breast Cancer Cells. Biomacromolecules 2020, 21, 1222–1233. [Google Scholar] [CrossRef] [PubMed]
- Pawar, A.; Singh, S.; Rajalakshmi, S.; Shaikh, K.; Bothiraja, C. Development of fisetin-loaded folate functionalized pluronic micelles for breast cancer targeting. Artif. Cells Nanomed. Biotechnol. 2018, 46, 347–361. [Google Scholar] [CrossRef] [PubMed]
- Kesharwani, S.S.; Dachineni, R.; Bhat, G.J.; Tummala, H. Hydrophobically modified inulin-based micelles: Transport mechanisms and drug delivery applications for breast cancer. J. Drug Deliv. Sci. Technol. 2019, 54, 101254. [Google Scholar] [CrossRef]
- Xu, J.; Sun, J.; Ho, P.Y.; Luo, Z.; Ma, W.; Zhao, W.; Rathod, S.B.; Fernandez, C.A.; Venkataramanan, R.; Xie, W.; et al. Creatine based polymer for codelivery of bioengineered MicroRNA and chemodrugs against breast cancer lung metastasis. Biomaterials 2019, 210, 25–40. [Google Scholar] [CrossRef]
- Baidya, D.; Kushwaha, J.; Mahadik, K.; Patil, S. Chrysin-loaded folate conjugated PF127-F68 mixed micelles with enhanced oral bioavailability and anticancer activity against human breast cancer cells. Drug Dev. Ind. Pharm. 2019, 45, 852–860. [Google Scholar] [CrossRef]
- Cao, A.; Ma, P.; Yang, T.; Lan, Y.; Yu, S.; Liu, L.; Sun, Y.; Liu, Y. Multifunctionalized Micelles Facilitate Intracellular Doxorubicin Delivery for Reversing Multidrug Resistance of Breast Cancer. Mol. Pharm. 2019, 16, 2502–2510. [Google Scholar] [CrossRef]
- Mehnath, S.; Chitra, K.; Karthikeyan, K.; Jeyaraj, M. Localized delivery of active targeting micelles from nanofibers patch for effective breast cancer therapy. Int. J. Pharm. 2020, 584, 119412. [Google Scholar] [CrossRef]
- Lang, T.; Dong, X.; Zheng, Z.; Liu, Y.; Wang, G.; Yin, Q.; Li, Y. Tumor microenvironment-responsive docetaxel-loaded micelle combats metastatic breast cancer. Sci. Bull. 2018, 64, 91–100. [Google Scholar] [CrossRef] [Green Version]
- Chida, T.; Miura, Y.; Cabral, H.; Nomoto, T.; Kataoka, K.; Nishiyama, N. Epirubicin-loaded polymeric micelles effectively treat axillary lymph nodes metastasis of breast cancer through selective accumulation and pH-triggered drug release. J. Control. Release 2018, 292, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, X.; Wang, D.; Zou, Y.; Qu, X.; He, C.; Deng, Y.; Jin, Y.; Zhou, Y.; Zhou, Y.; et al. Concurrently suppressing multidrug resistance and metastasis of breast cancer by co-delivery of paclitaxel and honokiol with pH-sensitive polymeric micelles. Acta Biomater. 2017, 62, 144–156. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; De, G.; Yue, Q.; Ma, H.; Cheng, J.; Zhu, G.; Du, M.; Yi, H.; Zhao, Q.; Chen, Y. pH Responsive Polymer Micelles Enhances Inhibitory Efficacy on Metastasis of Murine Breast Cancer Cells. Front. Pharmacol. 2018, 9, 543. [Google Scholar] [CrossRef]
- Han, Y.; Pan, J.; Liang, N.; Gong, X.; Sun, S. A pH-Sensitive Polymeric Micellar System Based on Chitosan Derivative for Efficient Delivery of Paclitaxel. Int. J. Mol. Sci. 2021, 22, 6659. [Google Scholar] [CrossRef]
- Atale, S.S.; Dyawanapelly, S.; Jagtap, D.D.; Jain, R.; Dandekar, P. Understanding the nano-bio interactions using real-time surface plasmon resonance tool. Int. J. Biol. Macromol. 2018, 123, 97–107. [Google Scholar] [CrossRef]
