The Role of Hypoxia-Associated Long Non-Coding RNAs in Breast Cancer
Abstract
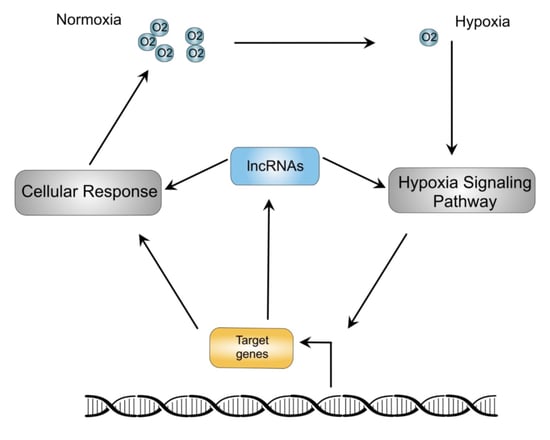
:1. Introduction
2. Hypoxia Signaling Pathway
2.1. Canonical Hypoxia Signaling Pathway
2.2. The Hypoxia Signaling Pathway and Breast Cancer
3. Hypoxia-Associated Long Non-Coding RNAs as Regulators of Breast Cancer
3.1. Growth
3.2. Migration, Invasion and Metastasis
3.3. Glycolysis
3.4. Stemness
4. Hypoxia-Associated lncRNAs as Breast Cancer Clinical Molecular Markers
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Pfeiffer, R.M.; Webb-Vargas, Y.; Wheeler, W.; Gail, M.H. Proportion of U.S. Trends in Breast Cancer Incidence Attributable to Long-term Changes in Risk Factor Distributions. Cancer Epidemiol. Biomark. Prev. 2018, 27, 1214–1222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Thomlinson, R.H.; Gray, L.H. The histological structure of some human lung cancers and the possible implications for radiotherapy. Br. J. Cancer 1955, 9, 539–549. [Google Scholar] [CrossRef] [Green Version]
- Ivan, M.; Fishel, M.L.; Tudoran, O.M.; Pollok, K.E.; Wu, X.; Smith, P.J. Hypoxia signaling: Challenges and opportunities for cancer therapy. Semin. Cancer Biol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Graham, A.M.; Presnell, J.S. Hypoxia Inducible Factor (HIF) transcription factor family expansion, diversification, divergence and selection in eukaryotes. PLoS ONE 2017, 12, e0179545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Myllyharju, J. Prolyl 4-hydroxylases, master regulators of the hypoxia response. Acta Physiol. 2013, 208, 148–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, M.; Lun, J.; Zhang, H.; Zhu, L.; Zhang, G.; Fang, J. The non-canonical functions of HIF prolyl hydroxylases and their dual roles in cancer. Int. J. Biochem. Cell Biol. 2021, 135, 105982. [Google Scholar] [CrossRef]
- Ortiz-Barahona, A.; Villar, D.; Pescador, N.; Amigo, J.; del Peso, L. Genome-wide identification of hypoxia-inducible factor binding sites and target genes by a probabilistic model integrating transcription-profiling data and in silico binding site prediction. Nucleic Acids Res. 2010, 38, 2332–2345. [Google Scholar] [CrossRef]
- Hockel, M.; Vaupel, P. Tumor hypoxia: Definitions and current clinical, biologic, and molecular aspects. J. Natl. Cancer Inst. 2001, 93, 266–276. [Google Scholar] [CrossRef] [Green Version]
- Vaupel, P.; Briest, S.; Hockel, M. Hypoxia in breast cancer: Pathogenesis, characterization and biological/therapeutic implications. Wien. Med. Wochenschr. 2002, 152, 334–342. [Google Scholar] [CrossRef]
- Dales, J.P.; Garcia, S.; Meunier-Carpentier, S.; Andrac-Meyer, L.; Haddad, O.; Lavaut, M.N.; Allasia, C.; Bonnier, P.; Charpin, C. Overexpression of hypoxia-inducible factor HIF-1alpha predicts early relapse in breast cancer: Retrospective study in a series of 745 patients. Int. J. Cancer 2005, 116, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Bos, R.; Zhong, H.; Hanrahan, C.F.; Mommers, E.C.; Semenza, G.L.; Pinedo, H.M.; Abeloff, M.D.; Simons, J.W.; van Diest, P.J.; van der Wall, E. Levels of hypoxia-inducible factor-1 alpha during breast carcinogenesis. J. Natl. Cancer Inst. 2001, 93, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Munoz, L.; Espinosa, M.; Quintanar-Jurado, V.; Hidalgo, A.; Melendez-Zajgla, J.; Maldonado, V. Paradoxial changes in the expression of estrogen receptor alpha in breast cancer multicellular spheroids. Tissue Cell 2010, 42, 334–337. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Galindo, L.; Melendez-Zajgla, J.; Pacheco-Fernandez, T.; Rodriguez-Sosa, M.; Mandujano-Tinoco, E.A.; Vazquez-Santillan, K.; Castro-Oropeza, R.; Lizarraga, F.; Sanchez-Lopez, J.M.; Maldonado, V. Changes in the transcriptome profile of breast cancer cells grown as spheroids. Biochem. Biophys. Res. Commun. 2019, 516, 1258–1264. [Google Scholar] [CrossRef]
