Abstract
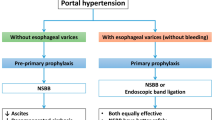
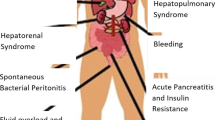
Treatment of the causes of many chronic liver diseases (CLDs) may not be possible. In this case, complications must be anticipated, prevented or at least controlled by the best available therapeutic modalities. There are three main goals for the management of portal hypertension: (i) prevention of the first episode of variceal bleeding largely by non-selective β-adrenoceptor antagonists, which is not generally recommended in children; (ii) control of bleeding by using a stepwise approach from the least to most invasive strategies; (iii) and prevention of re-bleeding using bypass operations, with particular enthusiasm for the use of meso-Rex bypass in the pediatric population. Hepatic encephalopathy management also consists of three main aspects: (i) ruling out other causes of encephalopathy; (ii) identifying and treating precipitating factors; and (iii) starting empiric treatment with drugs such as lactulose, rifaximin, sodium benzoate, and flumazenil. Treatment of mild ascites and peripheral edema should begin with the restriction of sodium and water, followed by careful diuresis, then large-volume paracentesis associated with colloid volume expansion in severe cases. Empiric broad spectrum antimicrobial therapy should be used for the treatment of spontaneous bacterial peritonitis, bacterial and fungal sepsis, and cholangitis, after taking appropriate cultures, with appropriate changes in therapy after sensitivity testing. Empirical therapies continue to be the standard practice for pruritus; these consist of bile acid binding agents, phenobarbital (phenobarbitone), ursodeoxycholic acid, antihistamines, rifampin (rifampicin), and carbamazepine. Partial external biliary diversion can be used in refractory cases. Once hepatorenal syndrome is suspected, treatment should be initiated early in order to prevent the progression of renal failure; approaches consist of general supportive measures, management of concomitant complications, screening for sepsis, treatment with antibiotics, use of vasopressin analogs (terlipressin), and renal replacement therapy if needed. Hepatopulmonary syndrome and portopulmonary hypertension are best managed by liver transplantation. Provision of an adequate caloric supply, nutrition, and vitamin/mineral supplements for the management of growth failure, required vaccinations, and special care for ensuring psychologic well-being should be ensured. Anticoagulation might be attempted in acute portal vein thrombosis.
Some CLDs, such as extrahepatic biliary atresia (EHBA), Crigler-Najjar syndrome, and Indian childhood cirrhosis, require special considerations. For EHBA, Kasai hepatoportoenterostomy is the current standard surgical approach in combination with nutritional therapy and supplemental fat and water soluble vitamins, minerals, and trace elements. In type 1 Crigler-Najjar syndrome, extensive phototherapy is the mainstay of treatment, in association with adjuvant therapy to bind photobilirubin such as calcium phosphate, cholestyramine, or agar, until liver transplantation can be carried out. Treating Indian childhood cirrhosis with penicillamine early in the course of the disease andat doses similar to those used to treat Wilson disease decreases the mortality rate by half.
New hopes for the future include extracorporeal liver support devices (the molecular adsorbent recirculating system [MARS®] and Prometheus®), hepatocyte transplantation, liver-directed gene therapy, genetically engineered enzymes, and therapeutic modalities targeting fibrogenesis. Hepapoietin, a naturally occurring cytokine that promotes hepatocyte growth, is under extensive research.




