1. Introduction
Dysmenorrhea is the most common gynecologic disorder in women of reproductive age, with a prevalence rate ranges from 16% to 91% [
1]. Dysmenorrhea has two types: primary and secondary. Primary dysmenorrhea is defined as painful menstruation, without pelvic disease, and significant symptoms such as sweating, vomiting, fatigue, back pain, headaches, and diarrhea; notably, severe pain does not commonly occur. Secondary dysmenorrhea refers to a pathological pelvic condition with painful menstruation [
2]. Meta-regression analysis reported that dysmenorrhea had higher rates of pain than dyspareunia and noncyclical pain in quality studies [
3]. Dysmenorrhea is among the leading causes with negative impact on absenteeism, limitations in daily living and socialization, and healthcare [
4]. Risk factors of dysmenorrhea include weight [
5], duration of menstrual flow [
5,
6,
7], earlier age at menarche [
5,
6,
7], cigarette smoking [
5,
6,
7,
8], and family history of dysmenorrhea [
7,
9]. Interestingly, severity of dysmenorrhea was not related to the length of menstrual cycle, weight, and age. Moreover, some psychological factors are associated with increased risk of dysmenorrhea such as depression, anxiety, stress, and somatic complaints [
9]. Treatment of dysmenorrhea include using non-steroidal anti-inflammatory drugs (NSAIDs) (38.5%) and oral pills (37.0%) to relieve menstrual pain [
4]. Some previous studies have reported that NSAIDs were not clearly proven with superior efficacy, and the side effect of gastrointestinal upset is common [
10,
11]. Lifestyle adjustments, diet, nutrient supplements, herbs, and complementary and alternative medicine are being explored as treatment options for dysmenorrhea.
The adlay (
Coix Lacryma-Jobi, Job’s tears) seed is a traditional Chinese medicine that has been widely used in Asian countries for thousands of years. They have been recognized for their diverse physiological activity such as anti-inflammatory, anti-obesity, anti-hyperlipidemia, anti-tumor, and anti-allergy effects in recent studies [
12]. These health benefits of adlay extracts are in part attributed to their unique phytochemical components including total phenolics and total flavonoids. For example, these phytochemicals contributed to antioxidant activity and proliferation inhibition on human liver cancer cells [
13]. Moreover, polyphenol extracts of adlay exert a cardioprotective effect via decreased serum levels of TC, LDL-C, and increased HDL-C [
14]. Our previous study showed that the ethyl acetate fraction of adlay hull significantly inhibited uterine myometrial hyperplasia in rats [
15]. The current study further investigates the inhibitory effect of ethyl acetate fraction of adlay testa on the uterine smooth muscle contraction both in an animal model and in human tissue.
Flavonoids are natural products found in plants, flowers, fruits, and leaves and belong to polyphenolic compounds [
16]. Flavonoids possess antioxidant, free radical scavenging, anti-proliferative activities [
17], and an anti-apoptotic effect [
18] to prevent or treat a variety of diseases such as renal diseases, cancer, cardiovascular disease, neurodegenerative disorders, ulcers, and gastritis [
19]. Previous studies have demonstrated that flavonoids from onion effectively attenuated uterine contraction induced by Prostaglandin F2α (PGF
2α) in rats [
20]. In addition, it was reported that the relaxant effect of flavonoid on colon smooth muscle was via blocking calcium (Ca
2+) influx [
21]. Our previous study showed that flavonoids quercetin and naringenin of active compounds have shown inhibitory effects on uterine contractions in rats [
22]. Therefore, adlay might show therapeutic potential against dysmenorrhea.
There is little information in the literature regarding the beneficial effect of adlay ethanolic extracts against dysmenorrhea. Most of the previous studies showed that adlay and its fractions focused on its chemopreventive effect [
23]. This study aimed to investigate the muscle-relaxing effect of adlay ethanolic extracts on uterine smooth muscle contraction induced by PGF
2α using ex vivo and in vivo as well as quantitative estimation of flavonoid, phytosterol, and fatty acid of ethyl acetate fraction of adlay hull and testa.
