Introduction: The lung is the essential respiration organ in many air-breathing animals with principal function of gas exchange between atmosphere and bloodstream. However, because of the lung cancer, chronic/acute inflammation, and other disease in lung, the function of lung will get irreversible destruction. Many techniques have been employed to reconstruct lung, such as hydrogel scaffolds[1], decellularized lung[2]-[4], while there is no perfect scaffold yet that can lead to biomimic alveoli. In this study, we generated biomimetic alveolar clusters through using inverse opal 3D scaffolds which have uniform pore structure, better gas permeability and mechanical property. The capacity of blood capillary forming in the cluster was also tested by co-culturing of epithelial cells and endothelial cells.
Materials and Method: Polyurethane (PU) and poly(L-lactic acid) (PLLA) inverse opal 3D scaffolds (IOS) were fabricated to mimic alveolar cluster[5]-[7]. PU and PLLA porous scaffolds by NaCl salt leaching method (SLS) were also generated as the control. Compression test was done to evaluate the mechanical property of IOS and SLS. We designed a new facile gas permeability testing device using a syringe pump and detergent film of which weight and friction can be negligible[8]. The rate of detergent film was recorded to evaluate the gas permeability qualitatively. Epithelial cells (NL20) labelled by PKH26 cell linker and endothelial cells (HUVECs) were seeded into the VEGF grafting scaffolds (IOS and SLS) sequentially. Cell viability was tested by CCK-8 kits and cell location was observed directly by confocal microscope. CD31 staining was used to demonstrate the location of HUVECs.
Results and Discussion: PU-IOS with pore diameter of 185μm, 240μm, 310μm, 432μm and PU-SLS, PLLASLS with pore size of 250-350μm were fabricated. The compression test showed that the Young’s modulus of PU-IOS were around 0.1MPa, which was much softer than PU-SLS but close to the modulus of alveolar. Under the optimized gas perfusion rate, the new gas permeability device showed sensitive measurement of gas permeability for the porous scaffolds. From the gas permeability test it can be seen that the gas permeability of PU-IOS was much better than PU-SLS and PLLA-SLS (Figure 1). The CCK-8 results showed that there were higher cell number in PU-IOS scaffolds than PU-SLS or PLLA-SLS. Confocal images conformed that cells were attached predominantly outside the scaffold than inside as the diameter of the pore of PU-IOS became larger. Within two week culture, NL20 became elongated, communicating with adjacent cells to form a sheet-structure in IOS scaffold. CD31 staining results showed that after 22 day culture, HUVECs became elongated as well in the inverse opal scaffolds, and co-localization with NL20 cells (Figure 2), while on PU-SLS and PLLA-SLS scaffolds, HUVECs and the epithelial cells showed rounded shape.
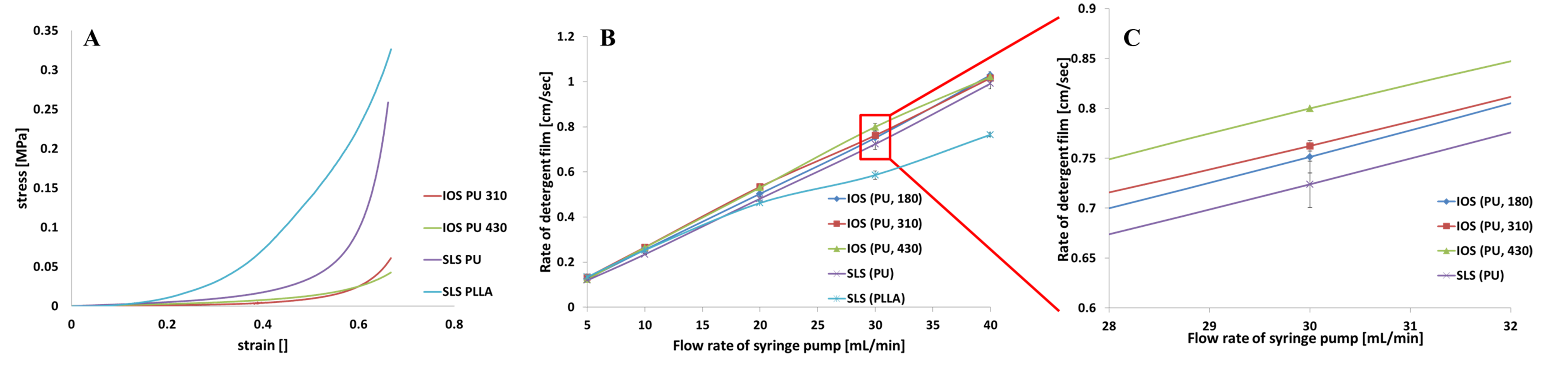
Figure 1 The mechanical test results (A); the gas permeability test results, (B) and (C).
