Abstract
Endothelial dysfunction and increased arterial stiffness occur early in the pathogenesis of diabetic vasculopathy. They are both powerful independent predictors of cardiovascular risk. Advances in non-invasive methodologies have led to widespread clinical investigation of these abnormalities in diabetes mellitus, generating a wealth of new knowledge concerning the mechanisms of vascular dysfunction, risk factor associations and potential treatment targets.
Endothelial dysfunction primarily reflects decreased availability of nitric oxide (NO), a critical endothelium-derived vasoactive factor with vasodilatory and anti-atherosclerotic properties. Techniques for assessing endothelial dysfunction include ultrasonographic measurement of flow-mediated vasodilatation of the brachial artery and plethysmography measurement of forearm blood flow responses to vasoactive agents. Arterial stiffness may be assessed using pulse wave analysis to generate measures of pulse wave velocity, arterial compliance and wave reflection.
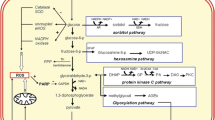
The pathogenesis of endothelial dysfunction in type 2 diabetes is multifactorial, with principal contributors being oxidative stress, dyslipidaemia and hyperglycaemia. Elevated blood glucose levels drive production of reactive oxidant species (ROS) via multiple pathways, resulting in uncoupling of mitochondrial oxidative phosphorylation and endothelial NO synthase (eNOS) activity, reducing NO availability and generating further ROS. Hyperglycaemia also contributes to accelerated arterial stiffening by increasing formation of advanced glycation end-products (AGEs), which alter vessel wall structure and function. Diabetic dyslipidaemia is characterised by accumulation of triglyceride-rich lipoproteins, small dense low-density lipoprotein (LDL) particles, reduced high-density lipoprotein (HDL)-cholesterol and increased postprandial free fatty acid flux. These lipid abnormalities contribute to increasing oxidative stress and may directly inhibit eNOS activity.
Although lipid-regulating agents such as HMG-CoA reductase inhibitors (statins), fibric acid derivatives (fibrates) and fish oils are used to treat diabetic dyslipidaemia, their impact on vascular function is less clear. Studies in type 2 diabetes have yielded inconsistent results, but this may reflect sampling variation and the potential over-riding influence of oxidative stress, dysglycaemia and insulin resistance on endothelial dysfunction. Results of positive intervention trials suggest that improvement in vascular function is mediated by both lipid and non-lipid mechanisms, including anti-inflammatory, anti-oxidative and direct effects on the arterial wall. Other treatments, such as renin-angiotensin-aldosterone system antagonists, insulin sensitisers and lifestyle-based interventions, have shown beneficial effects on vascular function in type 2 diabetes. Novel approaches, targeting eNOS and AGEs, are under development, as are new lipid-regulating therapies that more effectively lower LDL-cholesterol and raise HDL-cholesterol. Combination therapy may potentially increase therapeutic efficacy and permit use of lower doses, thereby reducing the risk of adverse drug effects and interactions. Concomitant treatments that specifically target oxidative stress may also improve endothelial dysfunction in diabetes. Vascular function studies can be used to explore the therapeutic potential and mechanisms of action of new and established interventions, and provide useful surrogate measures for cardiovascular endpoints in clinical trials.













Similar content being viewed by others
Notes
The use of trade names is for product identification purposes only and does not imply endorsement.
References
Haffner SM, Lehto S, Ronnemaa T, et al. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med 1998; 339: 229–34
Lim SC, Caballero AE, Smakowski P, et al. Soluble intercellular adhesion molecule, vascular cell adhesion molecule, and impaired microvascular reactivity are early markers of vasculopathy in type 2 diabetic individuals without microalbuminuria. Diabetes Care 1999; 22: 1865–70
Stehouwer CD, Nauta JJ, Zeldenrust GC, et al. Urinary albumin excretion, cardiovascular disease, and endothelial dysfunction in non-insulin-dependent diabetes mellitus. Lancet 1992; 340: 319–23
Ross R. The pathogenesis of atherosclerosis: a perspective for the 1990s. Nature 1993; 362: 801–9
Schram MT, Kostense PJ, Van Dijk RA, et al. Diabetes, pulse pressure and cardiovascular mortality: the Hoorn Study. J Hypertens 2002; 20: 1743–51
Schachinger V, Britten MB, Zeiher AM. Prognostic impact of coronary vasodilator dysfunction on adverse long-term outcome of coronary heart disease. Circulation 2000; 101: 1899–906
Suwaidi JA, Hamasaki S, Higano ST, et al. Long-term follow-up of patients with mild coronary artery disease and endothelial dysfunction. Circulation 2000; 101: 948–54
Neunteufl T, Heher S, Katzenschlager R, et al. Late prognostic value of flow-mediated dilation in the brachial artery of patients with chest pain. Am J Cardiol 2000; 86: 207–10
Heitzer T, Schlinzig T, Krohn K, et al. Endothelial dysfunction, oxidative stress, and risk of cardiovascular events in patients with coronary artery disease. Circulation 2001; 104: 2673–8
Perticone F, Ceravolo R, Pujia A, et al. Prognostic significance of endothelial dysfunction in hypertensive patients. Circula-tion 2001; 104: 191–6
Franklin SS, Khan SA, Wong ND, et al. Is pulse pressure useful in predicting risk for coronary heart disease? The Framingham Heart Study. Circulation 1999; 100: 354–60
Benetos A, Rudnichi A, Safar M, et al. Pulse pressure and cardiovascular mortality in normotensive and hypertensive subjects. Hypertension 1998; 32: 560–4
Laurent S, Boutouyrie P, Asmar R, et al. Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension 2001; 37: 1236–41
Boutouyrie P, Tropeano AI, Asmar R, et al. Aortic siffness is an independent predictor of primary coronary events in hypertensive patients: a longitudinal study. Hypertension 2002; 39: 10–5
Meaume S, Benetos A, Henry OF, et al. Aortic pulse wave velocity predicts cardiovascular mortality in subjects >70 years of age. Arterioscler Thromb Vasc Biol 2001; 21: 2046–50
London GM, Blacher J, Pannier B, et al. Arterial wave reflections and survival in end-stage renal failure. Hypertension 2001; 38: 434–8
Blacher J, Pannier B, Guerin AP, et al. Carotid arterial stiffness as a predictor of cardiovascular and all-cause mortality in end-stage renal disease. Hypertension 1998; 32: 570–4
Grey E, Bratteli C, Glasser SP, et al. Reduced small artery but not large artery elasticity is an independent risk marker for cardiovascular events. Am J Hypertens 2003; 16: 265–9
Woodman RJ, Watts GF. Measurement and application of arterial stiffness in clinical research: focus on new methodologies and diabetes mellitus. Med Sci Monit 2003; 9: RA81–9
Watts GF, Playford DA. Dyslipoproteinaemia and hyperoxidative stress in the pathogenesis of endothelial dysfunction in non-insulin dependent diabetes mellitus: an hypothesis. Atherosclerosis 1998; 141: 17–30
Laakso M, Lehto S, Penttila I, et al. Lipids and lipoproteins predicting coronary heart disease mortality and morbidity in patients with non-insulin-dependent diabetes. Circulation 1993; 88: 1421–30
American Diabetes Association Task Force for Writing Nutrition Principles and Recommendations for the Management of Diabetes and Related Complications. American Diabetes Association position statement: evidence-based nutrition principles and recommendations for the treatment and prevention of diabetes and related complications. J Am Diet Assoc 2002: 102: 109–18
American Diabetes Association. Diabetes and exercise. Diabetes Care 2001; 24: S51–5
American Diabetes Association. Management of dyslipidaemia in adults with diabetes. Diabetes Care 2003; 26: S83–5
Russo G, Leopold JA, Loscalzo J. Vasoactive substances: nitric oxide and endothelial dysfunction in atherosclerosis. Vascul Pharmacol 2002; 38: 259–69
Moncada S, Higgs A. The L-arginine-nitric oxide pathway. N Engl J Med 1993; 329: 2002–12
Vapaatalo H, Mervaala E. Clinically important factors influencing endothelial function. Med Sci Monit 2001; 7: 1075–85
Creager MA, Cooke JP, Mendelsohn ME, et al. Impaired vasodilation of forearm resistance vessels in hypercholester-olemic humans. J Clin Invest 1990; 86: 228–34
