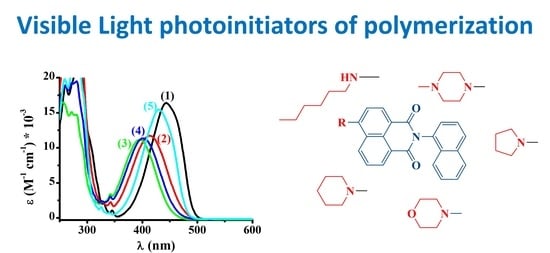
2.1. Synthesis of the Investigated Naphthalimides
All details concerning purchased chemicals are given in [
30]. Mass spectroscopy analyses were performed as detailed in [
31]. Details concerning NMR analyses are given in [
32].
2.1.1. Synthesis of 6-Bromo-2-(naphthalen-1-yl)-1H-benzo[de]isoquinoline-1,3(2H)-dione
A mixture 6-bromo-1H,3H-benzo[de]isochromene-1,3-dione (2.30 g, 8.30 mmol, M = 277.07 g/mol) and 1-naphthylamine (1.19 g, 8.30 mmol, M = 143.19 g/mol) was dissolved in acetic acid (50 mL) and the solution was refluxed overnight. After cooling, the solvent was removed under reduced pressure. The residue was purified by column chromatography (SiO2) using a mixture of eluent (DCM:THF). For higher purity, the product was dissolved in a minimum of ethanol and cooled in the fridge for one night. The yellow solid was filtered off, washed with cold ethanol, and dried under vacuum (2.87 g, 86% yield). 1H NMR (400 MHz, CDCl3) δ: 8.68 (dd, J = 8.5, 1.0 Hz, 1H), 8.63 (dd, J = 7.3, 1.0 Hz, 1H), 8.39 (d, J = 7.9 Hz, 1H), 8.30 (d, J = 7.9 Hz, 1H), 8.08 (dd, J = 11.4, 4.3 Hz, 3H), 7.82 (d, J = 8.4 Hz, 1H), 7.71–7.62 (m, 2H), 7.61–7.55 (m, 1H), 7.46 (ddd, J = 8.2, 6.8, 1.1 Hz, 1H)); 13C NMR (101 MHz, CDCl3) δ: 163.38, 163.31, 133.80, 132.88, 132.58, 131.75, 131.40, 131.12, 130.06, 130.04, 129.38, 129.19, 128.84, 128.20, 127.04, 126.90, 126.25, 125.71, 123.36, 122.58, 122.54; HRMS (ESI MS) m/z: theor: 401.0051 found: 401.0054 (M+ detected).
2.1.2. Synthesis of 2-(Naphthalen-1-yl)-6-(pyrrolidin-1-yl)-1H-benzo[de]isoquinoline1,3(2H)-dione (Napht-1)
A mixture of 6-bromo-2-(naphthalen-1-yl)-1H-benzo[de]isoquinoline-1,3(2H)-dione (1 g, 2.49 mmol, M = 402.25 g/mol) was dissolved in 2-ethoxyethanol (50 mL) and pyrrolidine (5 mL) was added. The solution was refluxed overnight. After cooling, the solvent was removed under reduced pressure. The residue was filtered on a plug of silicagel using DCM as the eluent. Upon evaporation of the solvent, a precipitate formed. It was filtered off, washed several times with pentane, and dried under vacuum (820 mg, 84% yield). 1H NMR (400 MHz, CDCl3) δ: 8.68 (dd, J = 8.6, 1.1 Hz, 1H), 8.63 (dd, J = 7.3, 1.1 Hz, 1H), 8.49 (d, J = 8.6 Hz, 1H), 7.95 (dd, J = 13.6, 8.2 Hz, 2H), 7.67–7.56 (m, 3H), 7.51–7.44 (m, 2H), 7.40 (ddd, J = 8.2, 6.8, 1.3 Hz, 1H), 6.88 (d, J = 8.7 Hz, 1H), 3.84 (t, J = 6.5 Hz, 4H), 2.19–2.06 (m, 4H); 13C NMR (101 MHz, CDCl3) δ: 165.16, 164.27, 153.04, 134.57, 133.95, 133.22, 132.51, 132.01, 131.61, 130.45, 129.04, 128.56, 126.94, 126.78, 126.13, 125.67, 123.12, 122.80, 122.70, 122.28, 110.52, 108.65, 53.27, 26.09; HRMS (ESI MS) m/z: theor: 392.1525 found: 392.1522 (M+ detected).