- Hashimoto, T.; Shibasaki, F. Hypoxia-inducible factor as an angiogenic master switch. Front. Pediatr. 2015, 3, 33. [Google Scholar] [CrossRef] [Green Version]
- Palazon, A.; Tyrakis, P.A.; Macias, D.; Velica, P.; Rundqvist, H.; Fitzpatrick, S.; Vojnovic, N.; Phan, A.T.; Loman, N.; Hedenfalk, I.; et al. An HIF-1alpha/VEGF-A Axis in Cytotoxic T Cells Regulates Tumor Progression. Cancer Cell 2017, 32, 669–683.e5. [Google Scholar] [CrossRef] [Green Version]
- Incio, J.; Ligibel, J.A.; McManus, D.T.; Suboj, P.; Jung, K.; Kawaguchi, K.; Pinter, M.; Babykutty, S.; Chin, S.M.; Vardam, T.D.; et al. Obesity promotes resistance to anti-VEGF therapy in breast cancer by up-regulating IL-6 and potentially FGF-2. Sci. Transl. Med. 2018, 10, eaag0945. [Google Scholar] [CrossRef] [Green Version]
- Semenza, G.L. The hypoxic tumor microenvironment: A driving force for breast cancer progression. Biochim. Biophys. Acta 2016, 1863, 382–391. [Google Scholar] [CrossRef]
- Tang, N.; Wang, L.; Esko, J.; Giordano, F.J.; Huang, Y.; Gerber, H.P.; Ferrara, N.; Johnson, R.S. Loss of HIF-1alpha in endothelial cells disrupts a hypoxia-driven VEGF autocrine loop necessary for tumorigenesis. Cancer Cell 2004, 6, 485–495. [Google Scholar] [CrossRef] [Green Version]
- Semenza, G.L. HIF-1 mediates metabolic responses to intratumoral hypoxia and oncogenic mutations. J. Clin. Investig. 2013, 123, 3664–3671. [Google Scholar] [CrossRef] [Green Version]
- Jamali, S.; Klier, M.; Ames, S.; Barros, L.F.; McKenna, R.; Deitmer, J.W.; Becker, H.M. Hypoxia-induced carbonic anhydrase IX facilitates lactate flux in human breast cancer cells by non-catalytic function. Sci. Rep. 2015, 5, 13605. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altemus, M.A.; Goo, L.E.; Little, A.C.; Yates, J.A.; Cheriyan, H.G.; Wu, Z.F.; Merajver, S.D. Breast cancers utilize hypoxic glycogen stores via PYGB, the brain isoform of glycogen phosphorylase, to promote metastatic phenotypes. PLoS ONE 2019, 14, e0220973. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boedtkjer, E. Na+,HCO3- cotransporter NBCn1 accelerates breast carcinogenesis. Cancer Metastasis Rev. 2019, 38, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Wykoff, C.C.; Beasley, N.J.; Watson, P.H.; Turner, K.J.; Pastorek, J.; Sibtain, A.; Wilson, G.D.; Turley, H.; Talks, K.L.; Maxwell, P.H.; et al. Hypoxia-inducible expression of tumor-associated carbonic anhydrases. Cancer Res. 2000, 60, 7075–7083. [Google Scholar] [PubMed]
- Ames, S.; Andring, J.T.; McKenna, R.; Becker, H.M. CAIX forms a transport metabolon with monocarboxylate transporters in human breast cancer cells. Oncogene 2020, 39, 1710–1723. [Google Scholar] [CrossRef] [PubMed]
- de Heer, E.C.; Jalving, M.; Harris, A.L. HIFs, angiogenesis, and metabolism: Elusive enemies in breast cancer. J. Clin. Investig. 2020, 130, 5074–5087. [Google Scholar] [CrossRef]
- Altman, B.J.; Stine, Z.E.; Dang, C.V. From Krebs to clinic: Glutamine metabolism to cancer therapy. Nat. Rev. Cancer 2016, 16, 619–634. [Google Scholar] [CrossRef] [Green Version]
- Butler, L.M.; Perone, Y.; Dehairs, J.; Lupien, L.E.; de Laat, V.; Talebi, A.; Loda, M.; Kinlaw, W.B.; Swinnen, J.V. Lipids and cancer: Emerging roles in pathogenesis, diagnosis and therapeutic intervention. Adv. Drug Deliv. Rev. 2020, 159, 245–293. [Google Scholar] [CrossRef]
- Rohrig, F.; Schulze, A. The multifaceted roles of fatty acid synthesis in cancer. Nat. Rev. Cancer 2016, 16, 732–749. [Google Scholar] [CrossRef]
- Choudhry, H.; Harris, A.L. Advances in Hypoxia-Inducible Factor Biology. Cell Metab. 2018, 27, 281–298. [Google Scholar] [CrossRef]