- Palanikumar, L.; Al-Hosani, S.; Kalmouni, M.; Nguyen, V.P.; Ali, L.; Pasricha, R.; Barrera, F.N.; Magzoub, M. pH-responsive high stability polymeric nanoparticles for targeted delivery of anticancer therapeutics. Commun. Biol. 2020, 3, 95. [Google Scholar] [CrossRef]
- Song, C.; Yang, F.; Ji, R.; Lv, Y.; Wei, Z. Construction of a Drug Delivery System via pH-Responsive Polymeric Nanomicelles Containing Ferrocene for DOX Release and Enhancement of Therapeutic Effects. ACS Omega 2021, 6, 28242–28253. [Google Scholar] [CrossRef]
- Gulfam, M.; Matini, T.; Monteiro, P.F.; Riva, R.; Collins, H.; Spriggs, K.; Howdle, S.M.; Jérôme, C.; Alexander, C. Bioreducible cross-linked core polymer micelles enhance in vitro activity of methotrexate in breast cancer cells. Biomater. Sci. 2017, 5, 532–550. [Google Scholar] [CrossRef]
- Chai, Z.; Teng, C.; Yang, L.; Ren, L.; Yuan, Z.; Xu, S.; Cheng, M.; Wang, Y.; Yan, Z.; Qin, C.; et al. Doxorubicin delivered by redox-responsive Hyaluronic Acid–Ibuprofen prodrug micelles for treatment of metastatic breast cancer. Carbohydr. Polym. 2020, 245, 116527. [Google Scholar] [CrossRef]
- Feng, L.; Yan, S.; Zhu, Q.; Chen, J.; Deng, L.; Zheng, Y.; Xue, W.; Guo, R. Targeted multifunctional redox-sensitive micelle co-delivery of DNA and doxorubicin for the treatment of breast cancer. J. Mater. Chem. B 2018, 6, 3372–3386. [Google Scholar] [CrossRef] [PubMed]
- Ren, L.; Nie, J.; Wei, J.; Li, Y.; Yin, J.; Yang, X.; Chen, G. RGD-targeted redox responsive nano micelle: Co-loading docetaxel and indocyanine green to treat the tumor. Drug Deliv. 2021, 28, 2024–2032. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Tang, L.; Tu, C.; Song, Z.; Yin, Q.; Yin, L.; Zhang, Z.; Cheng, J. Redox-Responsive, Core-Cross-Linked Micelles Capable of On-Demand, Concurrent Drug Release and Structure Disassembly. Biomacromolecules 2013, 14, 3706–3712. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Ai, X.; Shang, L.; Zhong, L.; Ji, B.; Wu, C.; Zhuo, L.; He, Z.; Sun, J. Doxorubicin-Loaded Redox-Responsive Polymeric Nanomicelles Delivery System to Reverse Multidrug Resistance in Drug-Resistant Breast Cancer Cells. J. Nanosci. Nanotechnol. 2016, 16, 8424–8430. [Google Scholar] [CrossRef]
- He, B.; Hu, H.-Y.; Tan, T.; Wang, H.; Sun, K.-X.; Li, Y.-P.; Zhang, Z.-W. IR-780-loaded polymeric micelles enhance the efficacy of photothermal therapy in treating breast cancer lymphatic metastasis in mice. Acta Pharmacol. Sin. 2017, 39, 132–139. [Google Scholar] [CrossRef]
- Li, W.; Peng, J.; Yang, Q.; Chen, L.; Zhang, L.; Chen, X.; Qian, Z. α-Lipoic acid stabilized DTX/IR780 micelles for photoacoustic/fluorescence imaging guided photothermal therapy/chemotherapy of breast cancer. Biomater. Sci. 2018, 6, 1201–1216. [Google Scholar] [CrossRef]
- Su, Y.; Huang, N.; Chen, D.; Zhang, L.; Dong, X.; Sun, Y.; Zhu, X.; Zhang, F.; Gao, J.; Wang, Y.; et al. Successful in vivo hyperthermal therapy toward breast cancer by Chinese medicine shikonin-loaded thermosensitive micelle. Int. J. Nanomed. 2017, 12, 4019–4035. [Google Scholar] [CrossRef]
- Sang, M.M.; Liu, F.L.; Wang, Y.; Luo, R.J.; Huan, X.X.; Han, L.F.; Zhang, Z.T.; Feng, F.; Qu, W.; Liu, W.; et al. A novel redox/pH dual-responsive and hyaluronic acid-decorated multifunctional magnetic complex micelle for targeted gambogic acid delivery for the treatment of triple negative breast cancer. Drug Deliv. 2018, 25, 1846–1857. [Google Scholar] [CrossRef]