2.1.3. Synthesis of 2-(Naphthalen-1-yl)-6-(piperidin-1-yl)-1H-benzo[de]isoquinoline-1,3(2H)-dione (Napht-2)
A mixture of 6-bromo-2-(naphthalen-1-yl)-1H-benzo[de]isoquinoline-1,3(2H)-dione (1 g, 2.49 mmol, M = 402.25 g/mol) was dissolved in 2-ethoxyethanol (50 mL) and piperidine (5 mL) was added. The solution was refluxed overnight. After cooling, the solvent was removed under reduced pressure. The residue was filtered on a plug of silicagel using DCM as the eluent. Upon evaporation of the solvent, a precipitate formed. It was filtered off, washed several times with pentane and dried under vacuum (930 mg, 92% yield). 1H NMR (400 MHz, CDCl3) δ: 8.64 (dd, J = 7.3, 1.2 Hz, 1H), 8.57 (d, J = 8.1 Hz, 1H), 8.49 (dd, J = 8.5, 1.2 Hz, 1H), 7.96 (dd, J = 11.8, 7.9 Hz, 2H), 7.74 (dd, J = 8.4, 7.3 Hz, 1H), 7.63 (dd, J = 8.2, 7.3 Hz, 2H), 7.52–7.45 (m, 2H), 7.41 (ddd, J = 8.2, 6.9, 1.4 Hz, 1H), 7.23 (s, 1H), 3.35–3.24 (m, 4H), 1.98–1.87 (m, 4H), 1.81–1.70 (m, 2H); 13C NMR (101 MHz, CDCl3) δ: 164.83, 164.30, 157.75, 134.57, 133.27, 132.77, 131.61, 131.12, 130.69, 130.34, 129.24, 128.62, 127.01, 126.79, 126.55, 126.20, 125.63, 125.49, 123.31, 122.10, 115.94, 114.88, 54.59, 26.27, 24.38; HRMS (ESI MS) m/z: theor: 406.1681 found: 406.1680 (M+ detected).
2.1.4. Synthesis of 6-Morpholino-2-(naphthalen-1-yl)-1H-benzo[de]isoquinoline-1,3(2H)-dione (Napht-3)
A mixture of 6-bromo-2-(naphthalen-1-yl)-1H-benzo[de]isoquinoline-1,3(2H)-dione (1 g, 2.49 mmol, M = 402.25 g/mol) was dissolved in 2-ethoxyethanol (50 mL) and morpholine (5 mL) was added. The solution was refluxed overnight. After cooling, the solvent was removed under reduced pressure. The residue was filtered on a plug of silicagel using DCM as the eluent. Upon evaporation of the solvent, a precipitate formed. It was filtered off, washed several times with pentane and dried under vacuum (860 mg, 85% yield). 1H NMR (400 MHz, CDCl3) δ: 8.66 (dd, J = 7.3, 1.2 Hz, 1H), 8.61 (d, J = 8.1 Hz, 1H), 8.53 (dd, J = 8.5, 1.2 Hz, 1H), 7.97 (dd, J = 12.2, 8.2 Hz, 2H), 7.78 (dd, J = 8.5, 7.3 Hz, 1H), 7.67–7.59 (m, 2H), 7.53–7.46 (m, 2H), 7.42 (ddd, J = 8.2, 6.9, 1.3 Hz, 1H), 7.30 (d, J = 8.1 Hz, 1H), 4.10–4.01 (m, 4H), 3.38–3.27 (m, 4H); 13C NMR (101 MHz, CDCl3) δ: 164.63, 164.14, 156.09, 134.57, 133.13, 132.60, 131.76, 130.60, 130.58, 130.27, 129.34, 128.67, 127.06, 126.79, 126.42, 126.25, 125.99, 125.64, 123.50, 122.01, 117.18, 115.13, 66.99, 53.48; HRMS (ESI MS) m/z: theor: 408.1474 found: 408.1479 (M+ detected).