- Choudhry, H.; Schodel, J.; Oikonomopoulos, S.; Camps, C.; Grampp, S.; Harris, A.L.; Ratcliffe, P.J.; Ragoussis, J.; Mole, D.R. Extensive regulation of the non-coding transcriptome by hypoxia: Role of HIF in releasing paused RNApol2. EMBO Rep. 2014, 15, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Platt, J.L.; Salama, R.; Smythies, J.; Choudhry, H.; Davies, J.O.; Hughes, J.R.; Ratcliffe, P.J.; Mole, D.R. Capture-C reveals preformed chromatin interactions between HIF-binding sites and distant promoters. EMBO Rep. 2016, 17, 1410–1421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thienpont, B.; Steinbacher, J.; Zhao, H.; D’Anna, F.; Kuchnio, A.; Ploumakis, A.; Ghesquiere, B.; Van Dyck, L.; Boeckx, B.; Schoonjans, L.; et al. Tumour hypoxia causes DNA hypermethylation by reducing TET activity. Nature 2016, 537, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.L.; Monje, M. Understanding the Deadly Silence of Posterior Fossa A Ependymoma. Mol. Cell 2020, 78, 999–1001. [Google Scholar] [CrossRef] [PubMed]
- Michealraj, K.A.; Kumar, S.A.; Kim, L.J.Y.; Cavalli, F.M.G.; Przelicki, D.; Wojcik, J.B.; Delaidelli, A.; Bajic, A.; Saulnier, O.; MacLeod, G.; et al. Metabolic Regulation of the Epigenome Drives Lethal Infantile Ependymoma. Cell 2020, 181, 1329–1345.e24. [Google Scholar] [CrossRef]
- Pennisi, E. Genomics. ENCODE project writes eulogy for junk DNA. Science 2012, 337, 1159–1161. [Google Scholar] [CrossRef]
- Ge, S.X. Exploratory bioinformatics investigation reveals importance of “junk” DNA in early embryo development. BMC Genom. 2017, 18, 200. [Google Scholar] [CrossRef] [Green Version]
- Bernardi, G. The “Genomic Code”: DNA Pervasively Moulds Chromatin Structures Leaving no Room for “Junk”. Life 2021, 11, 342. [Google Scholar] [CrossRef]
- Ling, H.; Vincent, K.; Pichler, M.; Fodde, R.; Berindan-Neagoe, I.; Slack, F.J.; Calin, G.A. Junk DNA and the long non-coding RNA twist in cancer genetics. Oncogene 2015, 34, 5003–5011. [Google Scholar] [CrossRef] [Green Version]
- Melendez-Zajgla, J.; Maldonado, V. The Role of lncRNAs in the Stem Phenotype of Pancreatic Ductal Adenocarcinoma. Int. J. Mol. Sci. 2021, 22, 6374. [Google Scholar] [CrossRef]
- Jiang, M.C.; Ni, J.J.; Cui, W.Y.; Wang, B.Y.; Zhuo, W. Emerging roles of lncRNA in cancer and therapeutic opportunities. Am. J. Cancer Res. 2019, 9, 1354–1366. [Google Scholar] [PubMed]
- Qian, Y.; Shi, L.; Luo, Z. Long Non-coding RNAs in Cancer: Implications for Diagnosis, Prognosis, and Therapy. Front. Med. 2020, 7, 612393. [Google Scholar] [CrossRef] [PubMed]
- Hon, C.C.; Ramilowski, J.A.; Harshbarger, J.; Bertin, N.; Rackham, O.J.; Gough, J.; Denisenko, E.; Schmeier, S.; Poulsen, T.M.; Severin, J.; et al. An atlas of human long non-coding RNAs with accurate 5’ ends. Nature 2017, 543, 199–204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiping, Z.; Bo, C.; Shifeng, Y.; Feijiang, Y.; Hongjian, Y.; Qihui, C.; Binbin, T. Roles of MALAT1 in development and migration of triple negative and Her-2 positive breast cancer. Oncotarget 2018, 9, 2255–2267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandez-Rojas, M.A.; Melendez-Zajgla, J.; Lagunas, V.M. lincRNA-RP11400K9.4 Regulates Cell Survival and Migration of Breast Cancer Cells. Cancer Genom. Proteom. 2020, 17, 769–779. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, X.; Hu, H.; Yao, M.; Song, Y.; Yang, A.; Xu, X.; Zhang, N.; Gao, J.; Liu, B. PCAT-1 facilitates breast cancer progression via binding to RACK1 and enhancing oxygen-independent stability of HIF-1α. Mol. Ther. Nucleic Acids 2021, 24, 310–324. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.; Cheng, Y.; Zhang, Y.; Guo, R.; Li, S.; Hong, X. Long non-coding RNA LINC00511/miR-150/MMP13 axis promotes breast cancer proliferation, migration and invasion. Biochim. Biophys. Acta Mol. Basis Dis. 2021, 1867, 165957. [Google Scholar] [CrossRef]