- Han, Z.; Gong, C.; Li, J.; Guo, H.; Chen, X.; Jin, Y.; Gao, S.; Tai, Z. Immunologically modified enzyme-responsive micelles regulate the tumor microenvironment for cancer immunotherapy. Mater. Today Bio 2021, 13, 100170. [Google Scholar] [CrossRef]
- Luo, L.; Xu, F.; Peng, H.; Luo, Y.; Tian, X.; Battaglia, G.; Zhang, H.; Gong, Q.; Gu, Z.; Luo, K. Stimuli-responsive polymeric prodrug-based nanomedicine delivering nifuroxazide and doxorubicin against primary breast cancer and pulmonary metastasis. J. Control. Release 2019, 318, 124–135. [Google Scholar] [CrossRef]
- Abdellatif, A.A.H.; Alsowinea, A.F. Approved and marketed nanoparticles for disease targeting and applications in COVID-19. Nanotechnol. Rev. 2021, 10, 1941–1977. [Google Scholar] [CrossRef]
- Zheng, X.; Xie, J.; Zhang, X.; Sun, W.; Zhao, H.; Li, Y.; Wang, C. An overview of polymeric nanomicelles in clinical trials and on the market. Chin. Chem. Lett. 2020, 32, 243–257. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. Evaluate the Efficacy and Safety of Genexol®-PM Compared to Genexol® in Recurrent or Metastatic Breast Cancer—Full Text View—ClinicalTrials.gov. 2012. Available online: https://clinicaltrials.gov/ct2/show/NCT00876486 (accessed on 1 July 2022).
- Fujiwara, Y.; Mukai, H.; Saeki, T.; Ro, J.; Lin, Y.-C.; Nagai, S.E.; Lee, K.S.; Watanabe, J.; Ohtani, S.; Kim, S.B.; et al. A multi-national, randomised, open-label, parallel, phase III non-inferiority study comparing NK105 and paclitaxel in metastatic or recurrent breast cancer patients. Br. J. Cancer 2019, 120, 475–480. [Google Scholar] [CrossRef]
- Lee, S.-W.; Yun, M.-H.; Jeong, S.W.; In, C.-H.; Kim, J.-Y.; Seo, M.-H.; Pai, C.-M.; Kim, S.-O. Development of docetaxel-loaded intravenous formulation, Nanoxel-PM™ using polymer-based delivery system. J. Control. Release 2011, 155, 262–271. [Google Scholar] [CrossRef]
- M.D. Anderson Cancer Center. Study of NC-4016 in Patients With Advanced Solid Tumors or Lymphoma. Clinicaltrials.Gov. Available online: https://clinicaltrials.gov/show/NCT03168035 (accessed on 1 July 2022).
- Sechi, M.; Sanna, V.; Pala, N. Targeted therapy using nanotechnology: Focus on cancer. Int. J. Nanomed. 2014, 9, 467–483. [Google Scholar] [CrossRef] [PubMed]
- Valle, J.W.; Armstrong, A.; Newman, C.; Alakhov, V.; Pietrzynski, G.; Brewer, J.; Campbell, S.; Corrie, P.; Rowinsky, E.K.; Ranson, M. A phase 2 study of SP1049C, doxorubicin in P-glycoprotein-targeting pluronics, in patients with advanced adenocarcinoma of the esophagus and gastroesophageal junction. Investig. New Drugs 2010, 29, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Chawla, S.P.; Goel, S.; Chow, W.; Braiteh, F.; Singh, A.S.; Olson, J.E.G.; Osada, A.; Bobe, I.; Riedel, R.F. A Phase 1b Dose Escalation Trial of NC-6300 (Nanoparticle Epirubicin) in Patients with Advanced Solid Tumors or Advanced, Metastatic, or Unresectable Soft-tissue Sarcoma. Clin. Cancer Res. 2020, 26, 4225–4232. [Google Scholar] [CrossRef]
- Home—ClinicalTrials.gov. Available online: http://clinicaltrials.gov/ (accessed on 10 August 2022).
- The ComprehensiveTable of Contents Headingsand Hierarchy. Available online: https://www.fda.gov/media/76444/download (accessed on 5 April 2022).
- ICH. CTD Triangle. ICH Harmonised Tripartite Guideline. 2010. Available online: https://admin.ich.org/sites/default/files/2021-02/CTD_triangle_color_Proofread.pdf (accessed on 5 April 2022).
