Potential Activity of Abrantes Pollen Extract: Biochemical and Cellular Model Studies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemical Reagents
2.2. Pollen Samples
2.3. Pollen Analysis
2.4. Pollen Extract Preparation
2.5. Identification of Phenolic Compounds via HPLC-DAD-ESI/MSn
2.6. Quantification of Phenolic Compounds via HPLC-DAD
2.7. Antioxidant Capacity Experiments
2.7.1. 1,1-Diphenyl-2-Picrylhydrazyl Radical (DPPH●) Assay
2.7.2. Nitric Oxide Radical (●NO) Assay
2.7.3. Superoxide Radical (O2●−) Assay
2.8. α-Glucosidase Inhibitory Activity
2.9. Biological Activity on Cellular Models
2.9.1. Isolation of Human Erythrocytes
2.9.2. Peroxyl Radical (ROO●)-Induced Oxidative Damage in Human Erythrocytes
2.9.3. Inhibition of Hemoglobin Oxidation
2.9.4. Inhibition of Lipid Peroxidation
2.9.5. Inhibition of Hemolysis
2.10. Cancer Cell Models
2.10.1. Cell Culture Conditions and Treatments
2.10.2. MTT Assay
2.10.3. Membrane Integrity Assay
2.11. Statistical Analysis
3. Results and Discussion
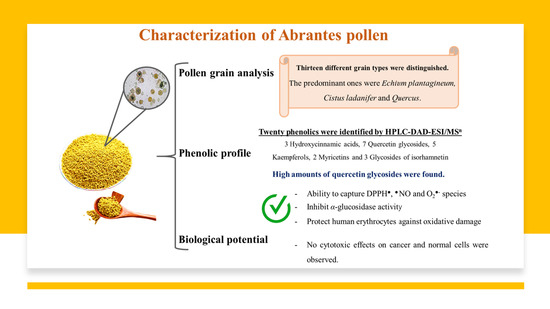
3.1. Pollen Composition
3.2. Phenolic Characterization
3.2.1. Hydroxycinnamic Acids
3.2.2. Flavonol Glycosides
3.3. Antioxidant Capacity
3.4. Antidiabetic Capacity
3.5. Protective Effects on Human Erythrocytes
3.6. Effect of Pollen Extracts in Mitochondrial Activity and Membrane Integrity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gonçalves, A.C.; Bento, C.; Nunes, A.R.; Simões, M.; Alves, G.; Silva, L.R. Multitarget protection of Pterospartum tridentatum phenolic-rich extracts against a wide range of free radical species, antidiabetic activity and effects on human colon carcinoma (Caco-2) cells. J. Food Sci. 2020, 85, 4377–4388. [Google Scholar] [CrossRef] [PubMed]
- Thakur, M.; Nanda, V. Composition and functionality of bee pollen: A review. Trends Food Sci. Technol. 2020, 98, 82–106. [Google Scholar] [CrossRef]
- Moita, E.; Sousa, C.; Andrade, P.B.; Fernandes, F.; Pinho, B.R.; Silva, L.R.; Valentão, P. Effects of Echium plantagineum L. bee pollen on basophil degranulation: Relationship with metabolic profile. Molecules 2014, 19, 10635–10649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chantarudee, A.; Phuwapraisirisan, P.; Kimura, K.; Okuyama, M.; Mori, H.; Kimura, A.; Chanchao, C. Chemical constituents and free radical scavenging activity of corn pollen collected from Apis mellifera hives compared to floral corn pollen at Nan, Thailand. BMC Complementary Altern. Med. 2012, 12, 45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silva, L.R.; Gonçalves, A.C.; Nunes, A.R.; Alves, G. Authentication of honeys from Caramulo region (Portugal): Pollen spectrum, physicochemical characteristics, mineral content, and phenolic profile. J. Food Sci. 2020, 85, 374–385. [Google Scholar] [CrossRef]
- Kaškonienė, V.; Ruočkuvienė, G.; Kaškonas, P.; Akuneca, I.; Maruška, A. Chemometric analysis of bee pollen based on volatile and phenolic compound compositions and antioxidant properties. Food Anal. Methods 2015, 8, 1150–1163. [Google Scholar] [CrossRef]