Similar content being viewed by others
References
El-Shabrawi MHF, Kamal NA. Medical management of chronic liver diseases in children (part I): focus on curable or potentially curable diseases. Pediatr Drugs 2011; 13(6): 357–70
Shneider B, Emre S, Groszmann R, et al. Portal hypertension in children: expert pediatric opinion on the report of the Baveno IV consensus workshop on the methodology of diagnosis and therapy in portal hypertension. Pediatr Transplant 2006; 10: 893–907
Shneider BL. Portal hypertension. In: Suchy FJ, Sokol RJ, Balistreri WF, editors. Liver disease in children. 3rd ed. New York: Cambridge University Press, 2007: 138–62
de Kolster CC, Rapa de Higuera M, Carvajal A, et al. Propranolol in children and adolescents with portal hypertension: its dosage and the clinical, cardiovascular and biochemical effects [in Spanish]. G E N 1992; 46: 199–207
Shashidhar H, Langhans N, Grand RJ. Propranolol in prevention of portal hypertensive hemorrhage in children: a pilot study. J Pediatr Gastroenterol Nutr 1999; 29: 12–7
Ozsoylu S, Kocak N, Demir H, et al. Propranolol for primary and secondary prophylaxis of variceal bleeding in children with cirrhosis. Turk J Pediatr 2000; 42: 31–3
Ozsoylu S, Kocak N, Yuce A. Propranolol therapy for portal hypertension in children. J Pediatr 1985; 106: 317–21
Hardy S, Kleinman RE. Cirrhosis and chronic liver failure. In: Suchy FJ, Sokol RJ, Balistreri WF, editors. Liver disease in children. 3rd ed. New York: Cambridge University Press, 2007: 97–137
Romero-Castro R, Jimenez-Saenz M, Pellicer-Bautista F, et al. Recombinant-activated factor VII as hemostatic therapy in eight cases of severe hemorrhage from esophageal varices. Clin Gastroenterol Hepatol 2004; 2: 78–84
Garcia-Tsao G, Lim J, Members of the Veterans Affairs Hepatitis C Resource Center Program. Management and treatment of patients with cirrhosis and portal hypertension: recommendations from the Department of Veterans Affairs hepatitis C resource center program and the national hepatitis C program. Am J Gastroenterol 2009; 104: 1802–29
D’Amico G, de Franchis R. Upper digestive bleeding in cirrhosis: post-therapeutic outcome and prognostic indicators. Hepatology 2003; 38: 599–612
Lo R, Austin A, Freeman J. Vasopressin in liver disease: should we turn on or off? Curr Clin Pharmacol 2008 Sep; 3(3): 156–65
Hill ID, Bowie MD. Endoscopic sclerotherapy for control of bleeding varices in children. Am J Gastroenterol 1991; 86: 472–6
Mowat AP. Liver disorders in childhood. 2nd ed. London: Buttersworth, 1987
Iwatsuki S, Starzl TE, Todo S, et al. Liver transplantation in the treatment of bleeding esophageal varices. Surgery 1988; 104: 697–705
Rasinska G, Wermenski K, Rajszys P. Percutaneous transsplenic embolization of esophageal varices in a 5-year-old child. Acta Radiol 1987; 28: 299–301
L’Hermine C, Chastanet P, Delemazure O, et al. Percutaneous transhepatic embolization of gastroesophageal varices: results in 400 patients. AJR Am J Roentgenol 1989; 152: 755–60
Superina R, Shneider B, Emre S, et al. Surgical guidelines for the management of extra-hepatic portal vein obstruction. Pediatr Transplant 2006; 10: 908–13
de Ville de Goyet J, Alberti D, Clapuyt P, et al. Direct bypassing of extra-hepatic portal venous obstruction in children: a new technique for combined hepatic portal revascularization and treatment of extrahepatic portal hypertension. J Pediatr Surg 1998; 33: 597–601
Dasgupta R, Roberts E, Superina RA, et al. Effectiveness of Rex shunt in the treatment of portal hypertension. J Pediatr Surg 2006; 41: 108–12