- Wang, X.; Chen, T.; Zhang, Y.; Zhang, N.; Li, C.; Li, Y.; Liu, Y.; Zhang, H.; Zhao, W.; Chen, B.; et al. Long noncoding RNA Linc00339 promotes triple-negative breast cancer progression through miR-377-3p/HOXC6 signaling pathway. J. Cell. Physiol. 2019, 234, 13303–13317. [Google Scholar] [CrossRef]
- Wang, Y.; Dong, T.; Wang, P.; Li, S.; Wu, G.; Zhou, J.; Wang, Z. LINC00922 regulates epithelial-mesenchymal transition, invasive and migratory capacities in breast cancer through promoting NKD2 methylation. Cell. Signal. 2021, 77, 109808. [Google Scholar] [CrossRef]
- Lu, G.; Li, Y.; Ma, Y.; Lu, J.; Chen, Y.; Jiang, Q.; Qin, Q.; Zhao, L.; Huang, Q.; Luo, Z.; et al. Long noncoding RNA LINC00511 contributes to breast cancer tumourigenesis and stemness by inducing the miR-185-3p/E2F1/Nanog axis. J. Exp. Clin. Cancer Res. 2018, 37, 289. [Google Scholar] [CrossRef]
- Du, T.; Shi, Y.; Xu, S.; Wan, X.; Sun, H.; Liu, B. Long Non-Coding RNAs in Drug Resistance of Breast Cancer. Onco Targets Ther. 2020, 13, 7075–7087. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022, 12, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Gagliano, T.; Brancolini, C. Epigenetic Mechanisms beyond Tumour-Stroma Crosstalk. Cancers 2021, 13, 914. [Google Scholar] [CrossRef]
- Li, J.J.; Tsang, J.Y.; Tse, G.M. Tumor Microenvironment in Breast Cancer-Updates on Therapeutic Implications and Pathologic Assessment. Cancers 2021, 13, 4233. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.C.; Yeh, C.C.; Chao, L.Y.; Tsai, M.H.; Chen, H.H.; Chuang, E.Y.; Lai, L.C. The hypoxia-responsive lncRNA NDRG-OT1 promotes NDRG1 degradation via ubiquitin-mediated proteolysis in breast cancer cells. Oncotarget 2018, 9, 10470–10482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, L.; Bao, L.; Niu, Y.; Wang, J.E.; Kumar, A.; Xing, C.; Wang, Y.; Luo, W. LncIHAT Is Induced by Hypoxia-Inducible Factor 1 and Promotes Breast Cancer Progression. Mol. Cancer Res. 2021, 19, 678–687. [Google Scholar] [CrossRef]
- Gong, P.J.; Shao, Y.C.; Huang, S.R.; Zeng, Y.F.; Yuan, X.N.; Xu, J.J.; Yin, W.N.; Wei, L.; Zhang, J.W. Hypoxia-Associated Prognostic Markers and Competing Endogenous RNA Co-Expression Networks in Breast Cancer. Front. Oncol. 2020, 10, 579868. [Google Scholar] [CrossRef]
- Liu, X.; Qiao, K.; Zhu, K.; Li, X.; Zhao, C.; Li, J.; Feng, D.; Fang, Y.; Wang, P.; Qian, C.; et al. Long Noncoding RNA HCG18 Promotes Malignant Phenotypes of Breast Cancer Cells via the HCG18/miR-103a-3p/UBE2O/mTORC1/HIF-1α-Positive Feedback Loop. Front. Cell Dev. Biol. 2021, 9, 675082. [Google Scholar] [CrossRef]
- Zheng, F.; Chen, J.; Zhang, X.; Wang, Z.; Chen, J.; Lin, X.; Huang, H.; Fu, W.; Liang, J.; Wu, W.; et al. The HIF-1α antisense long non-coding RNA drives a positive feedback loop of HIF-1α mediated transactivation and glycolysis. Nat. Commun. 2021, 12, 1341. [Google Scholar] [CrossRef]
- Choudhry, H.; Albukhari, A.; Morotti, M.; Haider, S.; Moralli, D.; Smythies, J.; Schödel, J.; Green, C.M.; Camps, C.; Buffa, F.; et al. Tumor hypoxia induces nuclear paraspeckle formation through HIF-2α dependent transcriptional activation of NEAT1 leading to cancer cell survival. Oncogene 2015, 34, 4482–4490. [Google Scholar] [CrossRef] [Green Version]
- Gómez-Maldonado, L.; Tiana, M.; Roche, O.; Prado-Cabrero, A.; Jensen, L.; Fernandez-Barral, A.; Guijarro-Muñoz, I.; Favaro, E.; Moreno-Bueno, G.; Sanz, L.; et al. EFNA3 long noncoding RNAs induced by hypoxia promote metastatic dissemination. Oncogene 2015, 34, 2609–2620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, A.; Li, C.; Xing, Z.; Hu, Q.; Liang, K.; Han, L.; Wang, C.; Hawke, D.H.; Wang, S.; Zhang, Y.; et al. The LINK-A lncRNA activates normoxic HIF1alpha signalling in triple-negative breast cancer. Nat. Cell Biol. 2016, 18, 213–224. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.C.; Luo, J.L.; Phan, N.N.; Cheng, Y.C.; Chow, L.P.; Tsai, M.H.; Chuang, E.Y.; Lai, L.C. Different effects of long noncoding RNA NDRG1-OT1 fragments on NDRG1 transcription in breast cancer cells under hypoxia. RNA Biol. 2018, 15, 1487–1498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, F.; Wang, J.H.; Fan, W.J.; Meng, Y.T.; Li, M.M.; Li, T.T.; Cui, B.; Wang, H.F.; Zhao, Y.; An, F.; et al. Glycolysis gatekeeper PDK1 reprograms breast cancer stem cells under hypoxia. Oncogene 2018, 37, 1062–1074. [Google Scholar] [CrossRef] [Green Version]