- Adaškevičiūtė, V.; Kaškonienė, V.; Kaškonas, P.; Barčauskaitė, K.; Maruška, A. Comparison of physicochemical properties of bee pollen with other bee products. Biomolecules 2019, 9, 819. [Google Scholar] [CrossRef] [Green Version]
- de Florio Almeida, J.; dos Reis, A.S.; Heldt, L.F.; Pereira, D.; Bianchin, M.; de Moura, C.; Plata-Oviedo, M.V.; Haminiuk, C.W.; Ribeiro, I.S.; da Luz, C.F.; et al. Lyophilized bee pollen extract: A natural antioxidant source to prevent lipid oxidation in refrigerated sausages. LWT-Food Sci. Technol. 2017, 76, 299–305. [Google Scholar] [CrossRef]
- Moreira, R.; Fernandes, F.; Valentão, P.; Pereira, D.M.; Andrade, P.B. Echium plantagineum L. honey: Search of pyrrolizidine alkaloids and polyphenols, anti-inflammatory potential and cytotoxicity. Food Chem. 2020, 328, 127169. [Google Scholar] [CrossRef]
- Silva, L.R.; Sousa, A.; Taveira, M. Characterization of Portuguese honey from Castelo Branco region according to their pollen spectrum, physicochemical characteristics and mineral contents. J. Food Sci. Technol. 2017, 54, 2551–2561. [Google Scholar] [CrossRef]
- Louveaux, J.; Maurizio, A.; Vorwohl, G. Methods of Melissopalynology. Bee World 1978, 59, 139–157. [Google Scholar] [CrossRef]
- Gonçalves, A.C.; Campos, G.; Alves, G.; Garcia-Viguera, C.; Moreno, D.A.; Silva, L.R. Physical and phytochemical composition of 23 Portuguese sweet cherries as conditioned by variety (or genotype). Food Chem. 2021, 335, 127637. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Giampieri, F.; González-Paramás, A.M.; Damiani, E.; Astolfi, P.; Martinez-Sanchez, G.; Bompadre, S.; Quiles, J.L.; Santos-Buelga, C.; Battino, M. Phenolics from monofloral honeys protect human erythrocyte membranes against oxidative damage. Food Chem. Toxicol. 2012, 50, 1508–1516. [Google Scholar] [CrossRef] [PubMed]
- De-Melo, A.A.; Estevinho, L.M.; Moreira, M.M.; Delerue-Matos, C.; Freitas, A.D.; Barth, O.M.; Almeida-Muradian, L.B. Phenolic profile by HPLC-MS, biological potential, and nutritional value of a promising food: Monofloral bee pollen. J. Food Biochem. 2018, 42, e12536. [Google Scholar] [CrossRef] [Green Version]
- Tang, J.; Dunshea, F.R.; Suleria, H.A.R. LC-ESI-QTOF/MS characterization of phenolic compounds from medicinal plants (Hops and Juniper berries) and their antioxidant activity. Foods 2020, 9, 7. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Liu, R.; Lu, Q. Separation and characterization of phenolamines and flavonoids from Rape bee pollen, and comparison of their antioxidant activities and protective effects. Molecules 2020, 25, 1264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silva, L.R.; Teixeira, R. Phenolic profile and biological potential of Endopleura uchi extracts. Asian Pac. J. Trop. Med. 2015, 8, 889–897. [Google Scholar] [CrossRef] [Green Version]
- Chisté, R.C.; Freitas, M.; Mercadante, A.Z.; Fernandes, E. Carotenoids inhibit lipid peroxidation and hemoglobin oxidation, but not the depletion of glutathione induced by ROS in human erythrocytes. Life Sci. 2014, 99, 52–60. [Google Scholar] [CrossRef]
- Jesus, F.; Gonçalves, A.C.; Alves, G.; Silva, L.R. Exploring the phenolic profile, antioxidant, antidiabetic and anti-hemolytic potential of Prunus avium vegetal parts. Food Res. Int. 2018, 116, 600–610. [Google Scholar] [CrossRef]