2. Materials and Methods
2.1. Plant Material
The Coix seeds were collected by commissioned farmers (No. 4 Coix, Taichung). The plant material was dehulled and separated into adlay hull, adlay testa, and adlay (red Job’s tears) by the air sieve method. Adlay was also further refined and divided into adlay bran and polished adlay for the experiment.
2.2. Preparation of Extracts
The plant materials (hull, testa, bran, and polished adlay) were soaked in 10 times the volume (w/v) of 95% ethanol at room temperature overnight. The filtrate was concentrated under reduced pressure after filtering the impurities and repeatedly extracted from the remaining residue twice. The resultants were collected from four parts referred to as adlay hull ethanolic extract (AHE), adlay testa ethanolic extract (ATE), adlay bran ethanolic extract (ABE), and polished adlay ethanolic extract (PAE).
2.3. Preparation of Ethanolic Extract and Its Fractions form Adlay Hull and Adlay Testa
The AHE and ATE with different polarities solvents were prepared by adding 5 times the amount (w/v) of distilled water and further fractionated through successive extraction with n-hexane, ethyl acetate, and n-butanol. Each fraction of AHE was concentrated to dryness under reduced pressure, which gives 0.22%, 0.1%, 0.07%, and a 0.12% yield in hexane (AHE-Hex), ethyl acetate (AHE-EA), n-butanol (AHE-Bu), and water (AHE-Wa), respectively. The same process was applied on ATE and obtained 1.57%, 0.29%, 0.2%, and 0.67% yields in hexane (ATE-Hex), ethyl acetate (ATE-EA), n-butanol (ATE-Bu), and water (ATE-Wa), respectively. Each fraction of AHE and ATE was prepared as 200 mg/mL stock in dimethyl sulfoxide (DMSO) and stored at 4 °C.
2.4. Identification and Quantification of Flavonoids in Ethyl Acetate Fraction of Adlay Hull (AHE-EA) and Ethyl Acetate Fraction of Adlay Testa (ATE-EA) by HPLC-MS (High-Performance Liquid Chromatography–Mass Spectrometry) and HPLC
HPLC coupled to MS was used to identify the flavonoid compounds through their retention times (by comparing them to those of reference standards) and the mass of the selected ions. The analytical HPLC system employed consisted of a Finnign MAT (P4000) high-performance liquid chromatography coupled with a UV–VIS detector (UV2000) and autosampler (AS3000). The separation was achieved on a Gemini C18 3 µm 2 × 150 mm column at ambient temperature. The separation of the extract solution was carried out with a flow of 0.3 mL/min. A 20 µL aliquot of extract solution was injected. The mobile phase consisted of water with 0.01% formic acid (solvent A) and methanol with acetonitrile (solvent B, 1:1 v/v). The gradient used for the determination of flavonoid was as follows: 80% A/20% B, 0–5 min; 60% A/40% B, 5–10 min; 40% A/60% B, 10–30 min; 35% A/65% B, 30–40 min; 1% A/99% B, 40–50 min. MS was performed on a Finnigan MAT LCQ (Thermoquest Corp, San Jose, CA, USA) equipped with a pneumatically assisted electrospray interface. The spray needle voltage was 5 kV, and the temperature of the heated inlet capillary 250 °C. Nebulizer pressure was 60 psi, and auxiliary gas was 30 psi.
2.5. Identification and Quantification of Fatty Acid and Phytosterol in Ethyl Acetate Fraction of Adlay Hull (AHE-EA) and Ethyl Acetate Fraction of Adlay Testa (ATE-EA) by GC
Gas chromatography (GC) analysis was conducted for qualitative analysis of phytosterol. GC was equipped with an FID and a CP-5 megabore capillary column (30 m × 0.53 mm 1 μm; J&W Scientific, Folsom, CA, USA). An initial oven temperature of 272 °C was maintained for 8 min. Then, the temperature was increased to 280 °C at a rate of 1.2 °C/min, and temperature was further increased to 300 °C at a rate of 10 °C/min for 5 min. Then, the final temperature was increased at 20 °C/min to reach 315 °C. 1 μL was injected into the instrument using a split ratio of 80:1. The injector port and detector temperatures were set at 330 °C and 340 °C, respectively. The column flow rate was 5.0 mL/min, and nitrogen was used as the carrier gas. Phytosterol and fatty acid compositions were determined on the basis of the relative chromatographic areas to compare calibration curves of authentic standards to calculate. The fatty acid content was detected by promulgation for confirmation of national standards.