- Stone, J.K.; Kim, J.H.; Vukadin, L.; Richard, A.; Giannini, H.K.; Lim, S.S.; Tan, M.; Ahn, E.E. Hypoxia induces cancer cell-specific chromatin interactions and increases MALAT1 expression in breast cancer cells. J. Biol. Chem. 2019, 294, 11213–11224. [Google Scholar] [CrossRef]
- Shih, C.H.; Chuang, L.L.; Tsai, M.H.; Chen, L.H.; Chuang, E.Y.; Lu, T.P.; Lai, L.C. Hypoxia-Induced MALAT1 Promotes the Proliferation and Migration of Breast Cancer Cells by Sponging MiR-3064-5p. Front. Oncol. 2021, 11, 658151. [Google Scholar] [CrossRef]
- Chen, F.; Chen, J.; Yang, L.; Liu, J.; Zhang, X.; Zhang, Y.; Tu, Q.; Yin, D.; Lin, D.; Wong, P.P.; et al. Extracellular vesicle-packaged HIF-1alpha-stabilizing lncRNA from tumour-associated macrophages regulates aerobic glycolysis of breast cancer cells. Nat. Cell Biol. 2019, 21, 498–510. [Google Scholar] [CrossRef]
- Cheng, L.; Xing, Z.; Zhang, P.; Xu, W. Long non-coding RNA LINC00662 promotes proliferation and migration of breast cancer cells via regulating the miR-497-5p/EglN2 axis. Acta Biochim. Pol. 2020, 67, 229–237. [Google Scholar] [CrossRef]
- Du, Y.; Wei, N.; Ma, R.; Jiang, S.H.; Song, D. Long Noncoding RNA MIR210HG Promotes the Warburg Effect and Tumor Growth by Enhancing HIF-1α Translation in Triple-Negative Breast Cancer. Front. Oncol. 2020, 10, 580176. [Google Scholar] [CrossRef]
- Garcia-Venzor, A.; Mandujano-Tinoco, E.A.; Ruiz-Silvestre, A.; Sanchez, J.M.; Lizarraga, F.; Zampedri, C.; Melendez-Zajgla, J.; Maldonado, V. lncMat2B regulated by severe hypoxia induces cisplatin resistance by increasing DNA damage repair and tumor-initiating population in breast cancer cells. Carcinogenesis 2020, 41, 1485–1497. [Google Scholar] [CrossRef]
- Liang, Y.; Song, X.; Li, Y.; Chen, B.; Zhao, W.; Wang, L.; Zhang, H.; Liu, Y.; Han, D.; Zhang, N.; et al. LncRNA BCRT1 promotes breast cancer progression by targeting miR-1303/PTBP3 axis. Mol. Cancer 2020, 19, 85. [Google Scholar] [CrossRef] [PubMed]
- Lozano-Romero, A.; Astudillo-de la Vega, H.; Terrones-Gurrola, M.; Marchat, L.A.; Hernández-Sotelo, D.; Salinas-Vera, Y.M.; Ramos-Payan, R.; Silva-Cázares, M.B.; Nuñez-Olvera, S.I.; Hernández-de la Cruz, O.N.; et al. HOX Transcript Antisense RNA HOTAIR Abrogates Vasculogenic Mimicry by Targeting the AngiomiR-204/FAK Axis in Triple Negative Breast Cancer Cells. Noncoding RNA 2020, 6, 19. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Bao, L.; Chen, Y.; Wang, C.; Luo, M.; Zhang, B.; Zhou, M.; Wang, J.E.; Fang, Y.V.; Kumar, A.; et al. HIF2-Induced Long Noncoding RNA RAB11B-AS1 Promotes Hypoxia-Mediated Angiogenesis and Breast Cancer Metastasis. Cancer Res. 2020, 80, 964–975. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.C.; Su, L.Y.; Chen, L.H.; Lu, T.P.; Chuang, E.Y.; Tsai, M.H.; Chuang, L.L.; Lai, L.C. Regulatory Mechanisms and Functional Roles of Hypoxia-Induced Long Non-Coding RNA MTORT1 in Breast Cancer Cells. Front. Oncol. 2021, 11, 663114. [Google Scholar] [CrossRef] [PubMed]
- Choudhry, H. UCA1 Overexpression Promotes Hypoxic Breast Cancer Cell Proliferation and Inhibits Apoptosis via HIF-1α Activation. J. Oncol. 2021, 2021, 5512156. [Google Scholar] [CrossRef] [PubMed]
- Chu, Z.; Huo, N.; Zhu, X.; Liu, H.; Cong, R.; Ma, L.; Kang, X.; Xue, C.; Li, J.; Li, Q.; et al. FOXO3A-induced LINC00926 suppresses breast tumor growth and metastasis through inhibition of PGK1-mediated Warburg effect. Mol. Ther. 2021, 29, 2737–2753. [Google Scholar] [CrossRef]