Chowienczyk PJ, Watts GF, Cockcroft JR, et al. Impaired endothelium-dependent vasodilation of forearm resistance vessels in hypercholesterolaemia. Lancet 1992; 340: 1430–2
Celermajer DS, Sorensen KE, Georgakopoulos D, et al. Ciga-rette smoking is associated with dose-related and potentially reversible impairment of endothelium-dependent dilation in healthy young adults. Circulation 1993; 88: 2149–55
Panza JA, Quyyumi AA, Brush JE, et al. Abnormal endothelium-dependent vascular relaxation in patients with essential hypertension. N Engl J Med 1990; 323: 22–7
Johnstone MT, Creager SJ, Scales KM, et al. Impaired endothelium-dependent vasodilation in patients with insulin-dependent diabetes mellitus. Circulation 1993; 88: 2510–6
Williams SB, Cusco JA, Roddy MA, et al. Impaired nitric oxide-mediated vasodilation in patients with non-insulin-dependent diabetes mellitus. J Am Coll Cardiol 1996; 27: 567–74
Celermajer DS, Sorensen KE, Bull C, et al. Endothelium-dependent dilation in the systemic arteries of asymptomatic subjects relates to coronary risk factors and their interaction. J Am Coll Cardiol 1994; 24: 1468–74
Lieberman EH, Gerhard MD, Uehata A, et al. Estrogen improves endothelium-dependent, flow-mediated vasodilation in postmenopausal women. Ann Intern Med 1994; 121: 936–41
Celermajer DS, Sorensen KE, Spiegelhalter DJ, et al. Aging is associated with endothelial dysfunction in healthy men years before the age-related decline in women. J Am Coll Cardiol 1994; 24: 471–6
Clarkson P, Celermajer DS, Powe AJ, et al. Endothelium-dependent dilatation is impaired in young healthy subjects with a family history of premature coronary disease. Circulation 1997; 96: 3378–83
Kari JA, Donald AE, Vallance DT, et al. Physiology and biochemistry of endothelial function in children with chronic renal failure. Kidney Int 1997; 52: 468–72
Woo KS, Chook P, Lolin YI, et al. Hyperhomocyst(e)inemia is a risk factor for arterial endothelial dysfunction in humans. Circulation 1997; 96: 2542–4
Bruel A, Oxlund H. Changes in biomechanical properties, composition of collagen and elastin, and advanced glycation end-products of the rat aorta in relation to age. Atherosclerosis 1996; 127: 155–65
Wilkinson IB, Qasem A, McEniery CM. Nitric oxide regulates local artery distensibility in vivo. Circulation 2002; 105: 213–7
Fitch RM, Vergona R, Sullivan ME, et al. Nitric oxide synthase inhibition increases aortic stiffness measured by pulse wave velocity in rats. Cardiovasc Res 2001; 51: 351–8
McVeigh GE, Allen PB, Morgan DR, et al. Nitric oxide modulation of blood vessel tone identified by arterial waveform analysis. Clin Sci 2001; 100: 387–93
McEniery CM, Qasem A, Schmitt M, et al. Endothelin-1 regulates arterial pulse wave velocity in vivo. J Am Coll Cardiol 2003; 42: 1975–81
Rehman A, Rahman AR, Rasool AH, et al. The effects of angiotensin II on pulse wave velocity in healthy humans. Int J Clin Pharmacol Ther 2001; 39: 423–30
Celermajer DS. Endothelial dysfunction: does it matter? Is it reversible? J Am Coll Cardiol 1997; 30: 325–33
Sorensen KE, Celermajer DS, Spiegelhalter DJ, et al. Non-invasive measurement of human endothelium dependent arterial responses: accuracy and reproducibility. Br Heart J 1995; 74: 247–53
Benjamin N, Calver A, Collier J, et al. Measuring forearm blood flow and interpreting the responses to drugs and mediators. Hypertension 1995; 25: 918–23
Woodman RJ, Playford DA, Watts GF, et al. Improved analysis of brachial artery ultrasound using a novel edge-detection software system. J Appl Physiol 2001; 91: 929–37
Irace C, Ceravolo R, Notarangelo L, et al. Comparison of endothelial function evaluated by strain gauge plethysmography and brachial artery ultrasound. Atherosclerosis 2001; 158: 53–9
Anderson TJ, Uehata A, Gerhard MD, et al. Close relation of endothelial function in the human coronary and peripheral circulations. J Am Coll Cardiol 1995; 26: 1235–41
Sax FL, Cannon RO, Hanson C, et al. Impaired forearm vasodilator reserve in patients with microvascular angina: evidence of a generalized disorder of vascular function? N Engl J Med 1987; 317: 1366–70
Zeiher AM,DrexlerH,SaurbierB,etal. Endothelium-mediated coronary blood flow modulation in humans: effects of age, atherosclerosis, hypercholesterolemia, and hypertension. J Clin Invest 1993; 92: 652–62
Kato M, Shiode N, Yamagata T, et al. Coronary segmental responses to acetylcholine and bradykinin in patients with atherosclerotic risk factors. Am J Cardiol 1997; 80: 751–5
Morris SJ, Shore AC, Tooke JE. Responses of the skin microcirculation to acetylcholine and sodium nitroprusside in patients with NIDDM. Diabetologia 1995; 38: 1337–44
Stehouwer CD, Stroes ES, Hackeng WH, et al. von Willebrand factor and development of diabetic nephropathy in IDDM. Diabetes 1991; 40: 971–6
Jensen T, Bjerre-Knudsen J, Feldt-Rasmussen B, et al. Features of endothelial dysfunction in early diabetic nephropathy. Lancet 1989; I: 461–3
Papaioannou GI, Seip RL, Grey NJ, et al. Brachial artery reactivity in asymptomatic patients with type 2 diabetes mellitus and microalbuminuria (from the Detection of Ischemia in Asymptomatic Diabetics-brachial artery reactivity study). Am J Cardiol 2004; 94: 294–9
Gearing AJ, Newman W. Circulating adhesion molecules in disease. Immunol Today 1993; 14: 506–12
Albertini JP, Valensi P, Lormeau B, et al. Elevated concentrations of soluble E-selectin and vascular cell adhesion mole-cule-1 in NIDDM: effect of intensive insulin treatment. Diabetes Care 1998; 21: 1008–13
Hwang SJ, Ballantyne CM, Sharrett AR, et al. Circulating adhesion molecules VCAM-1, ICAM-1, and E-selectin in carotid atherosclerosis and incident coronary heart disease cases: the Atherosclerosis Risk In Communities (ARIC) Study. Circulation 1997; 96: 4219–25
Hill JM, Zalos G, Halcox JP, et al. Circulating endothelial progenitor cells, vascular function, and cardiovascular risk. N Engl J Med 2003; 348: 593–600
Bramwell JC, Hill AV. Velocity of transmission of the pulse wave. Lancet 1922; I: 891–2
Lehmann ED. Noninvasive measurements of aortic stiffness: methodological considerations. Pathol Biol 1999; 47: 716–30
Kawasaki T, Sasayama S, Yagi S, et al. Non-invasive assessment of the age related changes in stiffness of major branches of the human arteries. Cardiovasc Res 1987; 21: 678–87
Cohn JN, Finkelstein S, McVeigh G, et al. Noninvasive pulse wave analysis for the early detection of vascular disease. Hypertension 1995; 26: 503–8
O’Rourke MF, Pauca A, Jiang XJ. Pulse wave analysis. Br J Clin Pharmacol 2001; 51: 507–22
Dart AM, Kingwell BA. Pulse pressure: a review of mechanisms and clinical relevance. J Am Coll Cardiol 2001; 37: 975–84
Nigam A, Mitchell GF, Lambert J, et al. Relation between conduit vessel stiffness (assessed by tonometry) and endothelial function (assessed by flow-mediated dilatation) in patients with and without coronary heart disease. Am J Cardiol 2003: 92: 395–9
Parvathaneni L, Harp J, Zelinger A, et al. Relation between brachial artery reactivity and noninvasive large and small arterial compliance in healthy volunteers. Am J Cardiol 2002; 89: 894–5
Cruickshank K, Riste L, Anderson SG, et al. Aortic pulse-wave velocity and its relationship to mortality in diabetes and glucose intolerance: an integrated index of vascular function? Circulation 2002; 106: 2085–90
Shoji T, Emoto M, Shinohara K, et al. Diabetes mellitus, aortic stiffness, and cardiovascular mortality in end-stage renal disease. J Am Soc Nephrol 2001; 12: 2117–24
McVeigh GE, Brennan GM, Johnston GD, et al. Impaired endothelium-dependent and independent vasodilation in patients with type 2 (non-insulin-dependent) diabetes mellitus. Diabetologia 1992; 35: 771–6
Watts GF, O’Brien SF, Silvester W, et al. Impaired endothelium-dependent and independent dilatation of forearm resistance arteries in men with diet-treated non-insulin-dependent diabetes: role of dyslipidaemia. Clin Sci 1996; 91: 567–73
Henry RM, Ferreira I, Kostense PJ, et al. Type 2 diabetes is associated with impaired endothelium-dependent, flow-mediated dilation, but impaired glucose metabolism is not: the Hoorn Study. Atherosclerosis 2004;174: 49–56
Nitenberg A, Valensi P, Sachs R, et al. Impairment of coronary vascular reserve and ACh-induced coronary vasodilation in diabetic patients with angiographically normal coronary arter-ies and normal left ventricular systolic function. Diabetes 1993; 42: 1017–25