2.6. Experimental Animal Studies
All animal studies were conducted according to the protocols approved by the Institutional Animal Care and Use Committee (IACUC) of Taipei Medical University (Permit No. LAC-2016-0167 and LAC-2016-0221). Female Sprague-Dawley (SD) rats were purchased from a commercial supplier (BioLASCO Taiwan Co., Ltd., Taipei, Taiwan). They were kept for 6~8 weeks under constant conditions of temperature (24 ± 2 °C), relative humidity (50~60%), and light and dark cycles of 12 h during the uterine contraction in vitro experiment. Animals were provided with a standard rodent chow diet (Rodent Laboratory Chow #5001, Ralston Purina Co., St. Louis, MO, USA), and the food and water were allowed ad libitum.
For in vivo uterine contractions experiment, female ICR mice (20–25 g) were purchased from a commercial supplier (BioLASCO Taiwan Co., Ltd., Taipei, Taiwan). The mice were confirmed at the estrous stage by microscopic examination of a vaginal smear in the experiment. The animals were housed and acclimatized under a normal 12 h light-dark cycle, temperature (24 ± 2 °C), and relative humidity (50–70%) for 1 week before the experiments. All mice had ad libitum access to food and water. The mice were then randomly divided into five groups (n = 8 per group) as follows: sesame oil (control), 2 mg/kg bw of stilbestrol (model control), 30 mg/day of AHE-EA, 30 mg/day of ATE-EA, and Ibuprofen 120 mg/kg (positive control) following the third day at the end of the experiment by oral gavage. Mice were treated with stilbestrol, a nonsteroidal estrogen medication, on the first six days in the oxytocin-induced mice writhing model. The mice received 20 IU/kg of oxytocin after treatment at 1 h before the writhing test.
Regarding the acetic acid-induced abdominal writhing test, mice were randomly separated into the following groups (8 mice/group): 0.5% CMC (control), AHE-EA 30 mg/day, ATE-EA 30 mg/day, and aspirin 200 mg/kg. All mice received only one time treatment by oral gavage 1 h before being induced by an intraperitoneal (i.p.) injection of acetic acid (0.6%).
2.7. Oxytocin-Induced Writhing Test
The test was performed on the basis of a previously standardized reference [
24]. Female mice were treated with stilbestrol 2 mg/kg by oral gavage for 6 days. The dose of 30 mg/day of AHE-EA and ATE-EA was selected on the basis of an IC
50 experiment of PGF
2α that resulted in a uterine contraction in vitro. AHE-EA, ATE-EA, and ibuprofen 120 mg/kg (positive control) were administered orally, beginning on day 3 and repeated every 24 h until the end of the experiment. Mice were injected intraperitoneally with 20 IU/kg oxytocin solution on day 7. The time that elapsed until the occurrence of the writhing response was recorded as pain latency as well as the number of writhing responses within 30 min after oxytocin injection. After the writhing test, uterine tissue was removed and collected to further analysis.
2.8. Acetic Acid-Induced Writhing Test
The acetic acid-induced writhing test was used to assess abdominal contraction on the basis of previous investigations [
25]. The modified test included a contraction of the abdominal muscle with an elevating and stretching of the hind limbs [
26]. AHE-EA, ATE-EA, and aspirin (200 mg/kg) were administered orally 1 h before providing 10 mL/kg 0.6% acetic acid by intraperitoneal injection. The frequencies and latency of writhing responses were counted and recorded in 30 min.