2.1.5. Synthesis of 6-(4-Methylpiperazin-1-yl)-2-(naphthalen-1-yl)-1H-benzo[de]isoquinoline-1,3(2H)-dione (Napht-4)
A mixture of 6-bromo-2-(naphthalen-1-yl)-1H-benzo[de]isoquinoline-1,3(2H)-dione (1 g, 2.49 mmol, M = 402.25 g/mol) was dissolved in 2-ethoxyethanol (50 mL) and N-methylpiperazine (5 mL) was added. The solution was refluxed overnight. After cooling, the solvent was removed under reduced pressure. The residue was filtered on a plug of silicagel using DCM as the eluent. Upon evaporation of the solvent, a precipitate formed. It was filtered off, washed several times with pentane and dried under vacuum (944 mg, 90% yield). 1H NMR (400 MHz, CDCl3) δ: 8.67 (dd, J = 7.3, 1.1 Hz, 1H), 8.61 (d, J = 8.1 Hz, 1H), 8.54 (dd, J = 8.5, 1.1 Hz, 1H), 7.99 (dd, J = 15.7, 8.3 Hz, 2H), 7.78 (dd, J = 8.4, 7.3 Hz, 1H), 7.67–7.63 (m, 2H), 7.51 (td, J = 6.9, 1.1 Hz, 2H), 7.44 (ddd, J = 8.2, 6.9, 1.2 Hz, 1H), 7.32 (d, J = 8.1 Hz, 1H), 3.45–3.36 (m, 4H), 2.83 (s, 4H), 2.50 (s, 3H); 13C NMR (101 MHz, CDCl3) δ: 164.73, 164.21, 156.42, 134.55, 133.20, 132.63, 131.70, 130.82, 130.61, 130.27, 129.31, 128.64, 127.05, 126.77, 126.41, 126.24, 125.77, 125.63, 123.39, 122.02, 116.67, 115.14, 55.15, 53.01, 46.14; HRMS (ESI MS) m/z: theor: 421.1790 found: 421.1792 (M+ detected).
2.1.6. Synthesis of 6-(Xexylamino)-2-(naphthalen-1-yl)-1H-benzo[de]isoquinoline-1,3(2H)-dione (Napht-5)
A mixture of 6-bromo-2-(naphthalen-1-yl)-1H-benzo[de]isoquinoline-1,3(2H)-dione (1 g, 2.49 mmol, M = 402.25 g/mol) was dissolved in 2-ethoxyethanol (50 mL) and hexylamine (5 mL) was added. The solution was refluxed overnight. The solution was refluxed overnight. After cooling, the solvent was removed under reduced pressure. The residue was filtered on a plug of silicagel using DCM as the eluent. Upon evaporation of the solvent, a precipitate formed. It was filtered off, washed several times with pentane, and dried under vacuum (852 mg, 81% yield). 1H NMR (400 MHz, CDCl3) δ: 8.65 (dd, J = 7.3, 0.8 Hz, 1H), 8.53 (d, J = 8.4 Hz, 1H), 8.22–8.12 (m, 1H), 7.95 (t, J = 9.0 Hz, 2H), 7.64 (ddd, J = 15.6, 10.8, 7.8 Hz, 3H), 7.52–7.34 (m, 3H), 6.78 (d, J = 8.5 Hz, 1H), 5.36 (s, 1H), 3.43 (dd, J = 11.5, 7.1 Hz, 2H), 1.83 (dt, J = 14.8, 7.2 Hz, 2H), 1.50–1.29 (m, 6H), 0.94 (t, J = 7.0 Hz, 3H); 13C NMR (101 MHz, CDCl3) δ: 165.06, 164.42, 150.10, 135.15, 134.53, 133.07, 131.67, 130.63, 130.40, 129.08, 128.55, 126.95, 126.75, 126.65, 126.15, 125.61, 124.67, 123.17, 122.15, 120.45, 109.80, 104.40, 43.75, 31.55, 28.83, 26.84, 22.61, 14.04; HRMS (ESI MS) m/z: theor: 422.1994 found: 422.1991 (M+ detected).