Caballero AE, Arora S, Saouaf R, et al. Microvascular and macrovascular reactivity is reduced in subjects at risk for type 2 diabetes. Diabetes 1999; 48: 1856–62
Steiner M, Reinhardt KM, Krammer B, et al. Increased levels of soluble adhesion molecules in type 2 (non-insulin dependent) diabetes mellitus are independent of glycaemic control. Thromb Haemost 1994; 72: 979–84
Bannan S, Mansfield MW, Grant PJ. Soluble vascular cell adhesion molecule-1 and E-selectin levels in relation to vascular risk factors and to E-selectin genotype in the first degree relatives of NIDDM patients and in NIDDM patients. Diabetologia 1998; 41: 460–6
Cominacini L, Fratta Pasini A, Garbin U, et al. Elevated levels of soluble E-selectin in patients with IDDM and NIDDM: relation to metabolic control. Diabetologia 1995; 38: 1122–4
Nannipieri M, Rizzo L, Rapuano A, et al. Increased transcapillary escape rate of albumin in microalbuminuric type II diabetic patients. Diabetes Care 1995; 18: 1–9
Cleland SJ, Petrie JR, Small M, et al. Insulin action is associated with endothelial function in hypertension and type 2 diabetes. Hypertension 2000; 35: 507–11
Petrie JR, Ueda S, Webb DJ, et al. Endothelial nitric oxide production and insulin sensitivity: a physiological link with implications for pathogenesis of cardiovascular disease. Circulation 1996; 93: 1331–3
Abe H, Yamada N, Kamata K, et al. Hypertension, hypertriglyceridemia, and impaired endothelium-dependent vascular relaxation in mice lacking insulin receptor substrate-1. J Clin Invest 1998; 101: 1784–8
Lupattelli G, Marchesi S, Roscini AR, et al. Direct association between high-density lipoprotein cholesterol and endothelial function in hyperlipemia. Am J Cardiol 2002; 90: 648–50
Iiyama K, Nagano M, Yo Y, et al. Impaired endothelial function with essential hypertension assessed by ultrasonography. Am Heart J 1996; 132: 779–82
Arcaro G, Zamboni M, Rossi L, et al. Body fat distribution predicts the degree of endothelial dysfunction in uncomplicated obesity. Int J Obes Relat Metab Disord 1999; 23: 936–42
Hogikyan RV, Galecki AT, Pitt B, et al. Specific impairment of endothelium-dependent vasodilation in subjects with type 2 diabetes independent of obesity. J Clin Endocrinol Metab 1998; 83: 1946–52
Ma L, Zhao S, Li J, et al. Interaction of hypertension and diabetes on impairment of endothelial function. Chin Med J 2001; 114: 563–7
Nappo F, Esposito K, Cioffi M, et al. Postprandial endothelial activation in healthy subjects and in type 2 diabetic patients: role of fat and carbohydrate meals. J Am Coll Cardiol 2002; 39: 1145–50
Anderson RA, Evans ML, Ellis GR, et al. The relationships between post-prandial lipaemia, endothelial function and oxidative stress in healthy individuals and patients with type 2 diabetes. Atherosclerosis 2001; 154: 475–83
Anderson RA, Jones CJ, Goodfellow J. Is the fatty meal a trigger for acute coronary syndromes. Atherosclerosis 2001: 159: 9–15
Kawano H, Motoyama T, Hirashima O, et al. Hyperglycemia rapidly suppresses flow-mediated endothelium-dependent vasodilation of brachial artery. J Am Coll Cardiol 1999; 34: 146–54
Shige H, Ishikawa T, Suzukawa M, et al. Endothelium-dependent flow-mediated vasodilation in the postprandial state in type 2 diabetes mellitus. Am J Cardiol 1999; 84: 1272–4
Haller H. Postprandial glucose and vascular disease. Diabet Med 1997; 14 Suppl. 3: S50-6
Nesto R. C-reactive protein, its role in inflammation, type 2 diabetes and cardiovascular disease, and the effects of insulin-sensitizing treatment with thiazolidinediones. Diabet Med 2004; 21: 810–7
Dandona P, Aljada A, Chaudhuri A. Endothelial dysfunction, inflammation and diabetes. Rev Endocr Metab Disord 2004; 5: 189–97
Aoun S, Blacher J, Safar ME, et al. Diabetes mellitus and renal failure: effects on large artery stiffness. J Hum Hypertens 2001; 15: 693–700
Lehmann ED, Gosling RG, Sonksen PH. Arterial wall compliance in diabetes. Diabetic Med 1992; 9: 114–9
Taniwaki H, Kawagishi T, Emoto M, et al. Correlation between the intima-media thickness of the carotid artery and aortic pulse-wave velocity in patients with type 2 diabetes: vessel wall properties in type 2 diabetes. Diabetes Care 1999; 22: 1851–7
Brooks BA, Molyneaux LM, Yue DK. Augmentation of central arterial pressure in type 2 diabetes. Diabet Med 2001; 18: 374–80
Salomaa V, Riley W, Kark JD, et al. Non-insulin-dependent diabetes mellitus and fasting glucose and insulin concentrations are associated with arterial stiffness indexes: the ARIC Study. Atherosclerosis Risk in Communities Study. Circulation 1995; 91: 1432–43
Henry RM, Kostense PJ, Spijkerman AM, et al. Arterial stiffness increases with deteriorating glucose tolerance status: the Hoorn Study. Circulation 2003; 107: 2089–95
McVeigh G, Brennan G, Hayes R, et al. Vascular abnormalities in non-insulin-dependent diabetes mellitus identified by arterial waveform analysis. Am J Med 1993; 95: 424–30
Weinberger MH, Fineberg NS, Fineberg SE. The influence of blood pressure and carbohydrate tolerance on vascular compliance in humans. Am J Hypertens 2002; 15: 678–82
Wilkinson IB, MacCallum H, Cockcroft JR, et al. Inhibition of basal nitric oxide synthesis increases aortic augmentation index and pulse wave velocity in vivo. Br J Clin Pharmacol 2002; 53: 189–92
Cohn JN. Vascular wall function as a risk marker for cardiovascular disease. J Hypertens 1999; 17 Suppl. 5: S41-4
Cameron JD, Bulpitt CJ, Pinto SE, et al. The aging of elastic and muscular arteries: a comparison of diabetic and nondiabetic subjects. Diabetes Care 2003; 26: 2133–88
Aronson D. Cross-linking of glycated collagen in the pathogenesis of arterial and myocardial stiffening of aging and diabetes. J Hypertens. 2003; 21: 3–12
Reiser KM. Nonenzymatic glycation of collagen in aging and diabetes. Proc Soc Exp Biol Med 1991; 196: 17–29
Atkinson J. Arterial calcification: mechanisms, consequences and animal models. Pathol Biol (Paris) 1999; 47: 677–84
Wahlqvist ML, Lo CS, Myers KA, et al. Putative determinants of arterial wall compliance in NIDDM. Diabetes Care 1988; 11: 787–90
Emoto M, Nishizawa Y, Kawagishi T, et al. Stiffness indexes beta of the common carotid and femoral arteries are associated with insulin resistance in NIDDM. Diabetes Care 1998; 21: 1178–82
Tanokuchi S, Okada S, Ota Z. Factors related to aortic pulse-wave velocity in patients with non-insulin-dependent diabetes mellitus. J Int Med Res 1995; 23: 423–30
Tedesco MA, Natale F, Di Salvo G, et al. Effects of coexisting hypertension and type II diabetes mellitus on arterial stiffness. J Hum Hypertens 2004; 18: 469–73
Lim HS, Lip GY. Arterial stiffness in diabetes and hypertension. J Hum Hypertens 2004; 18: 467–8
Van Dijk RA, Bakker SJ, Scheffer PG, et al. Associations of metabolic variables with arterial stiffness in type 2 diabetes mellitus: focus on insulin sensitivity and postprandial triglyceridaemia. Eur J Clin Invest 2003; 33: 307–15
Airaksinen KE, Salmela PI, Linnaluoto MK, et al. Diminished arterial elasticity in diabetes: association with fluorescent advanced glycosylation end products in collagen. Cardiovasc Res 1993; 27: 942–5
Vlassara H, Palace MR. Diabetes and advanced glycation end-products. J Intern Med. 2002; 251: 87–101
Doi T, Vlassara H, Kirstein M, et al. Receptor-specific increase in extracellular matrix production in mouse mesangial cells by advanced glycosylation end products is mediated via platelet-derived growth factor. Proc Natl Acad Sci U S A 1992; 89: 2873–7
Mott JD, Khalifah RG, Nagase H, et al. Nonenzymatic glycation of type IV collagen and matrix metalloproteinase susceptibility. Kidney Int 1997; 52: 1302–12
Andresen JL, Rasmussen LM, Ledet T. Diabetic macroangi-opathy and atherosclerosis. Diabetes 1996; 45 Suppl. 3: S91-4
Lehto S, Ronnemaa T, Haffner SM, et al. Dyslipidemia and hyperglycemia predict coronary heart disease events in middle-aged patients with NIDDM. Diabetes 1997; 46: 1354–9
Howard BV. Insulin resistance and lipid metabolism. Am J Cardiol 1999; 84(1A): 28J–32J
Pfeifer MA, Brunzell JD, Best JD, et al. The response of plasma triglyceride, cholesterol, and lipoprotein lipase to treatment in non-insulin-dependent diabetic subjects without familial hypertriglyceridemia. Diabetes 1983; 32: 525–31
Malmstrom R, Packard CJ, Caslake M, et al. Defective regulation of triglyceride metabolism by insulin in the liver in NIDDM. Diabetologia 1997; 40: 454–62
Lewis GF, Steiner G. Acute effects of insulin in the control of VLDL production in humans: implications for the insulin-resistant state. Diabetes Care 1996; 19: 390–3