2.9. Tissue Samples
Matched specimens of uterine leiomyoma and adjacent normal myometrium were obtained from women at the Department of Obstetrics and Gynecology at National Taiwan University Hospital from Dr. Lin-Hung Wei laboratory. Dissected specimens were immediately immersed in liquid saline and stored at 4 °C until we performed smooth muscle test contraction within 2 days. This study was approved by the IRB of National Taiwan University Hospital (IRB number: 201210072RIC).
2.10. Uterine Preparations and Measurement of Uterine Contraction
Rats and mice were sacrificed by carbon dioxide, and the uteri were removed and placed in Krebs’ solution (113 mM NaCl, 4.8 mM KCl, 2.5 mM CaCl2, 18 mM NaHCO3, 1.2 mM KH2PO4, 1.2 mM MgSO4, 5.5 mM glucose, 30 mM mannitol). After removing surrounding fats, we cut the uterine tissues into 0.5~1 cm segments of equal length. To maintain the tissue activity, we placed the segments in isolated organ baths containing Krebs’ solution at 37 °C with a 95% O2 and 5% CO2 supply. Equilibrate with 2 g of weight for at least 30 min for uterine smooth muscle contraction stability, PGF2α, oxytocin, and other drugs of smooth muscle contraction were added to each organ bath to measure their effects on uterine smooth muscle. Each concentration of adlay samples was added separately for 10 min in organ baths in a cumulative manner. Amplitude and frequency of uterine contractions were recorded with force displacement transducers by using the LabScribe software.
2.11. Statistical Analysis
The results of treatment effects were expressed as mean ± standard deviation (SD). Statistical comparisons were performed using Student’s t-test or one-way ANOVA test, followed by Duncan’s multiple range test post hoc analysis using SPSS (IBM Corporation, Armonk, NY, USA), and p-values less than or equal to 0.05 were considered significant.
4. Discussion
Most experimental and clinical research has reported that pathogenesis of primary dysmenorrhea identified uterine prostaglandins as a substantially contributing factor within the last 15 years [
27]. PGF
2α is the principal prostaglandin in the uterus, which functions to increase the contractility of the myometrium. Previous studies have been reported that PGF
2α plays an important role in parturition, including increased contractility, amplitude, duration, and frequency of spontaneous contractility as a myometrial stimulant [
28]. The cause for dysmenorrhea is tissue ischemia resulting from the vessel’s constriction, an increase of intrauterine pressure, and a decrease of uterine blood flow. Thus, prostaglandin theory is a convincing piece of evidence for achieving pain relief of inhibition prostaglandin synthesis and decreased intrauterine pressure [
27]. It has been demonstrated that various mechanisms regulate smooth muscle contractility by changing the intracellular calcium contraction ([Ca
2+]i) [
29,
30]. The extracellular Ca
2+ release and Ca
2+ influx are mediated by phospholipase C activation to IP3 generation, and the receptor-operated Ca
2+ channels (ROC) resulted in ATP-induced [Ca
2+]i increase [
31]. Meanwhile, voltage-operated Ca
2+ channels (VOC) are another factor predominantly responsible for the increase in the [Ca
2+]i, resulting in membrane depolarization to Ca
2+ influx such as high K
+-induced contractions. Oxytocin coupled to the receptor to stimulate inositol trisphosphate (InsP3) production for contraction as well as PGF
2α does not directly release Ca
2+ from store sites [
32]. The experiments were designed to investigate the potential inhibition induced by agonists such as PGF
2α, oxytocin, carbachol, or K
+ solution produced contractions by adlay ethanolic extracts. We then further examined the effect of four parts and their subfractions of ethanolic extracts of adlay in vitro and in vivo. We previously found that methanolic extracts of adlay hull inhibited uterine contractions induced by PGF
2α by blocking external Ca
2+ influx [
22]. Coix seed oil could prevent or reduce the contracture action of striated muscle, as previously described by Liu [
33]. Consistent with previous reports [
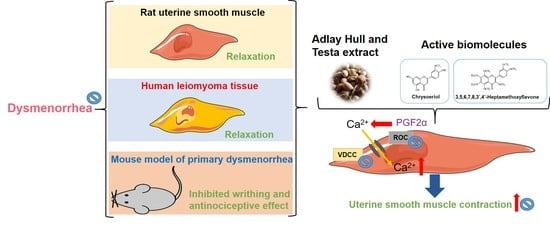
20,
22], our results suggested that the ATE-EA and AHE-EA from ethanolic extracts of adlay strongly suppressed smooth muscle by blocking ROCs or VOCs while inhibiting extracellular calcium ions influx.