- Du, P.; Luo, K.; Li, G.; Zhu, J.; Xiao, Q.; Li, Y.; Zhang, X. Long non-coding RNA VCAN-AS1 promotes the malignant behaviors of breast cancer by regulating the miR-106a-5p-mediated STAT3/HIF-1α pathway. Bioengineered 2021, 12, 5028–5044. [Google Scholar] [CrossRef]
- Stone, J.K.; Vukadin, L.; Ahn, E.E. eNEMAL, an enhancer RNA transcribed from a distal MALAT1 enhancer, promotes NEAT1 long isoform expression. PLoS ONE 2021, 16, e0251515. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, S. LncRNA GHET1 Promotes Hypoxia-Induced Glycolysis, Proliferation, and Invasion in Triple-Negative Breast Cancer Through the Hippo/YAP Signaling Pathway. Front. Cell Dev. Biol. 2021, 9, 643515. [Google Scholar] [CrossRef]
- Zhao, L.; Jiang, L.; Zhang, M.; Zhang, Q.; Guan, Q.; Li, Y.; He, M.; Zhang, J.; Wei, M. NF-κB-activated SPRY4-IT1 promotes cancer cell metastasis by downregulating TCEB1 mRNA via Staufen1-mediated mRNA decay. Oncogene 2021, 40, 4919–4929. [Google Scholar] [CrossRef]
- Zhu, P.; He, F.; Hou, Y.; Tu, G.; Li, Q.; Jin, T.; Zeng, H.; Qin, Y.; Wan, X.; Qiao, Y.; et al. A novel hypoxic long noncoding RNA KB-1980E6.3 maintains breast cancer stem cell stemness via interacting with IGF2BP1 to facilitate c-Myc mRNA stability. Oncogene 2021, 40, 1609–1627. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yang, J.; Ni, R.; Chen, J.; Zhou, Y.; Song, H.; Jin, L.; Pan, Y. Hypoxia-induced lncRNA RBM5-AS1 promotes tumorigenesis via activating Wnt/beta-catenin signaling in breast cancer. Cell Death Dis. 2022, 13, 95. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Du, C.; Zhang, L.; Wang, Y.; Zhang, Y.; Li, J. LncRNA LINC00649 promotes the growth and metastasis of triple-negative breast cancer by maintaining the stability of HIF-1alpha through the NF90/NF45 complex. Cell Cycle 2022, 21, 1034–1047. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhou, L.; Li, H.; Sun, T.; Wen, X.; Li, X.; Meng, Y.; Li, Y.; Liu, M.; Liu, S.; et al. Nuclear-Encoded lncRNA MALAT1 Epigenetically Controls Metabolic Reprogramming in HCC Cells through the Mitophagy Pathway. Mol. Ther. Nucleic Acids 2021, 23, 264–276. [Google Scholar] [CrossRef] [PubMed]
- Onoguchi-Mizutani, R.; Kirikae, Y.; Ogura, Y.; Gutschner, T.; Diederichs, S.; Akimitsu, N. Identification of a heat-inducible novel nuclear body containing the long noncoding RNA MALAT1. J. Cell Sci. 2021, 134, jcs253559. [Google Scholar] [CrossRef] [PubMed]
- Mondal, P.; Meeran, S.M. Long non-coding RNAs in breast cancer metastasis. Noncoding RNA Res. 2020, 5, 208–218. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, Y.; Lu, J. The roles of long noncoding RNAs in breast cancer metastasis. Cell Death Dis. 2020, 11, 749. [Google Scholar] [CrossRef]
- Zhang, T.; Hu, H.; Yan, G.; Wu, T.; Liu, S.; Chen, W.; Ning, Y.; Lu, Z. Long Non-Coding RNA and Breast Cancer. Technol. Cancer Res. Treat. 2019, 18, 1533033819843889. [Google Scholar] [CrossRef] [Green Version]
- Parsons, M.J.; Tammela, T.; Dow, L.E. WNT as a Driver and Dependency in Cancer. Cancer Discov. 2021, 11, 2413–2429. [Google Scholar] [CrossRef]
- Kandouz, M. The Eph/Ephrin family in cancer metastasis: Communication at the service of invasion. Cancer Metastasis Rev. 2012, 31, 353–373. [Google Scholar] [CrossRef]
- Xin, X.; Li, Q.; Fang, J.; Zhao, T. LncRNA HOTAIR: A Potential Prognostic Factor and Therapeutic Target in Human Cancers. Front. Oncol. 2021, 11, 679244. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhou, H.; Wang, K.; Wang, X.; Wang, M.; Zhao, W.; Xi, X.; Li, Y.; Cai, M.; Zhao, W.; et al. Role, molecular mechanism and the potential target of breast cancer stem cells in breast cancer development. Biomed. Pharmacother. 2022, 147, 112616. [Google Scholar] [CrossRef] [PubMed]