Ferenci P, Lockwood A, Mullen K, et al. Hepatic encephalopathy: definition, nomenclature, diagnosis, and quantification: final report of the working party at the 11th World Congresses of Gastroenterology, Vienna, 1998. Hepatology 2002 Mar; 35(3): 716–21
Prakash R, Mullen KD. Mechanisms, diagnosis and management of hepatic encephalopathy. Nat Rev Gastroenterol Hepatol 2010; 7: 515–25
Shawcross DL, Wright G, Olde Damink SW, et al. Role of ammonia and inflammation in minimal hepatic encephalopathy. Metab Brain Dis 2007; 22: 125–38
Shawcross DL, Davies NA, Williams R, et al. Systemic inflammatory response exacerbates the neuropsychological effects of induced hyper-ammonemia in cirrhosis. J Hepatol 2004; 40: 247–54
Haussinger D, Schliess F. Astrocyte swelling and protein tyrosine nitration in hepatic encephalopathy. Neurochem Int 2005; 47: 64–70
Moldawer LL, Marano MA, Wei H, et al. Cachectin/tumor necrosis factor-alpha alters red blood cell kinetics and induces anemia in vivo. FASEB J 1989 Mar; 3(5): 1637–43
de Vries HE, Blom-Roosemalen MC, van Oosten M, et al. The influence of cytokines on the integrity of the blood-brain barrier in vitro. J Neuro-immunol 1996 Jan; 64(1): 37–43
Didier N, Romero IA, Créminon C, et al. Secretion of interleukin-1beta by astrocytes mediates endothelin-1 and tumour necrosis factor-alpha effects on human brain microvascular endothelial cell permeability. J Neurochem 2003 Jul; 86(1): 246–54
Duchini A, Govindarajan S, Santucci M, et al. Effects of tumor necrosis factor-alpha and interleukin-6 on fluid-phase permeability and ammonia diffusion in CNS-derived endothelial cells. J Investig Med 1996 Oct; 44(8): 474–82
Cagnin A, Taylor-Robinson SD, Forton DM, et al. In vivo imaging of cerebral “peripheral benzodiazepine binding sites” in patients with hepatic encephalopathy. Gut 2006; 55: 547–53
Ahboucha S, Butterworth RF. The neurosteroid system: implication in the pathophysiology of hepatic encephalopathy. Neurochem Int 2008; 52: 575–87
Cordoba J, Minguez B. Hepatic encephalopathy. Semin Liver Dis 2008; 28: 70–80
Muniyappa P, Gulati R, Mohr F, et al. Use and safety of rifaximin in children with inflammatory bowel disease. J Pediatr Gastroenterol Nutr 2009 Oct; 49(4): 400–4
Lee C, Custer JW, Rau RE. Formulary: neomycin. In: Custer JW, Rau RE, editors. The Harriet Lane handbook. 18th ed. Philadelphia (PA): Mosby Elsevier, 2008
Lee C, Custer JW, Rau RE. Formulary: Metronidazole. In: Custer JW, Rau RE, editors. The Harriet Lane handbook. 18th ed. Philadelphia (PA): Mosby Elsevier, 2008: 902–4
Rezvani I. Urea cycle and hyperammonemia (arginine, citrulline, ornithine). In: Kliegman RM, Behrman RE, Jenson HB, et al., editors. Nelson text book of pediatrics. 18th ed. Philadelphia (PA): Saunders Elsevier, 2008: 555–8
Lee C, Custer JW, Rau RE. Formulary: Flumazenil. In: Custer JW, Rau RE, editors. The Harriet Lane Handbook. 18th ed. Philadelphia (PA): Mosby Elsevier, 2008: 835
European Association for the Study of the Liver. EASL clinical practice guidelines on the management of ascites, spontaneous bacterial peritonitis, and hepatorenal syndrome in cirrhosis. J Hepatol 2010 Sep; 53(3): 397–417
Rimola A, Soto R, Bory F, et al. Reticuloendothelial system phagocytic activity in cirrhosis and its relation to bacterial infections and prognosis. Hepatology 1984; 4: 53–8
Rajkovic IA, Williams R. Abnormalities of neutrophil phagocytosis, intra-cellular killing, and metabolic activity in alcoholic cirrhosis and hepatitis. Hepatology 1986; 6: 252–62