- Forbes-Hernández, T.Y.; Gasparrini, M.; Afrin, S.; Cianciosi, D.; González-Paramás, A.M.; Santos-Buelga, C.; Mezzetti, B.; Quiles, J.L.; Battino, M.; Giampieri, F.; et al. Strawberry (cv. Romina) methanolic extract and anthocyanin-enriched fraction improve lipid profile and antioxidant status in HepG2 cells. Int. J. Mol. Sci. 2017, 18, 1149. [Google Scholar] [CrossRef] [Green Version]
- Gonçalves, A.C.; Rodrigues, M.; Santos, A.O.; Alves, G.; Silva, L.R. Antioxidant status, antidiabetic properties and effects on Caco-2 cells of colored and non-colored enriched extracts of sweet cherry fruits. Nutrients 2018, 10, 1688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Estevinho, L.M.; Feás, X.; Seijas, J.A.; Pilar Vázquez-Tato, M. Organic honey from Trás-Os-Montes region (Portugal): Chemical, palynological, microbiological and bioactive compounds characterization. Food Chem. Toxicol. 2012, 50, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.R.; Videira, R.; Monteiro, A.P.; Valentão, P.; Andrade, P.B. Honey from Luso region (Portugal): Physicochemical characteristics and mineral contents. Microchem. J. 2009, 93, 73–77. [Google Scholar] [CrossRef]
- Karabagias, I.K.; Maia, M.; Karabagias, V.K.; Gatzias, I.; Badeka, A.V. Quality and origin characterisation of Portuguese, Greek, Oceanian, and Asian honey, based on poly-parametric analysis hand in hand with dimension reduction and classification techniques. Eur. Food Res. Technol. 2020, 246, 987–1006. [Google Scholar] [CrossRef]
- Escuredo, O.; Silva, L.R.; Valentão, P.; Seijo, M.C.; Andrade, P.B. Assessing Rubus honey value: Pollen and phenolic compounds content and antibacterial capacity. Food Chem. 2012, 130, 671–678. [Google Scholar] [CrossRef]
- Bento, C.; Gonçalves, A.C.; Silva, B.; Silva, L.R. Assessing the phenolic profile, antioxidant, antidiabetic and protective effects against oxidative damage in human erythrocytes of peaches from Fundão. J. Funct. Foods 2018, 43, 224–233. [Google Scholar] [CrossRef]
- Bento, C.; Gonçalves, A.C.; Jesus, F.; Simões, M.; Silva, L.R. Phenolic compounds: Sources, properties and applications. In Bioactive Compounds: Sources, Properties and Applications; Porter, R., Parker, N., Eds.; Nova Science Publishers: New York, NY, USA, 2017; pp. 271–299. [Google Scholar]
- Sousa, C.; Moita, E.; Valentão, P.; Fernandes, F.; Monteiro, P.; Andrade, P.B. Effects of colored and noncolored phenolics of Echium plantagineum L. bee pollen in Caco-2 cells under oxidative stress induced by tert -butyl hydroperoxide. J. Agric. Food Chem. 2015, 63, 2083–2091. [Google Scholar] [CrossRef]
- Oyarzún, J.E.; Andia, M.E.; Uribe, S.; Núñez Pizarro, P.; Núñez, G.; Montenegro, G.; Bridi, R. Honeybee pollen extracts reduce oxidative stress and steatosis in hepatic cells. Molecules 2020, 26, 6. [Google Scholar] [CrossRef] [PubMed]
- Morais, M.; Moreira, L.; Feás, X.; Estevinho, L.M. Honeybee-collected pollen from five Portuguese Natural Parks: Palynological origin, phenolic content, antioxidant properties and antimicrobial activity. Food Chem. Toxicol. 2011, 49, 1096–1101. [Google Scholar] [CrossRef] [Green Version]