Reaven GM, Greenfield MS. Diabetic hypertriglyceridemia: evidence for three clinical syndromes. Diabetes 1981; 30: 66–75
Taskinen MR, Beltz WF, Harper I, et al. Effects of NIDDM on very-low-density lipoprotein triglyceride and apolipoprotein B metabolism: studies before and after sulfonylurea therapy. Diabetes 1986; 35: 1268–77
Lewis GF, Uffelman KD, Szeto LW, et al. Interaction between free fatty acids and insulin in the acute control of very low density lipoprotein production in humans. J Clin Invest 1995; 95: 158–66
Adeli K, Taghibiglou C, Van Iderstine SC, et al. Mechanisms of hepatic very low-density lipoprotein overproduction in insulin resistance. Trends Cardiovasc Med 2001; 11: 170–6
Cassader M, Gambino R, Musso G, et al. Postprandial triglycer-ide-rich lipoprotein metabolism and insulin sensitivity in nonalcoholic steatohepatitis patients. Lipids 2001; 36: 1117–24
Tacikowski T, Dzieniszewski J, Nowicka G, et al. Comparative analysis of lipid profiles assessed by ultracentrifugation in patients with various hyperlipoproteinaemia types in correlation with hepatic steatosis. Med Sci Monit 2002; 8: CR697–701
Lewis GF, Uffelman KD, Szeto LW, et al. Effects of acute hyperinsulinemia on VLDL triglyceride and VLDL apoB production in normal weight and obese individuals. Diabetes 1993; 42: 833–42
Boden G. Effects of free fatty acids (FFA) on glucose metabol-ism: significance for insulin resistance and type 2 diabetes. Exp Clin Endocrinol Diabetes 2003; 111: 121–4
Kraegen EW, Cooney GJ, Ye JM, et al. The role of lipids in the pathogenesis of muscle insulin resistance and beta cell failure in type II diabetes and obesity. Exp Clin Endocrinol Diabetes 2001; 109 Suppl. 2: S189-201
Taskinen MR. Lipoprotein lipase in diabetes. Diabetes Met Rev 1987; 3: 551–70
Abbate SL, Brunzell JD. Pathophysiology of hyperlipidemia in diabetes mellitus. J Cardiovasc Pharmacol 1990; 16 Suppl. 9: S1-7
De Man FH, Cabezas MC, Van Barlingen HH, et al. Triglyceride-rich lipoproteins in non-insulin-dependent diabetes mellitus: post-prandial metabolism and relation to premature atherosclerosis. Eur J Clin Invest 1996; 26: 89–108
Chen YD, Swami S, Skowronski R, et al. Differences in postprandial lipemia between patients with normal glucose tolerance and noninsulin-dependent diabetes mellitus. J Clin Endocrinol Metab 1993; 76: 172–7
Erkelens DW. Diabetic dyslipidaemia. Eur Heart J 1998; 19 Suppl. H: H27-40
Elchebly M, Porokhov B, Pulcini T, et al. Alterations in composition and concentration of lipoproteins and elevated cholesteryl ester transfer in non-insulin-dependent diabetes mellitus (NIDDM). Atherosclerosis 1996; 123: 93–101
Kahri J, Syvanne M, Taskinen MR. Plasma cholesteryl ester transfer protein activity in non-insulin-dependent diabetic patients with and without coronary artery disease. Metabolism 1994; 43: 1498–502
Dullaart RP, De Vries R, Scheek L, et al. Type 2 diabetes mellitus is associated with differential effects on plasma cholesteryl ester transfer protein and phospholipid transfer protein activities and concentrations. Scand J Clin Lab Invest 2004; 64: 205–15
Borggreve SE, De Vries R, Dullaart RP. Alterations in high-density lipoprotein metabolism and reverse cholesterol transport in insulin resistance and type 2 diabetes mellitus: role of lipolytic enzymes, lecithinxholesterol acyltransferase and lipid transfer proteins. Eur J Clin Invest 2003; 33: 1051–69
Riemens SC, van Tol A, Sluiter WJ, et al. Plasma phospholipid transfer protein activity is related to insulin resistance: impaired acute lowering by insulin in obese type II diabetic patients. Diabetologia 1998; 41: 929–34
Baynes C, Henderson AD, Anyaoku V, et al. The role of insulin insensitivity and hepatic lipase in the dyslipidaemia of type 2 diabetes. Diabet Med 1991; 8: 560–6
De Vries R, Borggreve SE, Dullaart RP. Role of lipases, lecithinxholesterol acyltransferase and cholesteryl ester transfer protein in abnormal high density lipoprotein metabolism in insulin resistance and type 2 diabetes mellitus. Clin Lab 2003; 49: 601–13
Syvanne M, Ahola M, Lahdenpera S, et al. High density lipoprotein subfractions in non-insulin-dependent diabetes mellitus and coronary artery disease. J Lipid Res 1995; 36: 573–82
Feingold KR, Grunfeld C, Pang M, et al. LDL subclass pheno-types and triglyceride metabolism in non-insulin-dependent diabetes. Arterioscler Thromb 1992; 12: 1496–502
Singh AT, Rainwater DL, Haffner SM, et al. Effect of diabetes on lipoprotein size. Arterioscler Thromb Vasc Biol 1995; 15: 1805–11
Tan KC, Cooper MB, Ling KL, et al. Fasting and postprandial determinants for the occurrence of small dense LDL species in non-insulin-dependent diabetic patients with and without hypertriglyceridaemia: the involvement of insulin, insulin precursor species and insulin resistance. Atherosclerosis 1995; 113: 273–87
Krentz AJ. Lipoprotein abnormalities and their consequences for patients with type 2 diabetes. Diabetes Obes Metab 2003; }5 Suppl. 1: S19–27
Watts GF, Mandalia S, Brunt JN, et al. Independent associations between plasma lipoprotein subfraction levels and the course of coronary artery disease in the St. Thomas’ Atherosclerosis Regression Study (STARS). Metabolism 1993; 42: 1461–7
Tribble DL, Holl LG, Wood PD, et al. Variations in oxidative susceptibility among six low density lipoprotein subfractions of differing density and particle size. Atherosclerosis 1992; 93: 189–99
de Graaf J, Hak-Lemmers HL, Hectors MP, et al. Enhanced susceptibility to in vitro oxidation of the dense low density lipoprotein subfraction in healthy subjects. Arterioscl Thromb 1991; 11: 298–306
Griffin BA. Lipoprotein atherogenicity: an overview of current mechanisms. Proc Nutr Soc 1999; 58: 163–9
Syvanne M, Kahri J, Virtanen KS, et al. HDLs containing apolipoproteins A-I and A-II (LpA-I: A-II) as markers of coronary artery disease in men with non-insulin-dependent diabetes mellitus. Circulation 1995; 92: 364–70
Nofer JR, Kehrel B, Fobker M, et al. HDL and arteriosclerosis: beyond reverse cholesterol transport. Atherosclerosis 2002; 161: 1–16
Wilson DE, Hata A, Kwong LK, et al. Mutations in exon 3 of the lipoprotein lipase gene segregating in a family with hypertriglyceridemia, pancreatitis, and non-insulin-dependent diabetes. J Clin Invest 1993; 92: 203–11
Laakso M, Kesaniemi A, Kervinen K, et al. Relation of coronary heart disease and apolipoprotein E phenotype in patients with non-insulin dependent diabetes. BMJ 1991; 303: 1159–62
Li WW, Dammerman MM, Smith JD, et al. Common genetic variation in the promoter of the human apo CIII gene abolishes regulation by insulin and may contribute to hypertriglyceridemia. J Clin Invest 1995; 96: 2601–5
Rigoli L, Raimondo G, Di Benedetto A, et al. Apolipoprotein AI-CIII-AIV genetic polymorphisms and coronary heart disease in type 2 diabetes mellitus. Acta Diabetol 1995; 32: 251–6
Ukkola O, Savolainen MJ, Salmela PI, et al. DNA polymorphisms at the locus for human cholesteryl ester transfer protein (CETP) are associated with macro-and microangiopathy in non-insulin-dependent diabetes mellitus. Clin Genet 1994; 46: 217–27
Gotto AM. Lipid management in diabetic patients: lessons from prevention trials. Am J Med 2002; 112 Suppl. 8A: 19S-26S
Beckman JA, Creager MA, Libby P. Diabetes and atherosclerosis: epidemiology, pathophysiology, and management. JAMA 2002; 287: 2570–81
Mooradian AD. Cardiovascular disease in type 2 diabetes mellitus: current management guidelines. Arch Intern Med 2003; 163: 33–40
Grundy SM, Garber A, Goldberg R, et al. Prevention Conference VI: diabetes and cardiovascular disease. Writing Group IV: lifestyle and medical management of risk factors. Circulation 2002; 105: el53–8
Gaede P, Vedel P, Larsen N, et al. Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med 2003; 348: 383–93
National Heart Foundation of Australia and the Cardiac Society of Australia and New Zealand. Lipid management guidelines: 2001._National Heart Foundation of Australia, The Cardiac Society of Australia and New Zealand. Med J Aust 2001; 175 Suppl.: S57–85
Prevention of coronary heart disease in clinical practice: recommendations of the Second Joint Task Force of European and other Societies on coronary prevention. Eur Heart J 1998; 19: 1434–503
Expert Panel. Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). JAMA 2001; 285: 2486–97