Kourilsky O, Leroy C, Peltier AP. Complement and liver cell function in 53 patients with liver disease. Am J Med 1973; 55: 783–90
Fox RA, Dudley FJ, Sherlock S. The serum concentration of the third component of complement beta-1C-beta-1A in liver disease. Gut 1971; 12: 574–8
Larcher VF, Manolaki N, Vegnente A, et al. Spontaneous bacterial peritonitis in children with chronic liver disease: clinical features and etiologic factors. J Pediatr 1985; 106: 907–12
Leggiadro RJ, Lazar LF. Spontaneous bacterial peritonitis due to Neisseria meningitidis serogroup Z in an infant with liver failure. Clin Pediatr (Phila) 1991; 30: 350–2
Clark JH, Fitzgerald JF, Kleiman MB. Spontaneous bacterial peritonitis. J Pediatr 1984; 104: 495–500
Runyon BA. Management of adult patients with ascites caused by cirrhosis. Hepatology 1998; 27: 264–72
Felisart J, Rimola A, Arroyo V, et al. Cefotaxime is more effective than is ampi-cillin-tobramycin in cirrhotics with severe infections. Hepatology 1985; 5: 457–62
Sort P, Navasa M, Arroyo V, et al. Effect of intravenous albumin on renal impairment and mortality in patients with cirrhosis and spontaneous bacterial peritonitis. N Engl J Med 1999; 341: 403–9
Lee C, Custer JW, Rau RE. Formulary: cefotaxime. In: Custer JW, Rau RE, editors. The Harriet Lane handbook, 18th ed. Philadelphia (PA): Mosby Elsevier, 2008: 769–70
Lee C, Custer JW, Rau RE. Formulary: ceftriaxone. In: Custer JW, Rau RE, editors. The Harriet Lane handbook, 18th ed. Philadelphia (PA): Mosby Elsevier, 2008: 773–4
Lee C, Custer JW, Rau RE. Formulary: ampicillin/sulbactam. In: Custer JW, Rau RE, editors. The Harriet Lane handbook, 18th ed. Philadelphia (PA): Mosby Elsevier, 2008: 736
Lee C, Custer JW, Rau RE. Formulary: ciprofloxacin. In: Custer JW, Rau RE, editors. The Harriet Lane handbook, 18th ed. Philadelphia (PA): Mosby Elsevier, 2008: 783–4
Lee C, Custer JW, Rau RE. Formulary: levofloxacin. In: Custer JW, Rau RE, editors. The Harriet Lane handbook, 18th ed. Philadelphia (PA): Mosby Elsevier, 2008: 880
Lee C, Custer JW, Rau RE. Formulary: norfloxacin. In: Custer JW, Rau RE, editors. The Harriet Lane handbook, 18th ed. Philadelphia (PA): Mosby Elsevier, 2008: 921–2
Lee C, Custer JW, Rau RE. Formulary: sulfamethoxazole/trimethoprim. In: Custer JW, Rau RE, editors. The Harriet Lane handbook, 18th ed. Philadelphia (PA): Mosby Elsevier, 2008: 995
El-Shabrawi MH, El-Sisi O, Okasha S, et al. Diagnosis of spontaneous bacterial peritonitis in infants and children with chronic liver disease: a cohort study. Ital J Pediatr 2011; 37: 26
Novak DA, Lauwers GY, Kradin RL. Bacterial, parasitic, and fungal infections of the liver. In: Suchy FJ, Sokol RJ, Balistreri WF, editors. Liver disease in children. 3rd ed. NewYork: Cambridge University Press, 2007: 97–137
Ng VL, Balistreri WF. Treatment options for chronic cholestasis in infancy and childhood. Curr Treat Options Gastroenterol 2005 Oct; 8(5): 419–30
Cies JJ, Giamalis JN. Treatment of cholestatic pruritus in children. Am J Health Syst Pharm. 2007 Jun 1; 64(11): 1157–62
Wolfhagen FH, Sternieri E, Hop WC, et al. Oral naltrexone treatment for cholestatic pruritus: a double-blind, placebo-controlled study. Gastroenterology 1997; 113(4): 1264–9
Zellos A, Roy A, Schwarz KB. Use of oral naltrexone for severe pruritus due to cholestatic liver disease in children. J Pediatr Gastroenterol Nutr 2010 Dec; 51(6): 787–9
Sharma D, Shah UH, Sibal A, et al. Cholecystoappendicostomy for progressive familial intrahepatic cholestasis. Indian Pediatr 2010 Jul 7; 47(7): 626–8