- Thakur, M.; Nanda, V. Screening of Indian bee pollen based on antioxidant properties and polyphenolic composition using UHPLC-DAD-MS/MS: A multivariate analysis and ANN based approach. Food Res. Int. 2021, 140, 110041. [Google Scholar] [CrossRef]
- Duarte, A.W.F.; Vasconcelos, M.R.D.S.; Oda-Souza, M.; Oliveira, F.F.; López, A.M.Q. Honey and bee pollen produced by Meliponini (Apidae) in Alagoas, brazil: Multivariate analysis of physicochemical and antioxidant profiles. Food Sci. Technol. 2018, 38, 493–503. [Google Scholar] [CrossRef] [Green Version]
- Stagos, D.; Soulitsiotis, N.; Tsadila, C.; Papaeconomou, S.; Arvanitis, C.; Ntontos, A.; Karkanta, F.; Adamou-Androulaki, S.; Petrotos, K.; Spandidos, D.A.; et al. Antibacterial and antioxidant activity of different types of honey derived from Mount Olympus in Greece. Int. J. Mol. Med. 2018, 42, 726–734. [Google Scholar] [CrossRef] [Green Version]
- Daudu, O.M. Bee pollen extracts as potential antioxidants and inhibitors of α-amylase and α-glucosidase enzymes-In vitro assessment. J. Apic. Sci. 2019, 63, 315–325. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Wang, G.; Beta, T.; Dong, J. Inhibitory properties of aqueous ethanol extracts of propolis on alpha-glucosidase. Evid. -Based Complementary Altern. Med. 2015, 2015, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hassan, A.K.; El-Kotby, D.A.; Tawfik, M.M.; Badr, R.E.; Bahgat, I.M. Antidiabetic effect of the Egyptian honey bee (Apis mellifera) venom in alloxan-induced diabetic rats. J. Basic Appl. Zool. 2019, 80, 58. [Google Scholar] [CrossRef] [Green Version]
- Enginyurt, O.; Cakir, L.; Karatas, A.; Cankaya, S.; Kaya, Y.; Handan Tugcu, H.; Dogan Iscanli, M.; Cankaya, N.; Yarilgac, S. The role of pure honey in the treatment of diabetes mellitus. Biomed. Res. 2017, 28, 3305–3312. [Google Scholar]
- Tadera, K.; Minami, Y.; Takamatsu, K.; Matsuoka, T. Inhibition of α-glucosidase and α-amylase by flavonoids. J. Nutr. Sci. Vitaminol. 2006, 52, 149–153. [Google Scholar] [CrossRef] [Green Version]
- Blasa, M.; Candiracci, M.; Accorsi, A.; Piacentini, M.P.; Piatti, E. Honey flavonoids as protection agents against oxidative damage to human red blood cells. Food Chem. 2007, 104, 1635–1640. [Google Scholar] [CrossRef]
- Barbieri, D.; Gabriele, M.; Summa, M.; Colosimo, R.; Leonardi, D.; Domenici, V.; Pucci, L. Antioxidant, nutraceutical properties, and fluorescence spectral profiles of bee pollen samples from different botanical origins. Antioxidants 2020, 9, 1001. [Google Scholar] [CrossRef] [PubMed]
- Peyrat-Maillard, M.N.; Cuvelier, M.E.; Berset, C. Antioxidant activity of phenolic compounds in 2,2′-Azobis (2-amidinopropane) Dihydrochloride (AAPH)-induced oxidation: Synergistic and antagonistic effects. J. Am. Oil Chem. Soc. 2003, 80, 1007–1012. [Google Scholar] [CrossRef]