- Tiwari, G.; Tiwari, R.; Sriwastawa, B.; Bhati, L.; Pandey, S.; Pandey, P.; Bannerjee, S.K. Drug delivery systems: An updated review. Int. J. Pharm. Investig. 2012, 2, 2–11. [Google Scholar] [CrossRef]
- Cho, H.; Lai, T.C.; Tomoda, K.; Kwon, G.S. Polymeric Micelles for Multi-Drug Delivery in Cancer. AAPS PharmSciTech 2014, 16, 10–20. [Google Scholar] [CrossRef]
- Yan, X.; Sedykh, A.; Wang, W.; Yan, B.; Zhu, H. Construction of a web-based nanomaterial database by big data curation and modeling friendly nanostructure annotations. Nat. Commun. 2020, 11, 2519. [Google Scholar] [CrossRef]
- Zhang, H.; Zhu, Y.; Shen, Y. Microfluidics for Cancer Nanomedicine: From Fabrication to Evaluation. Small 2018, 14, e1800360. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.V.; Ansari, M.H.D.; Rosenkranz, D.; Maharjan, R.S.; Kriegel, F.L.; Gandhi, K.; Kanase, A.; Singh, R.; Laux, P.; Luch, A. Artificial Intelligence and Machine Learning in Computational Nanotoxicology: Unlocking and Empowering Nanomedicine. Adv. Health Mater. 2020, 9, e1901862. [Google Scholar] [CrossRef] [PubMed]










| S.No. | Characterization Method | Property Measured | Reference |
|---|---|---|---|
| 1 | NMR | Nature of amphiphilic components | [42,43] |
| 2 | FTIR | Nature of amphiphilic components | [44] |
| 3 | DLS | PM size | [45,46] |
| 4 | AFM | PM morphology | [47] |
| 5 | Cryo-TEM | PM morphology | [48] |
| 6 | Surface tension | CMC of PM | [49] |
| 7 | Absorbance (Sudan III, 1,6-diphenyl-1,3,5-hexatriene (DPH)) | CMC of PM | [50] |
| 8 | Fluorescence (Pyrene, Coumarin-6, Nile Red, 1,6-diphenyl-1,3,5-hexatriene (DPH)) | CMC of PM | [51] |
| 9 | DLS | Surface charge of PM | [14,15] |
| 10 | FRET | Drug release, Protein corona formation, Stability in biological fluids | [52] |
| 11 | Chromatography Techniques | Drug release, Protein corona formation, Stability in biological fluids, Tissue concentrations | [14,15] |
| 12 | Symmetrical flow field-flow fractionation (AF4) | Stability in biological fluids (serum, cerebrospinal fluid, vitreous (eye)) | [14,15] |
| 13 | X-rays | PM size | [53] |
| S.No. | Polymer | Therapeutic Agent | Disease Type | Ref. |
|---|---|---|---|---|
| 1 | PLA-PEG | LAP, PTX | HER-2-negative, HER-2-positive | [67] |
| 2 | P(MeOx-b-BuOx-b-MeOx | PTX, alkylated cisplatin | Multidrug-resistant breast cancer | [68] |
| 3 | PEG-G3-PEITC | PTX | Breast cancer | [69] |
| 4 | Nonionic pluronic block copolymers | DOX | TNBC | [70] |
| 5 | PEG-PGlu-PPhA | DOX, ALN | Late-stage breast cancer bone metastasis | [71] |
| 6 | Zein with lactoferrin protein | RAP, WOG | Breast cancer | [72] |
| 7 | MPEG-PCL | GA | Breast cancer | [73] |
| 8 | Pluronic F127 block copolymer and Vitamin E-TPGS | RES | Breast cancer | [74] |
| 9 | Pluronic F127 block copolymer | Zileuton | TNBC | [75] |
| 10 | POEGMEMA-PtBuMA | Estrogen | Hormone-responsive breast cancer | [76] |
| 11 | PF-FA | FS | Breast cancer | [77] |
| 12 | INT | DOX, PTX | Breast cancer | [78] |
| 13 | POEG-VBC | DOX, tRNA-mir-34a | Metastatic breast cancer | [79] |
| 14 | Pluronic PF127-pluronic F68 with folate | CH | Breast cancer | [80] |
| 15 | Pluronic F127 with HA-DOCA-His-PF | DOX | Multidrug-resistant breast cancer | [81] |
| 16 | Cholic acid with poly (bis (carboxyphenoxy) phosphazene | PTX | Breast cancer | [82] |
| 17 | BD-PEI and BD-peptide-PEG | DTX | Metastatic breast cancer | [83] |
| 18 | PEG-poly(amino acid) block | EPI | ALNM of TNBC | [84] |