Collins R, Armitage J, Parish S, et al. MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5963 people with diabetes: a randomised placebo-controlled trial. Heart Protection Study Collaborative Group. Lancet 2003: 361: 2005–16
Pyorala K, Pedersen TR, Kjekshus J, et al. Cholesterol lowering with simvastatin improves prognosis of diabetic patients with coronary heart disease: a subgroup analysis of the Scandinavian Simvastatin Survival Study (4S). Diabetes Care 1997; 20: 614–20
Keech A, Colquhoun D, Best J, et al. Secondary prevention of cardiovascular events with long-term pravastatin in patients with diabetes or impaired fasting glucose. Diabetes Care 2003; 26: 2713–21
Goldberg RB, Mellies MJ, Sacks FM, et al. Cardiovascular events and their reduction with pravastatin in diabetic and glucose-intolerant myocardial infarction survivors with average cholesterol levels: subgroup analyses in the cholesterol and recurrent events (CARE) trial. The Care Investigators. Circulation 1998; 98: 2513–9
Wolfram S, Jensen KS, Liao JK. Endothelium-dependent effects of statins. Arterioscler Thromb Vasc Biol 2003; 3: 29–36
Waters DD, Hsue PY. What is the role of intensive cholesterol lowering in the treatment of acute coronary syndromes? Am J Cardiol 2001; 88: 7J–16J
Takemoto M, Liao JK. Pleiotropic effects of 3-hydroxy-3-methylglutaryl coenzyme a reductase inhibitors. Arterioscler Thromb Vasc Biol 2001; 21: 1712–9
Shepherd J, Hunninghake DB, Barter P, et al. Guidelines for lowering lipids to reduce coronary artery disease risk: a com-parison of rosuvastatin with atorvastatin, pravastatin, and simvastatin for achieving lipid-lowering goals. Am J Cardiol 2003; 91: 11C-7C
Jones PH, Davidson MH, Stein EA, et al. Comparison of the efficacy and safety of rosuvastatin versus atorvastatin, simvastatin, and pravastatin across doses (STELLAR* Trial). Am J Cardiol 2003; 92: 152–60
McKenney JM, Jones PH, Adamczyk MA, et al. Comparison of the efficacy of rosuvastatin versus atorvastatin, simvastatin, and pravastatin in achieving lipid goals: results from the STELLAR trial. STELLAR Study Group. Curr Med Res Opin 2003; 19: 689–98
Liao JK. Beyond lipid lowering: the role of statins in vascular protection. Int J Cardiol 2002; 86: 5–18
Brewer Jr HB. Benefit-risk assessment of rosuvastatin 10 to 40 milligrams. Am J Cardiol 2003; 92: 23K–9K
Duez H, Frachart JC, Staels B. PPARS in inflammation, atherosclerosis and thrombosis. J Cardiovasc Risk 2001; 8: 187–94
Watts GF, Barrett PH, Ji J, et al. Differential regulation of lipoprotein kinetics by atorvastatin and fenofibrate in subjects with the metabolic syndrome. Diabetes 2003; 52: 803–11
Staels B, Dallongeville J, Auwerx J, et al. Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation 1998; 98: 2088–93
Watts GF, Dimmitt SB. Fibrates, dyslipoproteinaemia and cardiovascular disease. Curr Opin Lipidol 1999; 10: 561–74
Garg A, Grundy SM. Gemfibrozil alone and in combination with lovastatin for treatment of hypertriglyceridemia in NIDDM. Diabetes 1989; 38: 364–72
Vakkilainen J, Steiner G, Ansquer JC, et al. Relationships between low-density lipoprotein particle size, plasma lipoproteins, and progression of coronary artery disease: the Diabetes Atherosclerosis Intervention Study (DAIS). Circulation 2003; 107: 1733–7
Forcheron F, Cachefo A, Thevenon S, et al. Mechanisms of the triglyceride-and cholesterol-lowering effect of fenofibrate in hyperlipidemic type 2 diabetic patients. Diabetes 2002; 51: 3486–91
Koskinen P, Manttari M, Manninen V, et al. Coronary heart disease incidence in NIDDM patients in the Helsinki Heart Study. Diabetes Care 1992; 15: 820–5
Rubins HB, Robins SJ, Collins D, et al. Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol: Veterans Affairs High-Density Lipoprotein Cholesterol Intervention Trial Study Group. N Engl J Med 1999; 341: 410–8
Rubins HB, Robins SJ, Collins D, et al. Diabetes, plasma insulin, and cardiovascular disease: subgroup analysis from the Department of Veterans Affairs high-density lipoprotein intervention trial (VA-HIT). Arch Intern Med 2002; 162: 2597–604
Robins SJ, Collins D, Wittes JT, et al. Relation of gemfibrozil treatment and lipid levels with major coronary events. VA-HIT: a randomized controlled trial. VA HIT Study Group. JAMA 2001; 285: 1585–91
Effect of fenofibrate on progression of coronary-artery disease in type 2 diabetes: the Diabetes Atherosclerosis Intervention Study, a randomised study. Lancet 2001; 357: 905–910
Keating GM, Ormrod D. Micronised fenofibrate: an updated review of its clinical efficacy in the management of dys-lipidaemia. Drugs 2002; 62(13): 1909–44
Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto miocardico. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: results of the GISSI-Prevenzione trial. Lancet 1999; 354: 447–55
Burr ML, Fehily AM, Gilbert JF, et al. Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: Diet And Reinfarction Trial (DART). Lancet 1989; II: 757–61
Daviglus ML, Stamler J, Orencia AJ, et al. Fish consumption and the 30-year risk of fatal myocardial infarction. N Engl J Med 1997; 336: 1046–53
Kromhout D, Bosschieter EB, de Lezenne C, et al. The inverse relation between fish consumption and 20-year mortality from coronary heart disease. N Engl J Med 1985; 312: 1205–9
Norell SE, Ahlbom A, Feychting M, et al. Fish consumption and mortality from coronary heart disease. BMJ (Clin Res Ed) 1986; 293: 426
Dolecek TA. Epidemiological evidence of relationships between dietary polyunsaturated fatty acids and mortality in the multiple risk factor intervention trial. Proc Soc Exp Biol Med 1992; 200: 177–82
Rodriguez BL, Sharp DS, Abbott RD, et al. Fish intake may limit the increase in risk of coronary heart disease morbidity and mortality among heavy smokers: the Honolulu Heart Program. Circulation 1996; 94: 952–6
Albert CM, Hennekens CH, O’Donnell CJ, et al. Fish consumption and risk of sudden cardiac death. JAMA 1998; 279: 23–8
Petersen M, Pedersen H, Major-Pedersen A, et al. Effect offish oil versus corn oil supplementation on LDL and HDL subclasses in type 2 diabetic patients. Diabetes Care 2002; 25: 1704–8
Sirtori CR, Crepaldi G, Manzato E, et al. One-year treatment with ethyl esters of n-3 fatty acids in patients with hypertrig-lyceridemia and glucose intolerance: reduced triglyceridemia, total cholesterol and increased HDL-C without glycemic alterations. Atherosclerosis 1998; 137: 419–27
Goh YK, Jumpsen JA, Ryan EA, et al. Effect of omega 3 fatty acid on plasma lipids, cholesterol and lipoprotein fatty acid content in NIDDM patients. Diabetologia 1997; 40: 45–52
Montori VM, Farmer A, Wollan PC, et al. Fish oil supplementa-tion in type 2 diabetes: a quantitative systematic review. Diabetes Care 2000; 23: 1407–15
Harris WS. Fish oils and plasma lipid and lipoprotein metabolism in humans: a critical review. J Lipid Res 1989; 30: 785–807
Connor WE, Prince MJ, Ullmann D, et al. The hypotriglyceridemic effect offish oil in adult-onset diabetes without adverse glucose control. Ann N Y Acad Sci 1993; 683: 337–40
Price PT, Nelson CM, Clarke SD. Omega-3 polyunsaturated fatty acid regulation of gene expression. Curr Opin Lipidol 2000; 11: 3–7
Woodman RJ, Mori TA, Burke V, et al. Effects of purified eicosapentaenoic acid and docosahexaenoic acid on glycemic control, blood pressure and serum lipids in type 2 diabetic patients with treated hypertension. Am J Clin Nutr 2002; 76: 1007–15
Suzukawa M, Abbey M, Howe PR, et al. Effects of fish oil fatty acids on low density lipoprotein size, oxidizability, and uptake by macrophages. J Lipid Res 1995; 36: 473–84
Mori TA, Burke V, Puddey IB, et al. Purified eicosapentaenoic and docosahexaenoic acids have differential effects on serum lipids and lipoproteins, LDL particle size, glucose, and insulin in mildly hyperlipidemic men. Am J Clin Nutr 2000; 71: 1085–94
Woodman RJ, Mori TA, Burke V, et al. Docosahexaenoic acid but not eicosapentaenoic acid increases LDL particle size in treated hypertensive type 2 diabetic patients [letter]. Diabetes Care 2003; 26: 253
Canner PL, Berge KG, Wenger NK, et al. Fifteen year mortality in Coronary Drug Project patients: long-term benefit with niacin. J Am Coll Cardiol 1986; 8: 1245–55
Elam MB, Hunninghake DB, Davis KB, et al. Effect of niacin on lipid and lipoprotein levels and glycemic control in patients with diabetes and peripheral arterial disease. The ADMIT study: a randomized trial. Arterial Disease Multiple Intervention Trial. JAMA 2000; 284: 1263–70