- Kitagawa, S.; Sakamoto, H.; Tano, H. Inhibitory effects of flavonoids on free radical-induced hemolysis and their oxidative effects on hemoglobin. Chem. Pharm. Bull. 2004, 52, 999–1001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, I.W.; Kuo, S.M. Flavonoid structure affects the inhibition of lipid peroxidation in Caco-2 intestinal cells at physiological concentrations. J. Nutr. 2003, 133, 2184–2187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laranjinha, J.; Vieira, O.; Almeida, L.; Madeira, V. Inhibition of metmyoglobin/H2O2-dependent low density lipoprotein lipid peroxidation by naturally occurring phenolic acids. Biochem. Pharmacol. 1996, 51, 395–402. [Google Scholar] [CrossRef]



| Peak | Compounds Identification | HPLC-DAD-ESI-MSn Data | Quantification | |||
|---|---|---|---|---|---|---|
| Rt (min) | λmax (nm) | Molecular Ion [M-H] (m/z) | Fragments MS/MS (m/z) | |||
| 1 | Caffeoyl di-hexoside | 10.0 | 320 | 635 | 341, 179 | nq |
| 2 | Coumaroyl hexose | 13.8 | 320 | 325 | 145, 163, 205, 235 | nq |
| 3 | Caffeoyl hexose | 14.0 | 320 | 341 | 179, 135 | 0.056 ± 0.0057 |
| 4 | Quercetin 7-glucoside-3-O-rutinoside | 24.0 | 350 | 771 | 609, 301 | 0.35 ± 0.020 |
| 5 | Kaempferol di-hexoside | 24.3 | 350 | 609 | 447, 285 | nq |
| 6 | Myricetin rhamno-hexoside | 25.0 | 350 | 625 | 316, 271, 287 | nq |
| 7 | Quercetin 3-O-rutinoside | 25.9 | 350 | 609 | 271, 301 | 0.76 ± 0.037 |
| 8 | Kaempferol 3-O-rutinoside-O-hexoside | 26.2 | 350 | 755 | 593, 447, 285 | nq |
| 9 | Quercetin derivative 1 | 26.3 | 350 | 639 | 314, 301, 150 | 0.49 ± 0.031 |
| 10 | Myricetin derivative | 26.4 | 350 | 521 | 316; 271 | nq |
| 11 | Isorhamnetin 3-O-rutinoside 1 | 26.8 | 350 | 623 | 315 | nq |
| 12 | Kaempferol 3-O-rutinoside | 27.3 | 350 | 447 | 285, 256 | nq |
| 13 | Quercetin 3-O-glucoside | 28.4 | 350 | 463 | 300/301, 271 | nq |
| 14 | Isorhamnetin 3-O-rutinoside 2 | 28.5 | 350 | 623 | 315 | nq |
| 15 | Quercetin derivative 2 | 28.6 | 350 | 609 | 315, 300, 271 | nq |
| 16 | Quercetin acetyl rhamnoside | 29.3 | 350 | 505 | 463, 301 | 1.33 ± 0.022 |
| 17 | Isorhamnetin acetyl hexoside | 31.7 | 350 | 519 | 315 | 0.82 ± 0.013 |
| 18 | Kaempferol acetyl hexoside | 31.9 | 350 | 447 | 235 | nq |
| 19 | Kaempferol hexoside | 32.6 | 350 | 489 | 447, 285 | nq |
| 20 | Quercetin hexoside | 35.0 | 350 | 463 | 301 | 0.36 ± 0.032 |
| Σ | 4.17 | |||||
| DPPH● | ●NO | O2●− | α-Glucosidase | Haemoglobin Oxidation | Haemolysis | Lipid Peroxidation | |
|---|---|---|---|---|---|---|---|
| Extract | 695.99 ± 1.69 | 1115.28 ± 5.32 | 449.04 ± 2.01 | 1192.71 ± 8.14 | 311.50 ± 1.37 | 103.48 ± 2.23 | 277.03 ± 2.52 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonçalves, A.C.; Lahlou, R.A.; Alves, G.; Garcia-Viguera, C.; Moreno, D.A.; Silva, L.R. Potential Activity of Abrantes Pollen Extract: Biochemical and Cellular Model Studies. Foods 2021, 10, 2804. https://doi.org/10.3390/foods10112804
Gonçalves AC, Lahlou RA, Alves G, Garcia-Viguera C, Moreno DA, Silva LR. Potential Activity of Abrantes Pollen Extract: Biochemical and Cellular Model Studies. Foods. 2021; 10(11):2804. https://doi.org/10.3390/foods10112804
Chicago/Turabian StyleGonçalves, Ana C., Radhia Aitfella Lahlou, Gilberto Alves, Cristina Garcia-Viguera, Diego A. Moreno, and Luís R. Silva. 2021. "Potential Activity of Abrantes Pollen Extract: Biochemical and Cellular Model Studies" Foods 10, no. 11: 2804. https://doi.org/10.3390/foods10112804