| 19 | PEOz-PLA | PTX, HNK | Multidrug-resistant and metastatic breast cancer | [85] |
| 20 | PELA-PBAE | PTX | Metastatic breast cancer | [86] |
| 21 | mPEG-CS-Hz-CH | PTX | Breast cancer | [87] |
| 22 | PLGA-BS-ATRAM | DOX-TPP | Breast cancer | [89] |
| 23 | mPEG-Dlabile-PCL-Fc | DOX | Breast cancer | [90] |
| 24 | mPEG-PCL | MTX | Breast cancer | [60] |
| 25 | mPEG-b-poly(εCL-co-αN3εCL | MTX | Breast cancer | [91] |
| 26 | HA-ss-BF | DOX | Breast cancer | [92] |
| 27 | PEG-PEI-PCL-S–S-PCL-PEG (PPPT) | DOX | Breast cancer | [93] |
| 28 | PEG, PPE | DOX | Multidrug-resistant breast cancer | [64] |
| 29 | PEG, polycaprolactone, RGD peptide | DOX, ICG | Breast cancer | [94] |
| 30 | poly(Tyrosine(alkynyl)-OCA), monomethoxy poly(ethylene glycol)-b-poly(Tyrosine(alkynyl)-OCA) | Camptothecin | Breast cancer | [95] |
| 31 | P5kSSLV | DOX | Multidrug-resistant breast cancer | [96] |
| 32 | DSPE-PEG2000 | IR780 | Breast cancer lymphatic metastasis | [97] |
| 33 | mPEG-PCL copolymer | DTX, IR780 | Breast cancer | [98] |
| 34 | Poly(N-isopropylacrymaide) and polylactic acid | Shikonin | Breast cancer | [99] |
| 35 | Zein-lactoferrin copolymer, Fe3O4 nanoparticles | DAS | TNBC | [52] |
| 36 | Folate-conjugated dextran/retinoic acid copolymer, oleic acid | DOX | Breast cancer | [65] |
| 37 | mPEG-b-PHEP copolymer, iron oxide nanocubes | Emodin | Breast cancer | [66] |
| 38 | CSO-SS-Hex, HA, Speg copolymer, Fe3O4 nanoparticles | GA | TNBC | [100] |
| 39 | SHR, PLGLAGK-Mal, coated with anti-CD28 antibody | CH223191 | Breast cancer | [101] |
| 40 | HPMA, OEGMA, GFLG peptide | DOX, Nifuroxazide | Metastatic breast cancer | [102] |
| S.No. | PM | Excipient | Drug Release | Therapeutic Agent | Disease Type | NCT Number/ Clinical Phase (Status) |
|---|---|---|---|---|---|---|
| 1 | Genexol-PM | PEG-b-PLA | Controlled release with no specific triggering mechanism | PTX | Breast Cancer | NCT00876486/Phase III (Completed) |
| PTX, PM with DOX | Metastatic Breast Cancer | NCT01784120/Phase II (Unknown) | ||||
| PTX | Recurrent Breast Cancer | NCT00912639/Phase IV (Unknown) | ||||
| PTX | Breast Cancer | NCT01169870/Phase II (Withdrawn) | ||||
| PTX, PM with Carboplatin | Ovarian Cancer | NCT00877253/ Phase I (Completed) | ||||
| 2 | NK105 | PEG-b-PPBA | Sustained release equivalent dose of <1 μg mL−1 with no specific triggering mechanism | PTX | Metastatic or Recurrent Breast Cancer | NCT01644890/ Phase III (Completed) |
| 3 | Nanoxel-PM | PEG-PDLLA | DTX was released immediately from micelle to bind to the plasma proteins. Pharmacokinetics equivalent to Taxotere® | DTX | Breast Cancer, Non-small Cell Lung Cancer | NCT04066335/(Recruiting) |
| No specific triggering mechanism | PTX | Advanced Breast Cancer | NCT00915369/ Phase I(Unknown) | |||
| 4 | Docecal | XR-17 block polymer | Controlled release with no specific triggering mechanism | DTX | Breast Cancer | 2012-005161-12 (EudraCT)/Phase III (results not published) |
| 5 | NK012 | PEG-b-poly(glutamate) | Releases SN-38 under neutral conditions even without the presence of a hydrolytic enzyme | SN-38 an active metabolite of irinotecan hydrochloride (CPT-11) | Triple Negative Breast Cancer | NCT00951054/Phase II/Completed |
| Releases SN-38 under neutral conditions | NK012 and Carboplatin | Metastatic Triple Negative Breast Cancer | NCT01238952/Phase 1/Completed |
| S.No. | Quality Attribute | Target Terms | Criticality | Justification |
|---|---|---|---|---|
| 1 | Description | Examples: A sterile, translucent red/green/yellow micellar dispersion | Yes |