Garg A, Grundy SM. Nicotinic acid as therapy for dyslipidemia in non-insulin-dependent diabetes mellitus. JAMA 1990; 264: 723–6
Alvarsson M, Grill V. Impact of nicotinic acid treatment on insulin secretion and insulin sensitivity in low and high insulin responders. Scand J Clin Lab Invest 1996; 56: 563–70
Wang W, Basinger A, Neese RA, et al. Effects of nicotinic acid on fatty acid kinetics, fuel selection, and pathways of glucose production in women. Am J Physiol Endocrinol Metab 2000; 279: E50–9
Grundy SM, Vega GL, McGovern ME, et al. Efficacy, safety, and tolerability of once-daily niacin for the treatment of dys-lipidemia associated with type 2 diabetes: results of the assess-ment of diabetes control and evaluation of the efficacy of niaspan trial. Diabetes Multicenter Research Group. Arch Intern Med 2002; 162: 1568–76
Bays HE, Moore PB, Drehobl MA, et al. Effectiveness and tolerability of ezetimibe in patients with primary hypercholes-terolemia: pooled analysis of two phase II studies. Ezetimibe Study Group. Clin Ther 2001; 23: 1209–30
Gagne C, Bays HE, Weiss SR, et al. Efficacy and safety of ezetimibe added to ongoing statin therapy for treatment of patients with primary hypercholesterolemia: Ezetimibe Study Group. Am J Cardiol 2002; 90: 1084–91
Knopp RH, Gitter H, Truitt T, et al. Effects of ezetimibe, a new cholesterol absorption inhibitor, on plasma lipids in patients with primary hypercholesterolemia: Ezetimibe Study Group. Eur Heart J 2003; 24: 729–41
Dujovne CA, Ettinger MP, McNeer JF, et al. Efficacy and safety of a potent new selective cholesterol absorption inhibitor, ezetimibe, in patients with primary hypercholesterolemia: Ezetimibe Study Group. Am J Cardiol 2002; 90: 1092–7
Athyros VG, Papageorgiou AA, Athyrou VV, et al. Atorvastatin and micronized fenofibrate alone and in combination in type 2 diabetes with combined hyperlipidemia. Diabetes Care. 2002; 25: 1198–202
Haffner SM, Goldberg RB. New strategies for the treatment of diabetic dyslipidemia. Diabetes Care 2002; 25: 1237–9
Jeu L, Cheng JW. Pharmacology and therapeutics of ezetimibe (SCH 58235), a cholesterol-absorption inhibitor. Clin Ther 2003; 25: 2352–87
Davidson MH, McGarry T, Bettis R, et al. Ezetimibe coadministered with simvastatin in patients with primary hypercholes-terolemia. J Am Coll Cardiol 2002; 40: 2125–34
Melani L, Mills R, Hassman D, et al. Efficacy and safety of ezetimibe coadministered with pravastatin in patients with primary hypercholesterolemia: a prospective, randomized, double-blind trial. Ezetimibe Study Group. Eur Heart J 2003; 24: 717–28
Bays HE, Dujovne CA. Drug interactions of lipid-altering drugs. Drug Saf 1998; 19: 355–71
Kaplan F, Al-Majali K, Betteridge DJ. PPARS, insulin resistance and type 2 diabetes. J Cardiovasc Risk 2001; 8: 211–7
Nass CM, Blumenthal RS. Glitazones and the potential improvement of lipid profiles in diabetes patients at high risk for cardiovascular disease. Am J Manag Care 2000; 6: S1247–56
Hsueh WA, Bruemmer D. Peroxisome proliferator-activated receptor gamma: implications for cardiovascular disease. Hypertension 2004; 43: 297–305
Rosenblatt S, Miskin B, Glazer NB, et al. The impact of pioglitazone on glycemic control and atherogenic dyslipidemia in patients with type 2 diabetes mellitus. Pioglitazone 026 Study Group. Coron Artery Dis 2001; 12: 413–23
Aronoff S, Rosenblatt S, Braithwaite S, et al. Pioglitazone hydrochloride monotherapy improves glycemic control in the treatment of patients with type 2 diabetes: a 6-month randomized placebo-controlled dose-response study. The Pioglitazone 001 Study Group. Diabetes Care 2000; 23: 1605–11
Winkler K, Friedrich I, Nauck M, et al. Pioglitazone reduces dense LDL-particles in patients with type 2 diabetes [abstract]. Diabetes 2001; 50 Suppl. 1: A147
King AB, Armstrong DU. Lipid response to pioglitazone in diabetic patients: clinical observations from a retrospective chart review. Diabetes Technol Ther 2002; 4: 145–51
Rosenstock J, Einhorn D, Hershon K, et al. Efficacy and safety of pioglitazone in type 2 diabetes: a randomised, placebo-controlled study in patients receiving stable insulin therapy. Pioglitazone 014 Study Group. Int J Clin Pract 2002; 56: 251–7
Ko SH, Song KH, Ahn YB, et al. The effect of rosiglitazone on serum lipoprotein(a) levels in Korean patients with type 2 diabetes mellitus. Metabolism 2003; 52: 731–4
Freed MI, Ratner R, Marcovina SM, et al. Effects of rosiglitazone alone and in combination with atorvastatin on the metabolic abnormalities in type 2 diabetes mellitus. Rosiglitazone Study 108 investigators. Am J Cardiol 2002; 90: 947–52
Tack CJ, Smits P, Demacker PN, et al. Troglitazone decreases the proportion of small, dense LDL and increases the resis-tance of LDL to oxidation in obese subjects. Diabetes Care 1998; 21: 796–9
Shirai K, Itoh Y, Sasaki H, et al. The effect of insulin sensitizer, troglitazone, on lipoprotein lipase mass in preheparin serum. Diabetes Res Clin Pract 1999; 46: 35–41
Olansky L, Marchetti A, Lau H. Multicenter retrospective as-sessment of thiazolidinedione monotherapy and combination therapy in patients with type 2 diabetes: comparative subgroup analyses of glycemic control and blood lipid levels. Clin Ther 2003; 25 Suppl. B: B64-80
Gegick CG, Altheimer MD. Comparison of effects of thiazolidinediones on cardiovascular risk factors: observations from a clinical practice. Endocr Pract 2001; 7: 162–9
Khan MA, St Peter JV, Xue JL. A prospective, randomized comparison of the metabolic effects of pioglitazone or rosiglitazone in patients with type 2 diabetes who were previously treated with troglitazone. Diabetes Care 2002; 25: 708–11
Delea TE, Edelsberg JS, Hagiwara M, et al. Use of thiazolidenediones and risk of heart failure in people with type 2 diabetes: a retrospective study. Diabetes Care 2004; 26: 2983–9
Wulffele MG, Kooy A, de Zeeuw D, et al. The effect of metformin on blood pressure, plasma cholesterol and triglycerides in type 2 diabetes mellitus: a systematic review. J Intern Med 2004; 256: 1–14
Giannarelli R, Aragona M, Coppelli A, et al. Reducing insulin resistance with metformin: the evidence today. Diabetes Metab 2003; 29: 6S28-9
Li XP, Zhao SP, Zhang XY, et al. Protective effect of high density lipoprotein on endothelium-dependent vasodilatation. Int J Cardiol 2000; 73: 231–6
Toikka JO, Ahotupa M, Viikari JS, et al. Constantly low HDL-cholesterol concentration relates to endothelial dysfunction and increased in vivo LDL-oxidation in healthy young men. Atherosclerosis 1999; 147: 133–8
Watts GF, Burnett JR. An ABC of HDL: a paradigm shift in managing lipid disorders. Med Today 2003; 4: 44–53
Barter PJ, Brewer HBJ, Chapman MJ, et al. Cholesteryl ester transfer protein: a novel target for raising HDL and inhibiting atherosclerosis. Arterioscler Thromb Vasc Biol 2003; 23: 160–7
Lund EG, Menke JG, Sparrow CP. Liver X receptor agonists as potential therapeutic agents for dyslipidemia and atherosclerosis. Arterioscler Thromb Vasc Biol 2003; 23: 1169–77
Laffitte BA, Chao LC, Li J, et al. Activation of liver X receptor improves glucose tolerance through coordinate regulation of glucose metabolism in liver and adipose tissue. Proc Natl Acad Sci U S A 2003; 100: 5419–24
Lin KY, Ito A, Asagami T, et al. Impaired nitric oxide synthase pathway in diabetes mellitus: role of asymmetric dimethy-larginine and dimethylarginine dimethylaminohydrolase. Circulation 2002; 106: 987–92
Fenster BE, Tsao PS, Rockson SG. Endothelial dysfunction: clinical strategies for treating oxidant stress. Am Heart J 2003; 146: 218–26
Christison J, Karjalainen A, Brauman J, et al. Rapid reduction and removal of HDL-but not LDL-associated cholesteryl ester hydroperoxides by rat liver perfused in situ. Biochem J 1996; 314: 739–42
Spitaler MM, Graier WF. Vascular targets of redox signalling in diabetes mellitus. Diabetologia 2002; 45: 476–94
Giugliano D, Ceriello A, Paolisso G. Oxidative stress and diabetic vascular complications. Diabetes Care 1996; 19: 257–67
Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001; 414: 813–20
Guzik TJ, Mussa S, Gastaldi D, et al. Mechanisms of increased vascular Superoxide production in human diabetes mellitus: role of NAD(P)H oxidase and endothelial nitric oxide synthase. Circulation 2002; 105: 1656–62
Hayaishi-Okano R, Yamasaki Y, Kajimoto Y, et al. Association of NAD(P)H oxidase p22 phox gene variation with advanced carotid atherosclerosis in Japanese type 2 diabetes. Diabetes Care 2003; 26: 458–63