When examining the effect of ATE-EA and AHE-EA in modulating smooth muscle contraction for protection, we found the presence of ROC and VOC drugs to increase cumulative concentration. The current findings showed PGF
2α or oxytocin-induced contraction of the AHE-EA was a better strongly protective effect of smooth muscle hypercontraction than ATE-EA. However, ATE-EA and AHE-EA had no protective effect on the carbachol-mediated contraction. A competitive antagonist blocks the chain of reactions produced by an agonist, as the antagonist acts on a different site in the receptor within the effector system in pharmacology [
34]. Thus, the ATE-EA supposed aspect of inhibitory effect might have a less active compound, quercetin, in comparison with AHE-EA in terms of explaining the observed effect. These results are similar to our previous study, which indicated that naringenins are the major pure components of AHM-EA in terms of suppressing contraction [
22]. This is reported in patients with uterine leiomyoma complaints, including menorrhagia, dysmenorrhea, pain, and mild renal impairment [
35]. Moreover, the effect of 375 μg/mL ATE-EA and AHE-EA showed a consistent and highly significant reduced contractility of human leiomyoma and normal myometrial smooth muscle.
Chrysoeriol (luteolin 3′-methyl ether), a bioactive flavonoid, is found in several tropical, medicinal plants known for potent antioxidant [
36], anti-inflammatory, antitumor, antimicrobial, and antiviral activity. In the literature, some reports show selective bronchodilator effect by K
+-induced contractions in the trachea and aorta [
37], as well as smooth muscle-relaxing effects [
38]. No data to date are available regarding the question of whether 3,5,6,7,8,3′,4′-heptamethoxyflavone inhibits contractility on uterine smooth muscle. This study is the first report showing that 3,5,6,7,8,3′,4′-heptamethoxyflavone has an inhibitory effect on uterine contraction.
The oxytocin-induced writhing response preserved for about 7 days was used to evaluate clinical features of primary dysmenorrhea in estrogen-treated mice. When oxytocin was given within 0~30 min, 90% of the twisting body reacted with concentration. This model for dysmenorrhea in mice was simple, reliable, and economical [
24]. In the present study, we demonstrated that ATE-EA and AHE-EA inhibited the contraction of isolated uterus induced by oxytocin. In addition, we demonstrated that ATE-EA and AHE-EA significantly reduced oxytocin-induced writhing response. Our results were similar to those reported previously [
39,
40]. Taken together, these data suggest that ATE-EA and AHE-EA may have potential therapeutic agents for dysmenorrhea.
Administration of acetic acid of writhing resulted in profound pain of endogenous nature for a prolonged period of time because writhing induced the overt response by nociceptors. The writhing characterized the retraction of the abdomen and stretching of hind limbs. The writhing test is the animal model commonly used to evaluate the analgesic effect [
41]. Collier et al. reported that writhing might partly result from stimulation of prostaglandin biosynthesis [
42] as well as the mechanism accounting for the efficacy of non-steroidal anti-inflammatory drugs. Treatment with coix seed oil for cancer patients effectively reduced the efficacy of pain relief in a clinical study [
43]. Adlay also was found to exert many functions, such as anticancer effects, blood sugar reduction, and pain relief, for human health [
12]. In this study, ATE-EA alone exhibited antinociceptive activity in the acetic acid-induced writhing test. In addition, ATE-EA may have more antinociceptive activity than AHE-EA in mice. Further studies are needed to elucidate the mechanism of the antinociceptive action of ATE-EA and AHE-EA.