- Saha, T.; Lukong, K.E. Breast Cancer Stem-Like Cells in Drug Resistance: A Review of Mechanisms and Novel Therapeutic Strategies to Overcome Drug Resistance. Front. Oncol. 2022, 12, 856974. [Google Scholar] [CrossRef]
- Al-Hajj, M.; Wicha, M.S.; Benito-Hernandez, A.; Morrison, S.J.; Clarke, M.F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl. Acad. Sci. USA 2003, 100, 3983–3988. [Google Scholar] [CrossRef] [Green Version]
- Flores-Huerta, N.; Silva-Cazares, M.B.; Arriaga-Pizano, L.A.; Prieto-Chavez, J.L.; Lopez-Camarillo, C. LncRNAs and microRNAs as Essential Regulators of Stemness in Breast Cancer Stem Cells. Biomolecules 2021, 11, 380. [Google Scholar] [CrossRef]
- Xiang, L.; Gilkes, D.M.; Chaturvedi, P.; Luo, W.; Hu, H.; Takano, N.; Liang, H.; Semenza, G.L. Ganetespib blocks HIF-1 activity and inhibits tumor growth, vascularization, stem cell maintenance, invasion, and metastasis in orthotopic mouse models of triple-negative breast cancer. J. Mol. Med. 2014, 92, 151–164. [Google Scholar] [CrossRef] [Green Version]
- Shiraishi, A.; Tachi, K.; Essid, N.; Tsuboi, I.; Nagano, M.; Kato, T.; Yamashita, T.; Bando, H.; Hara, H.; Ohneda, O. Hypoxia promotes the phenotypic change of aldehyde dehydrogenase activity of breast cancer stem cells. Cancer Sci. 2017, 108, 362–372. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.Y.; Lang, Y.D.; Lin, H.N.; Liu, Y.R.; Liao, C.C.; Nana, A.W.; Yen, Y.; Chen, R.H. miR-103/107 prolong Wnt/beta-catenin signaling and colorectal cancer stemness by targeting Axin2. Sci. Rep. 2019, 9, 9687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, M.; Xing, L.Q.; Liu, Y.J. A three-long noncoding RNA signature as a diagnostic biomarker for differentiating between triple-negative and non-triple-negative breast cancers. Medicine 2017, 96, e6222. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, L.; Zhao, J.; Du, X.; Yu, Q.; Wu, J.; Wang, B.; Ou, R. Construction and Verification of a Hypoxia-Related 4-lncRNA Model for Prediction of Breast Cancer. Int. J. Gen. Med. 2021, 14, 4605–4617. [Google Scholar] [CrossRef]
- Gu, P.; Zhang, L.; Wang, R.; Ding, W.; Wang, W.; Liu, Y.; Wang, W.; Li, Z.; Yan, B.; Sun, X. Development and Validation of a Novel Hypoxia-Related Long Noncoding RNA Model With Regard to Prognosis and Immune Features in Breast Cancer. Front. Cell Dev. Biol. 2021, 9, 796729. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, G.; Han, J. HIF1A-AS2 predicts poor prognosis and regulates cell migration and invasion in triple-negative breast cancer. J. Cell. Biochem. 2019, 120, 10513–10518. [Google Scholar] [CrossRef] [PubMed]

| Year | lncRNA | Expression in Breast Cancer | Regulated by Hypoxia | Regulates Hypoxia Signaling | Molecular | Phenotype | References |
|---|---|---|---|---|---|---|---|
| 2015 | NEAT1 | Paraspeckle formation | Proliferation, apoptosis inhibition | [60] | |||
| 2015 | EFNA3 | Induce Ephrin-A3 accumulation | Increased metastatic potential | [61] | |||
| 2016 | LINK-A | Increased in TNBC | Allows HIF-1α phosphorylation and stabilization by BRK and LRRK2 | Glycolysis | [62] | ||
| 2018 | NDRG-OT1 | Inhibition of NDRG1 by ubiquitination-mediated proteolysis. Inhibition of NDRG1 promoter activation | [55,63] | ||||
| 2018 | H19 | miRNA let-7 endogenous competitor. HIF-1α activator | PDK-1-mediated Increased glycolysis | [64] | |||
| 2018, 2019, 2021 | MALAT1, TALAM | Increased | XBP1-HIF-1α and HER2 pathway-mediated effects. miRNA-3064-5p endogenous competitor. Chromatin remodeling | Proliferation and invasion | [44,65,66] | ||
| 2019 | HISLA | Increased in breast cancer TAMs | HIF-1α stabilization by blocking PHD2 | Macrophage-mediated enhanced glycolysis | [67] | ||
| 2020 | LINC00662 | Increased | miR-497-5p endogenous competitor mediated EglN2 regulation | Proliferation and migration | [68] | ||