Vijayalingam S, Parthiban A, Shanmugasundaram KR, et al. Abnormal antioxidant status in impaired glucose tolerance and non-insulin-dependent diabetes mellitus. Diabet Med 1996; 13: 715–9
Sundaram RK, Bhaskar A, Vijayalingam S, et al. Antioxidant status and lipid peroxidation in type II diabetes mellitus with and without complications. Clin Sci 1996; 90: 255–60
Bhatia S, Shukla R, Venkata Madhu S, et al. Antioxidant status, lipid peroxidation and nitric oxide end products in patients of type 2 diabetes mellitus with nephropathy. Clin Biochem 2003; 36: 557–62
Memisogullari R, Taysi S, Bakan E, et al. Antioxidant status and lipid peroxidation in type II diabetes mellitus. Cell Biochem Funct 2003;21: 291–6
Raitakari OT, Pitkanen OP, Lehtimaki T, et al. In vivo low density lipoprotein oxidation relates to coronary reactivity in young men. J Am Coll Cardiol 1997; 30: 97–102
Anderson TJ, Meredith IT, Yeung AC, et al. The effect of cholesterol-lowering and antioxidant therapy on endothelium-dependent coronary vasomotion. N Engl J Med 1995; 332: 488–93
O’Brien SF, Watts GF, Playford DA, et al. Low-density lipoprotein size, high-density lipoprotein concentration, and endo-thelial dysfunction in non-insulin-dependent diabetes. Diabet Med 1997; 14: 974–8
Steinberg HO, Baron AD. Vascular function, insulin resistance and fatty acids. Diabetologia 2002; 45: 623–34
Kawamura M, Heinecke JW, Chait A. Pathophysiological concentrations of glucose promote oxidative modification of low density lipoprotein by a superoxide-dependent pathway. J Clin Invest 1994; 94: 771–8
Bowie A, Owens D, Collins P, et al. Glycosylated low density lipoprotein is more sensitive to oxidation: implications for the diabetic patient? Atherosclerosis 1993; 102: 63–7
Lopes-Virella MF, Virella G. Modified lipoproteins, cytokines and macrovascular disease in non-insulin-dependent diabetes mellitus. Ann Med 1996; 28: 347–54
Picard S. Lipoprotein glyco-oxidation. Diabete Metab 1995; 21: 89–94
Liao JK, Shin WS, Lee WY, et al. Oxidized low-density lipoprotein decreases the expression of endothelial nitric oxide synthase. J Biol Chem 1995; 270: 319–24
Mukherjee S, Coaxum SD, Maleque M, et al. Effects of oxidized low density lipoprotein on nitric oxide synthetase and protein kinase C activities in bovine endothelial cells. Cell Mol Biol 2001; 47: 1051–8
Liao JK. Inhibition of Gi proteins by low density lipoprotein attenuates bradykinin-stimulated release of endothelial-derived nitric oxide. J Biol Chem 1994; 269: 12987–92
Parhami F, Fang ZT, Yang B, et al. Stimulation of Gs and inhibition of Gi protein functions by minimally oxidized LDL. Arterioscler Thromb Vasc Biol 1995; 15: 2019–24
Jessup W. Oxidized lipoproteins and nitric oxide. Curr Opin Lipidol 1996; 7: 274–80
Ju H, Zou R, Venema VJ, et al. Direct interaction of endothelial nitric-oxide synthase and caveolin-1 inhibits synthase activity. J Biol Chem 1997; 272: 18522–5
Garcia-Cardena G, Martasek P, Masters BS, et al. Dissecting the interaction between nitric oxide synthase (NOS) and caveolin: functional significance of the nos caveolin binding domain in vivo. J Biol Chem 1997; 272: 25437–40
Michel JB, Feron O, Sase K, et al. Caveolin versus calmodulin: counterbalancing allosteric modulators of endothelial nitric oxide synthase. J Biol Chem 1997; 272: 25907–12
Bist A, Fielding PE, Fielding CJ. Two sterol regulatory element-like sequences mediate up-regulation of caveolin gene transcription in response to low density lipoprotein free cholesterol. Proc Nat Acad Sci U S A 1997; 94: 10693–8
Boulanger CM, Tanner FC, Bea ML, et al. Oxidized low density lipoproteins induce mRNA expression and release of endothelin from human and porcine endothelium. Circ Res 1992; 70: 1191–7
Pritchard KAJ, Groszek L, Smalley DM, et al. Native low-density lipoprotein increases endothelial cell nitric oxide synthase generation of Superoxide anion. Circ Res 1995; 77: 510–8
Boger RH, Sydow K, Borlak J, et al. LDL cholesterol upregulates synthesis of asymmetrical dimethylarginine in human endothelial cells: involvement of S-adenosylmethionine-dependent methyltransferases. Circ Res 2000; 87: 99–105
Takahara N, Kashiwagi A, Nishio Y, et al. Oxidized lipoproteins found in patients with NIDDM stimulate radical-induced monocyte chemoattractant protein-1 mRNA expression in cultured human endothelial cells. Diabetologia 1997; 40: 662–70
Doi H, Kugiyama K, Ohgushi M, et al. Remnants of chylomicron and very low density lipoprotein impair endothe-lium-dependent vasorelaxation. Atherosclerosis 1998; 137: 341–9
Vogel RA, Corretti MC, Plotnick GD. Effect of a single high-fat meal on endothelial function in healthy subjects. Am J Cardiol 1997; 79: 350–4
Lundman P, Eriksson M, Schenck-Gustafsson K, et al. Transient triglyceridemia decreases vascular reactivity in young, healthy men without risk factors for coronary heart disease. Circulation 1997; 96: 3266–8
Grieve DJ, Avella MA, Elliott J, et al. The influence of chylomicron remnants on endothelial cell function in the isolated perfused rat aorta. Atherosclerosis 1998; 139: 273–81
Kugiyama K, Doi H, Motoyama T, et al. Association of remnant lipoprotein levels with impairment of endothelium-dependent vasomotor function in human coronary arteries. Circulation 1998; 97: 2519–26
Nestel PJ, Shige H, Pomeroy S, et al. Post-prandial remnant lipids impair arterial compliance. J Am Coll Cardiol 2001; 37: 1929–35
Plotnick GD, Corretti MC, Vogel RA. Effect of antioxidant vitamins on the transient impairment of endothelium-dependent brachial artery vasoactivity following a single high-fat meal. JAMA 1997; 278: 1682–6
Kugiyama K, Motoyama T, Doi H, et al. Improvement of endothelial vasomotor dysfunction by treatment with alpha-tocopherol in patients with high remnant lipoproteins levels. J Am Coll Cardiol 1999; 33: 1512–8
Bae JH, Bassenge E, Kim KB, et al. Postprandial hypertriglyceridemia impairs endothelial function by enhanced oxidant stress. Atherosclerosis 2001; 155: 517–23
Evans M, Anderson RA, Graham J, et al. Ciprofibrate therapy improves endothelial function and reduces postprandial lipemia and oxidative stress in type 2 diabetes mellitus. Circulation 2000; 101: 1773–9
Ceriello A, Taboga C, Tonutti L, et al. Evidence for an independent and cumulative effect of postprandial hypertriglyceridemia and hyperglycemia on endothelial dysfunction and oxidative stress generation: effects of short-and long-term simvastatin treatment. Circulation 2002; 106: 1211–8
Navab M, Berliner JA, Watson AD, et al. The Yin and Yang of oxidation in the development of the fatty streak: a review based on the 1994 George Lyman Duff Memorial Lecture. Arterioscl Thromb Vasc Biol 1996; 16: 831–42
Parthasarathy S, Barnett J, Fong LG. High-density lipoprotein inhibits the oxidative modification of low-density lipoprotein. Biochim Biophys Acta 1990; 1044: 275–83
Mackness MI, Abbott C, Arrol S, et al. The role of high-density lipoprotein and lipid-soluble antioxidant vitamins in inhibiting low-density lipoprotein oxidation. Biochem J 1993; 294: 829–34
Barter PJ, Rye KA. High density lipoproteins and coronary heart disease. Atherosclerosis 1996; 121: 1–12
Klimov AN, Gurevich VS, Nikiforova AA, et al. Antioxidative activity of high density lipoproteins in vivo. Atherosclerosis 1993; 100: 13–8
Mackness MI, Durrington PN. HDL, its enzymes and its potential to influence lipid peroxidation. Atherosclerosis 1995; 115: 243–53
Abbott CA, Mackness MI, Kumar S, et al. Serum paraoxonase activity, concentration, and phenotype distribution in diabetes mellitus and its relationship to serum lipids and lipoproteins. Arterioscler Thromb Vasc Biol 1995; 15: 1812–8
Mackness MI, Durrington PN. Paraoxonase: another factor in NIDDM cardiovascular disease. Lancet 1995; 346: 856
Odawara M, Tachi Y, Yamashita K. Paraoxonase polymorphism (Gln192-Arg) is associated with coronary heart disease in Japanese noninsulin-dependent diabetes mellitus. J Clin Endocrinol Metab 1997; 82: 2257–60
Fleisher LN, Tall AR, Witte LD, et al. Stimulation of arterial endothelial cell prostacyclin synthesis by high density lipoproteins. J Biol Chem 1982; 257: 6653–5
Galle J, Ochslen M, Schollmeyer P, et al. Oxidized lipoproteins inhibit endothelium-dependent vasodilation: effects of pressure and high-density lipoprotein. Hypertension 1994; 23: 556–64