Bustorff-Silva J, Sbraggia Neto L, Olímpio H, et al. Partial internal biliary diversion through a cholecystojejunocolonic anastomosis: a novel surgical approach for patients with progressive familial intrahepatic cholestasis: a preliminary report. J Pediatr Surg 2007 Aug; 42(8): 1337–40
Gün F, Erginel B, Durmaz O, et al. An outstanding non-transplant surgical intervention in progressive familial intrahepatic cholestasis: partial internal biliary diversion. Pediatr Surg Int 2010 Aug; 26(8): 831–4
Tissières P, Sasbón JS, Devictor D. Liver support for fulminant hepatic failure: is it time to use the molecular adsorbents recycling system in children? Pediatr Crit Care Med 2005 Sep; 6(5): 585–91
Mitzner SR, Stange J, Peszynski P, et al. Extracorporeal support of the failing liver. Curr Opin Crit Care. 2002 Apr; 8(2): 171–7
Jost U. Einsatz eines zellfreien Leberunterstützungssystems zur Elimination albumin-gebundener toxischer Substanzen beim akuten Leberversagen [abstract]. Germ Interdisciplin J Intensive Care Med 2000; 37: 435
Klammt S, Stange J, Mitzner SR, et al. Extracorporeal liver support by recirculating albumin dialysis: analysing the effect of the first clinically used generation of the MARS system. Liver 2002; 22Suppl. 2: 30–4
Arroyo V, Ginès P, Gerbes AL, et al. Definition and diagnostic criteria of refractory ascites and hepatorenal syndrome in cirrhosis. International Ascites Club. Hepatology 1996 Jan; 23(1): 164–76
Yousef N, Habes D, Ackermann O, et al. Hepatorenal syndrome: diagnosis and effect of terlipressin therapy in 4 pediatric patients. J Pediatr Gastroenterol Nutr 2010 Jul; 51(1): 100–2
Ginès P, Schrier RW. Renal failure in cirrhosis. N Engl J Med 2009; 361: 1279–90
Moreau R, Lebrec D. The use of vasoconstrictors in patients with cirrhosis: type 1 HRS and beyond. Hepatology 2006; 43: 385–94
Keller F, Heinze H, Jochimson F, et al. Risk factors and outcome of 107 patients with decompensated liver disease and acute renal failure (including 26 patients with hepatorenal syndrome): the role of hemodialysis. Ren Fail 1995; 17: 135–46
Capling RK, Bastani B. Theclinical course of patients with type1 hepatorenal syndrome maintained on hemodialysis. Ren Fail 2004; 26: 563–8
Mitzner SR, Stange J, Klammt S, et al. Improvement of hepatorenal syndrome with extracorporeal albumin dialysis MARS: results of a prospective, randomized, controlled clinical trial. Liver Transpl 2000; 6: 287–9
Restuccia T, Ortega R, Guevara M, et al. Effects of treatment of hepatorenal syndrome before transplantation on posttransplantation outcome: a case-control study. J Hepatol 2004 Jan; 40(1): 140–6
Fallon MB, Arguedas MR. Pulmonary manifestations of liver disease. In: Schiff ER, Sorrell MF, Maddrey WC, editors. Schiffs diseases of the liver. 10th ed. Philadelphia (PA): Lippincott Williams & Wilkins; 2007: 514–27
Abrams G, Fallon M. The hepatopulmonary syndrome. Clin Liver Dis 1997; 1: 185–200
Song JY, Choi JY, Ko JT, et al. Long-term aspirin therapy for hepatopulmonary syndrome. Pediatrics 1996; 97 (6 Pt 1): 917–20
Caldwell SH, Jeffers LJ, Narula OS, et al. Ancient remedies revisited: does Allium sativum (garlic) palliate the hepatopulmonary syndrome? J Clin Gastroenterol 1992; 15: 248–50
Abrams GA, Fallon MB. Treatment of hepatopulmonary syndrome with allium sativum L. (garlic): a pilot trial. J Clin Gastroenterol 1998; 27: 232–5
Rolla G, Bucca C, Brussino L. Methylene blue in the hepatopulmonary syndrome. N Engl J Med 1994; 331: 1098
Schenk P, Madl C, Rezale-Majd S, et al. Methylene blue improves the hepatopulmonary syndrome. Ann Int Med 2000; 133: 701–6