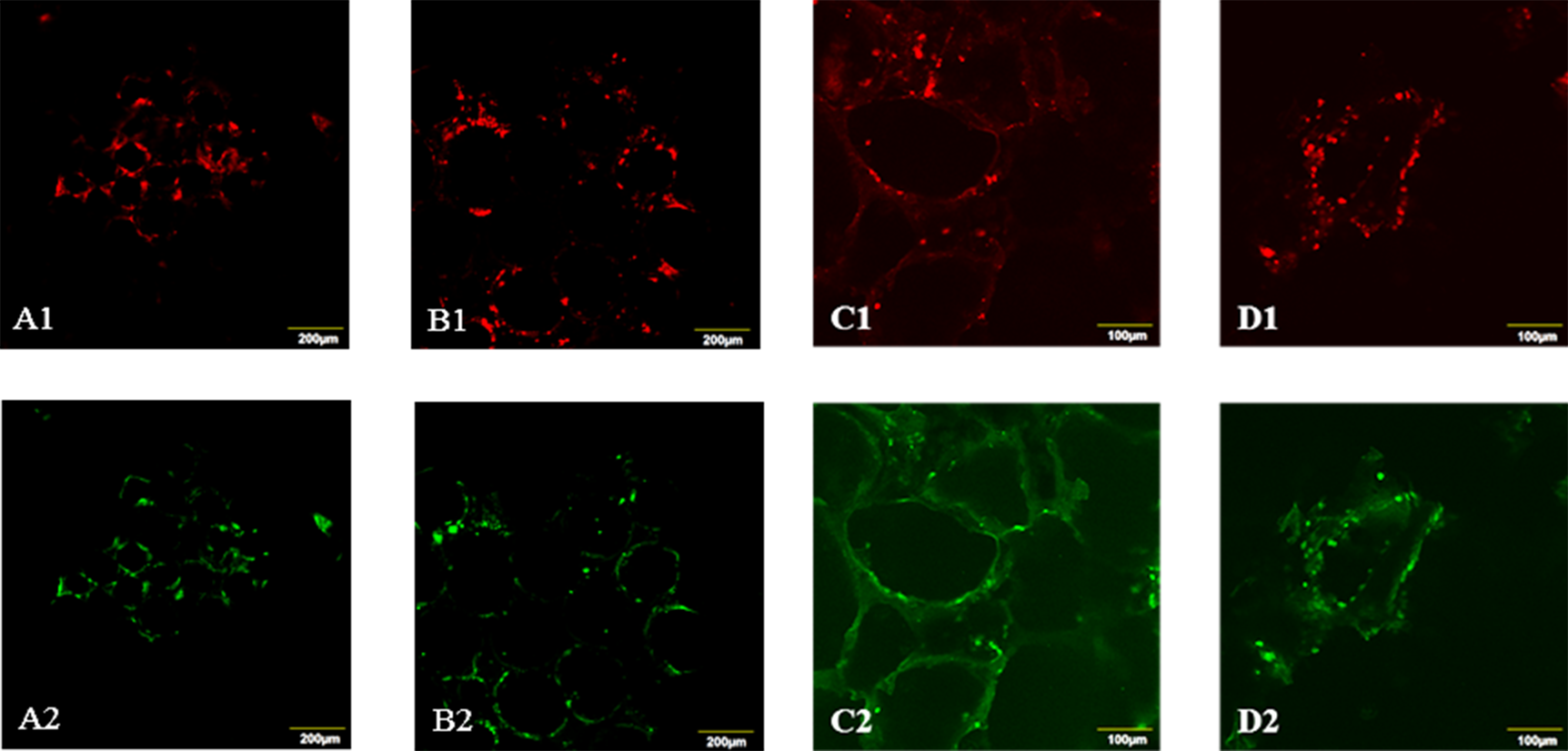
Figure 2 Co-localization of NL20 and HUVECs on different scaffolds. PU-IOS with pore diameter of 185 μm (A1 and A2), 240 μm (B1 and B2): PU-SLS (C1 and C2), and PLLA-SLS (D1 and D2). A1-D1: PKH26 cell linker showing the NL20 location; A2-D2: CD31 staining showing the location of HUVECs.
Conclusion: This study demonstrates that PU-IOS scaffolds can lead to biomimetic alveolar clusters with outstanding mechanical property and gas permeability, also good surface property for epithelial and endothelial cells attachment and proliferation, which could be a good candidate for lung tissue engineering.
References:
[1] Zhang, W. J., Lin, Q. X., Zhang, Y., Liu, C. T., Qiu, L. Y., Wang, H. B., ... & Wang, C. Y. (2011). The reconstruction of lung alveolus‐like structure in collagen‐matrigel/microcapsules scaffolds in vitro. Journal of cellular and molecular medicine, 15(9), 1878-1886.
[2] Girard, E. D., Jensen, T. J., Vadasz, S. D., Blanchette, A. E., Zhang, F., Moncada, C., ... & Finck, C. M. (2013). Automated procedure for biomimetic de-cellularized lung scaffold supporting alveolar epithelial transdifferentiation.Biomaterials, 34(38), 10043-10055.
[3] Petersen, T. H., Calle, E. A., Zhao, L., Lee, E. J., Gui, L., Raredon, M. B., ... & Niklason, L. E. (2010). Tissue-engineered lungs for in vivo implantation.Science, 329(5991), 538-541.
[4] Wagner, D. E., Bonenfant, N. R., Sokocevic, D., DeSarno, M. J., Borg, Z. D., Parsons, C. S., ... & Weiss, D. J. (2014). Three-dimensional scaffolds of acellular human and porcine lungs for high throughput studies of lung disease and regeneration. Biomaterials, 35(9), 2664-2679.
[5] Zhang, Y. S., Choi, S. W., & Xia, Y. (2013). Inverse opal scaffolds for applications in regenerative medicine. Soft Matter, 9(41), 9747-9754.
[6] Choi, S. W., Zhang, Y., MacEwan, M. R., & Xia, Y. (2013). Neovascularization in biodegradable inverse opal scaffolds with uniform and precisely controlled pore sizes. Advanced healthcare materials, 2(1), 145-154.
[7] Choi, S. W., Cheong, I. W., Kim, J. H., & Xia, Y. (2009). Preparation of uniform microspheres using a simple fluidic device and their crystallization into close‐packed lattices. Small, 5(4), 454-459.
[8] Levy, A. (1964). The accuracy of the bubble meter method for gas flow measurements. Journal of Scientific Instruments, 41(7), 449.