| 2020 | MIR210HG | Increased | HIF-1α translation enhancement | Glycolysis | [69] | ||
| 2020 | lncMat2B | Stemness | [70] | ||||
| 2020 | BRCT1 | Increased | miR-1303 endogenous competitor-mediated stabilization of PTBP3 | Proliferation, migration, increased metastatic potential | [71] | ||
| 2020 | HOTAIR | Increased | miR-204 endogenous competitor-mediated FAK induction | Migration | [72] | ||
| 2020 | RAB11B-AS1 | Increased | Induces the expression of VEGFA and ANGPTL4 by recruitment of RNA polymerase II | Migration, invasion, and angiogenesis | [73] | ||
| 2021 | LncIHAT | Promotes expression of PDK1 and ITGA6 | Tumor growth and metastasis | [56] | |||
| 2021 | MTORT1 | miR-26a-5p endogenous competitor-mediated CREB1 and STK4 regulation | Proliferation and migration | [74] | |||
| 2021 | UCA1 | Increased | Proliferation and apoptosis | [75] | |||
| 2021 | LINC00926 | Regulation of PGK1 via ubiquitination mediated by STUB1 | Glycolysis | [76] | |||
| 2021 | VCAN-AS1 | Increased | miR-106a-5p endogenous competitor-mediated regulation of STAT3 | Proliferation, migration, invasion, and EMT. | [77] | ||
| 2021 | HCG18 | Increased | miR-103a-3p endogenous competitor-mediated UBE2O/AMPKα2/mTORC1 activation | Proliferation, invasion and stemness | [58] | ||
| 2021 | eNEMAL | Alternative splicing of NEAT1 | [78] | ||||
| 2021 | PCAT-1 | Increased | Stabilizes HIF-1α by preventing RACK1 binding | [46] | |||
| 2021 | GHET1 | Increased in TNBC | Decreased phosphorylation of LATS1 allowing YAP nuclear translocation | Proliferation, viability, invasion, glycolysis | [79] | ||
| 2021 | SPRY4-IT1 | STAU1 recruitment to TCEB1, upregulating HIF-1α | Metastasis | [80] | |||
| 2021 | HIFAL | Increased | Recruitment of PHD3 to PKM2 to enhance HIF-1α activity | Tumor growth | [59] | ||
| 2021 | KB-1980E6.3 | Increased | Activation of IGF2BP1/c-Myc signaling axis to stabilize c-Myc | Stemness | [81] | ||
| 2022 | RBM5-AS1 | Increased | Activation of the Wnt pathway | Proliferation, migration, invasion, EMT, and stemness | [82] | ||
| 2022 | LINC00649 | Increased in TNBC | Stabilization of HIF-1α via NF90/NF45 interaction | Proliferation, migration, and invasion | [83] |
| Year | Marker Genes | Signature Derived From | Features | Reference |
|---|---|---|---|---|
| 2017 | 3-gene signature. ANRIL, HIF1A-AS2, UCA1 | Plasma | Differentiate TNBC vs. other | [99] |
| 2020 | 13-gene signature that includes SNHG16, LINC00899, PSMG3-AS1 and PAXIP-AS1 lncRNAs | Tumoral tissue | Prognosis | [57] |
| 2021 | 12-gene signature. LINC01614, LINCO2384, AL109955.1, AC044849.1, LINCO2084, LINC01615, YTHDF3-AS1, AL451085.3, AL512380.1, HSD11B1-AS1, AC110995.1, AC004847.1, TRG-AS1, AC011978.2, CDK6-AS1, TDRKH-AS1, OTUD6B-AS1, MIR4435-2HG, AP003774.2, SLC12A5-AS1 | Tumoral tissue | Prognosis and immune features | [101] |
| 2021 | 4-gene signature. AL031316.1, AC004585.1, LINC01235, and ACTA2-AS1 | Tumoral tissue | Prognosis | [100] |
| 2018 | MALAT1 | Tumoral tissue | Number of metastatic lymph nodes | [44] |
| 2019 | HIF1A-AS2 | Tumoral tissue | Lymph node metastasis, distant metastasis, and unfavorable histological grade. Shorter overall survival | [102] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maldonado, V.; Melendez-Zajgla, J. The Role of Hypoxia-Associated Long Non-Coding RNAs in Breast Cancer. Cells 2022, 11, 1679. https://doi.org/10.3390/cells11101679
Maldonado V, Melendez-Zajgla J. The Role of Hypoxia-Associated Long Non-Coding RNAs in Breast Cancer. Cells. 2022; 11(10):1679. https://doi.org/10.3390/cells11101679
Chicago/Turabian StyleMaldonado, Vilma, and Jorge Melendez-Zajgla. 2022. "The Role of Hypoxia-Associated Long Non-Coding RNAs in Breast Cancer" Cells 11, no. 10: 1679. https://doi.org/10.3390/cells11101679