Brussino L, Bucca C, Morello M, et al. Effect on dyspnoea and hypoxaemia of inhaled NG-nitro-L-arginine methyl ester in hepatopulmonary syndrome. Lancet 2003; 362: 43–4
Anel RM, Sheagren JN. Novel presentation and approach to management of hepatopulmonary syndrome with use of antimicrobial agents. Clin Infect Dis 2001; 32: E131–6
Allgaier HP, Haag K, Ochs A, et al. Hepatopulmonary syndrome: successful treatment by transjugular intrahepatic portosystemic stent-shunt (TIPS). J Hepatol 1995; 23: 102–5
Paramesh A, Hussain S, Shneider B, et al. Improvement ofhepatopulmonary syndrome after transjugular intrahepatic portasystemic shunting: case report and review of literature. Pediatr Transpl 2003; 7: 157–62
Kuo P, Johnson L, Plotkin J, et al. Continuous intravenous epoprostenol for the treatment ofportopulmonary hypertension. Transplantation 1997; 63: 604–6
Krowka M, Frantz R, McGoon M, et al. Improvement in pulmonary hemodynamics during intravenous epoprostenol (prostacyclin): a study of 15 patients with moderate to severe portopulmonary hypertension. Hepatology 1999; 30: 641–8
Hoeper MM, Krowka MJ, Strassburg CP. Portopulmonary hypertension and hepatopulmonary syndrome. Lancet 2004; 363: 1461–8
Halank M, Miehlke S, Hoeffken G, et al. Use of oral endothelin-receptor antagonist bosentan in the treatment of portopulmonary hypertension. Transplantation 2004; 77: 1775–6
Hinterhuber L, Graziadei IW, Kähler CM, et al. Endothelin-receptor antagonist treatment of portopulmonary hypertension. Clin Gastroenterol Hepatol 2004; 2: 1039–42
Kuntzen C, Gülberg V, Gerbes AL. Use of a mixed endothelin receptor antagonist in portopulmonary hypertension: a safe and effective therapy? Gastroenterology 2005; 128: 164–8
Barst RJ, Rich S, Widlitz A, et al. Clinical efficacy of sitaxsentan, an endo-thelin-a receptor antagonist, in patients with pulmonary arterial hypertension: open-label pilot study. Chest 2002; 121: 1860–8
Makisalo H, Koivusalo A, Vakkuri A, et al. Sildenafil for portopulmonary hypertension in a patient undergoing liver transplantation. Liver Transpl 2004; 10: 945–50
Chua R, Keogh A, Miyashita M. Novel use of sildenafil in the treatment of portopulmonary hypertension. J Heart Lung Transplant 2005; 24: 498–500
Leonis MA, Balistreri WF. Evaluation and management of end-stage liver disease in children. Gastroenterology 2008 May; 134(6): 1741–51
Bavdekar A, Bhave S, Pandit A. Nutrition management in chronic liver disease. Indian J Pediatr. 2002 May; 69(5): 427–31
Moreno LA, Gottrand F, Hoden S, et al. Improvement of nutritional status in cholestatic children with supplemental nocturnal enteral nutrition. J Pediatr Gastroenterol Nutr 1991; 12: 213–6
Smith J, Horowitz J, Henderson JM, et al. Enteral hyperalimentation in undernourished patients with cirrhosis and ascites. Am J Clin Nutr 1982; 35: 56–72
Fuchs IG. Enteral support of the hospitalized child. In: Suskind R, Lewinter-Suskind L, editors. Textbook of pediatric nutrition. 2nd ed. New York: Raven, 1993: 239–48
Feranchak AP, Sokol RJ. Medical and nutritional management of cholestasis in infants and children. In: Suchy FJ, Sokol RJ, Balistreri WF, editors. Liver disease in children. 3rd ed. New York: Cambridge University Press; 2007: 190–231
A-Kader HH, Balistreri WF. Cholestasis. In: Kliegman RM, Behrman RE, Jenson HB, et al., editors. Nelson text book of pediatrics. 18th ed. Philadelphia (PA): Saunders Elsevier, 2008: 1668–75
Duane WC, Hunninghake DB, Freeman ML, et al. Simvastatin, a competitive inhibitor of HMG-CoA reductase, lowers cholesterol saturation index of gallbladder bile. Hepatology 1988; 8: 1147–50