|
| 2 | pH | Target pH | Yes |
|
| 3 | Osmolality (mOsm/kg) | Specific mOsm/kg of water | Yes |
|
| 4 | Average particle size | Particle size in nanometers (nm) | Yes |
|
| 5 | Particle size distribution | Particle size in nanometers (nm) Population distribution (D,10), (D,50), (D,90) | Yes |
|
| 6 | Identification (API) | Positive for specific drug substance | Yes |
|
| 7 | Identification of polymers | Positive for specific polymers (PLGA, PLA, etc.) | Yes |
|
| 8 | Assay (Dug substance) | Each mL contains a specific amount of labeled drug | Yes |
|
| 9 | Polymer content | Each mL contains the amount of polymer | Yes |
|
| 10 | Percentage of the entrapped drug | Should comply with the qualified limits. Example: Not less than 95.0% of the assay amount. | Yes |
|
| 11 | Percentage of Unentrapped drug | Should comply with the qualified limits Example: Not more than 5.0%. | Yes | |
| 12 | Ion content (ppm) | Total Ion content (ppm) (Ammonium, Iron, etc.). | Yes |
|
| 14 | Related substances (API) | Should comply with the qualified limits. | Yes |
|
| 15 | Related substances (polymers) | Should comply with the qualified limits. | Yes | |
| 16 | Drug release | Drug release profile. | Yes |
|
| 17 | Visible Particulate Matter Particulate Matter (sub visible)–Microscopic method | Free from visible particles. Not more than 3000 average number of particles should be greater than or equal to 10 µm. Not more than 300 average number of particles should be greater than or equal to 25 µm. | Yes |
|
| 18 | Uniformity of dosage units | Content uniformity method. | Yes |
|
| 20 | Zeta Potential (by Zeta Sizer) | Defined by innovator. | Yes |
|
| 21 | Sterility | The product should be sterile. | Yes |
|
| 22 | Bacterial Endotoxin Test | Specific limits. | Yes |
|
| 23 | Residual solvents | Based on process optimization. | Yes |
|
| S.No. | Unit Operation | Process Parameter |
|---|---|---|
| 1 | Polymer dissolution | Process Temperature |
| Stirring speed | ||
| Stirring Time | ||
| 2 | Preparation of micelles | Process Temperature |
| Homogenization Speed | ||
| Homogenization Time | ||
| 3 | Extrusion process | Process Temperature |
| Number of Cycles | ||
| Membrane Pore Size | ||
| Pump Speed | ||
| 4 | Dia-filtration (Removal of ions) | Conductivity of Filtrate |
| Process Temperature | ||
| Pump Speed | ||
| 5 | Drug Loading | Process Temperature |
| Stirring Speed | ||
| Incubation Time | ||
| 6 | Filtration | Filter Flushes |
| Filter Integrity | ||
| Filtration Time | ||
| Filtration Pressure |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Junnuthula, V.; Kolimi, P.; Nyavanandi, D.; Sampathi, S.; Vora, L.K.; Dyawanapelly, S. Polymeric Micelles for Breast Cancer Therapy: Recent Updates, Clinical Translation and Regulatory Considerations. Pharmaceutics 2022, 14, 1860. https://doi.org/10.3390/pharmaceutics14091860
Junnuthula V, Kolimi P, Nyavanandi D, Sampathi S, Vora LK, Dyawanapelly S. Polymeric Micelles for Breast Cancer Therapy: Recent Updates, Clinical Translation and Regulatory Considerations. Pharmaceutics. 2022; 14(9):1860. https://doi.org/10.3390/pharmaceutics14091860
Chicago/Turabian StyleJunnuthula, Vijayabhaskarreddy, Praveen Kolimi, Dinesh Nyavanandi, Sunitha Sampathi, Lalitkumar K. Vora, and Sathish Dyawanapelly. 2022. "Polymeric Micelles for Breast Cancer Therapy: Recent Updates, Clinical Translation and Regulatory Considerations" Pharmaceutics 14, no. 9: 1860. https://doi.org/10.3390/pharmaceutics14091860