Logan GM, Duane WC. Lovastatin added to ursodeoxycholic acid further reduces biliary cholesterol saturation. Gastroenterology 1990; 98: 1572–6
Mazzella G, Parini P, Festi D, et al. Effect of simvastatin, ursodeoxycholic acid and simvastatin plus ursodeoxycholic acid on biliary lipid secretion and cholic acid kinetics in nonfamilial hypercholesterolemia. Hepatology 1992; 15: 1072–8
El-Karaksy H, El-Sayed R, El-Raziky M, et al. Cost-effectiveness of prescreening versus empirical vaccination for hepatitis A in Egyptian children with chronic liver disease. East Mediterr Health J 2008 Jul–Aug; 14(4): 804–9
Joshi N, Rao S, Kumar A, et al. Hepatitis A vaccination in chronic liver disease: is it really required in a tropical country like India? Indian J Med Microbiol 2007 Apr; 25(2): 137–9
Song HJ, Kim TH, Song JH, et al. Emerging need for vaccination against hepatitis A virus in patients with chronic liver disease in Korea. J Korean Med Sci 2007 Apr; 22(2): 218–22
Arguedas MR, McGuire BM, Fallon MB. Implementation of vaccination in patients with cirrhosis. Dig Dis Sci 2002 Feb; 47(2): 384–7
Chawla Y, Duseja A, Dhiman RK. Review article: the modern management of portal vein thrombosis. Aliment Pharmacol Ther 2009 Nov 1; 30(9): 881–94
Balistreri WF, Grand R, Suchy FJ, et al. Biliary atresia: current concepts and research directions. Hepatology 1996; 23: 1682–92
Ohi R, Ibrahim M. Biliary atresia. Semin Liver Dis 1992; 1: 115–24
Ryckman FC, Alonso MH, Bucuvalas JC, et al. Biliary atresia: surgical management and treatment options as they relate to outcome. Liver Transplant Surg 1998; 4: S24–33
Khalil BA, Perera MT, Mirza DF. Clinical practice: management of biliary atresia. Eur J Pediatr 2010 Apr; 169(4): 395–402
US Department of Health & Human Services. National Institutes of Health Consensus Development Conference statement: liver transplantation. June 20–23, 1983. Hepatology 1983; 4: 107–10S
Balistreri WF, Bezerra JA. Biliary atresia and other disorders of the extrahepatic bile ducts. In: Suchy FJ, Sokol RJ, Balistreri WF, editors. Liver disease in children. 3rd ed. New York: Cambridge University Press, 2007: 247–69
Kelly DA, Davenport M. Current management of biliary atresia. Arch Dis Child 2007 Dec; 92(12): 1132–5
A-Kader HH, Balistreri WF. Cholestasis. In: Kliegman RM, Behrman RE, Jenson HB, et al., editors. Nelson textbook of pediatrics. 18th ed. Philadelphia (PA): Saunders Elsevier, 2008: 1668–75
Carey RG, Balistreri WF. Metabolic diseases of the liver. In: Kliegman RM, Behrman RE, Jenson HB, et al., editors. Nelson text book of pediatrics. 18th ed. Philadelphia (PA): Saunders Elsevier, 2008: 1675–80
Debray D, Yousef N, Durand P. New management options for end-stage chronic liver disease and acute liver failure: potential for pediatric patients. Paediatr Drugs 2006; 8(1): 1–13
Parola M, Pinzani M. Hepatic wound repair. Fibrogen T Rep 2009 Sep 25; 2(1): 1–6
Friedman SL. Mechanisms of hepatic fibrogenesis. Gastroenterol 2008 May; 134(6): 1655–69
Marino G, Rustgi VK, Salzberg G, et al. Pharmacokinetics and biochemical effects of hepapoietin in patients with chronic liver disease. Aliment Pharmacol Ther 2002 Feb; 16(2): 235–42
Acknowledgements
No sources of funding were used to assist in the preparation of this review. The authors have no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Shabrawi, M.H.F., Kamal, N.M. Medical Management of Chronic Liver Diseases (CLD) in Children (Part II). Pediatr-Drugs 13, 371–383 (2011). https://doi.org/10.2165/11591620-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11591620-000000000-00000