Zeiher AM, Schachlinger V, Hohnloser SH, et al. Coronary atherosclerotic wall thickening and vascular reactivity in humans: elevated high-density lipoprotein levels ameliorate abnormal vasoconstriction in early atherosclerosis. Circulation 1994; 89: 2525–32
Bisoendial RJ, Hovingh GK, Levels JH, et al. Restoration of endothelial function by increasing high-density lipoprotein in subjects with isolated low high-density lipoprotein. Circulation 2003; 107: 2944–8
Maier JA, Barenghi L, Pagani F, et al. The protective role of high-density lipoprotein on oxidized-low-density-lipoprotein-induced U937/endothelial cell interactions. Eur J Biochem 1994; 221: 35–41
Spieker LE, Sudano I, Hurlimann D, et al. High-density lipoprotein restores endothelial function in hypercholesterolemic men. Circulation 2002; 105: 1399–402
Syvanne M, Castro G, Dengremont C, et al. Cholesterol efflux from Fu5AH hepatoma cells induced by plasma of subjects with or without coronary artery disease and non-insulin-dependent diabetes: importance of LpA-I: A-II particles and phospholipid transfer protein. Atherosclerosis 1996; 127: 245–53
Calabresi L, Gomaraschi M, Franceschini G. Endothelial protection by high-density lipoproteins: from bench to bedside. Arterioscler Thromb Vasc 2003; 23: 1724–31
Steinberg HO, Tarshoby M, Monestel R, et al. Elevated circulating free fatty acid levels impair endothelium-dependent vasodilation. J Clin Invest 1997; 100: 1230–9
Steinberg HO, Paradisi G, Hook G, et al. Free fatty acid elevation impairs insulin-mediated vasodilation and nitric oxide production. Diabetes 2000; 49: 1231–8
Inoguchi T, Li P, Umeda F, et al. High glucose level and free fatty acid stimulate reactive oxygen species production through protein kinase C: dependent activation of NAD(P)H oxidase in cultured vascular cells. Diabetes 2000; 49: 1939–45
Balletshofer BM, Rittig K, Volk A, et al. Impaired nonesterified fatty acid suppression is associated with endothelial dysfunction in insulin resistant subjects. Horm Metab Res 2001; 33: 428–31
Sarabi M, Fugmann A, Karlstrom B, et al. An ordinary mixed meal transiently impairs endothelium-dependent vasodilation in healthy subjects. Acta Physiol Scand 2001; 172: 107–13
Harris WS. n-3 fatty acids and lipoproteins: comparison of results from human and animal studies. Lipids 1996; 31: 243–52
Nestel PJ. Fish oil and cardiovascular disease: lipids and arterial function. Am J Clin Nutr 2000; 71: 228S–31S
Caslake MJ, Stewart G, Day SP, et al. Phenotype-dependent and -independent actions of rosuvastatin on atherogenic lipoprotein subfractions in hyperlipidaemia. Atherosclerosis 2003 Dec; 171(2): 245–53
Wilmink HW, Twickler MB, Banga JD, et al. Effect of statin versus fibrate on postprandial endothelial dysfunction: role of remnant-like particles. Cardiovasc Res 2001; 50: 577–82
Agren JJ, Hanninen O, Julkunen A, et al. Fish diet, fish oil and docosahexaenoic acid rich oil lower fasting and postprandial plasma lipid levels. Eur J Clin Nutr 1996; 50: 765–71
Fruchart JC, Staels B, Duriez P. The role of fibric acids in atherosclerosis. Curr Atheroslerosis Rep 2001; 3: 83–92
De Caterina R, Zampolli A. n-3 fatty acids: antiatherosclerotic effects. Lipids 2001; 36 Suppl.: S69–78
Das UN. Beneficial effect(s) of n-3 fatty acids in cardiovascular diseases: but, why and how? Prostaglandins Leukot Essent Fatty Acids 2000; 63: 351–62
Duval C, Chinetti G, Trottein F, et al. The role of PPARs in atherosclerosis. Trends Mol Med 2002; 8: 422–30
McVeigh GE, Brennan GM, Johnston GD, et al. Dietary fish oil augments nitric oxide production or release in patients with type 2 (non-insulin-dependent) diabetes mellitus. Diabetologia 1993; 36: 33–8
Chin JP, Dart AM. How do fish oils affect vascular function? Clin Exp Pharmacol Physiol 1995; 22: 71–81
Kohno M, Yokokawa K, Horio T, et al. Release mechanism of endothelin-1 and big endothelin-1 after stimulation with thrombin in cultured porcine endothelial cells. J Vasc Res 1992; 29: 56–63
Christensen MS, Therkelsen K, Moller JM, et al. n-3 fatty acids do not decrease plasma endothelin levels in healthy individuals. Scan J Clin Lab Invest 1997; 57: 495–9
Playford DA, Watts GF, Croft KD, et al. Combined effect of coenzyme Q(10) and fenofibrate on forearm microcirculatory function in type 2 diabetes. Atherosclerosis 2003; 168: 169–79
Mori TA, Woodman RJ, Burke V, et al. Effect of eicosapentaenoic acid and docosahexaenoic acid on oxidative stress and inflammatory markers in treated-hypertensive type 2 diabetic subjects. Free Radie Biol Med 2003; 35: 772–81
Stroes ES, Koomans HA, de Bruin TW, et al. Vascular function in the forearm of hypercholesterolaemic patients off and on lipid-lowering medication. Lancet 1995; 346: 467–71
O’Driscoll G, Green D, Taylor RR. Simvastatin, an HMG-coenzyme A reductase inhibitor, improves endothelial function within 1 month. Circulation 1997; 95: 1126–31
Treasure CB, Klein JL, Weintraub WS, et al. Beneficial effects of cholesterol-lowering therapy on the coronary endothelium in patients with coronary artery disease. N Engl J Med 1995; 332: 481–7
Kontopoulos AG, Athyros VG, Pehlivanidis AN, et al. Long-term treatment effect of atorvastatin on aortic stiffness in hypercholesterolaemic patients. Curr Med Res Opin 2003; 19: 22–7
Leibovitz E, Hazanov N, Zimlichman R, et al. Treatment with atorvastatin improves small artery compliance in patients with severe hypercholesterolemia. Am J Hypertens 2001; 14: 1096–8
Raison J, Rudnichi A, Safar ME. Effects of atorvastatin on aortic pulse wave velocity in patients with hypertension and hypercholesterolaemia: a preliminary study. J Hum Hypertens 2002; 16: 705–10
Smilde TJ, van den Berkmortel FW, Wollersheim H, et al. The effect of cholesterol lowering on carotid and femoral artery wall stiffness and thickness in patients with familial hypercho-lesterolaemia. Eur J Clin Invest 2000; 30: 473–80
Karter Y, Curgunlu A, Erturk N, et al. Effects of low and high doses of atorvastatin on arterial compliance. Jpn Heart J 2003; 44: 953–61
Economides PA, Caselli A, Tiani E, et al. The effects if atorvastatin on endothelial function in diabetic patients and subjects at risk for type 2 diabetes. J Clin Endocrinol Metab 2004; 89: 740–7
Ichihara A, Hayashi M, Ryuzaki M, et al. Fluvastatin prevents development of arterial stiffness in haemodialysis patients with type 2 diabetes mellitus. Nephrol Dial Transplant 2002; 17: 1513–7
van Venrooij FV, van de Ree MA, Bots ML, et al. Aggressive lipid lowering does not improve endothelial function in type 2 diabetes: the Diabetes Atorvastatin Lipid Intervention (DALI) Study. A randomized, double-blind, placebo-controlled trial. DALI Study Group. Diabetes Care 2002; 25: 1211–6
van Etten RW, de Koning EJ, Honing ML, et al. Intensive lipid lowering by statin therapy does not improve vasoreactivity in patients with type 2 diabetes. Arterioscler Thromb Vasc Biol 2002; 22: 799–804
Tan KC, Chow WS, Tam SC, et al. Atorvastatin lowers C-reactive protein and improves endothelium-dependent vasodilation in type 2 diabetes mellitus. J Clin Endocrinol Metab 2002; 87: 563–8
Sheu WH, Chen YT, Lee WJ. Improvement in endothelial dysfunction with LDL cholesterol level <80 mg/dl in type 2 diabetic patients. Diabetes Care 2001; 24: 1499–501
van de Ree MA, Huisman MV, de Man FH, et al. Impaired endothelium-dependent vasodilation in type 2 diabetes mellitus and the lack of effect of simvastatin. Cardiovasc Res 2001; 52: 299–305
Tsunekawa T, Hayashi T, Kano H, et al. Cerivastatin, a hydroxymethylglutaryl coenzyme a reductase inhibitor, improves endothelial function in elderly diabetic patients within 3 days. Circulation 2001; 104: 376–9
Mansourati J, Newman LG, Roman SH, et al. Lipid lowering does not improve endothelial function in subjects with poorly controlled diabetes. Diabetes Care 2001; 24: 2152–3
Sheu WH, Juang BL, Chen YT, et al. Endothelial dysfunction is not reversed by simvastatin treatment in type 2 diabetic patients with hypercholesterolemia. Diabetes Care 1999; 22: 1224–5
Fichtischerer S, Rosenberger G, Walter DH, et al. Elevated C-reactive protein levels and impaired endothelial vasoreactivity in patients with coronary artery disease. Circulation 2000; 102: 1000–6
Jialal I, Stein D, Balis D, et al. Effect of hydroxymethyl glutaryl coenzyme A reductase inhibitor therapy on high sensitive C-reactive protein levels. Circulation 2001; 103: 1933–5
Bhakdi S, Torzewski M, Klouche M, et al. Complement and atherogenesis: binding of CRP to degraded, nonoxidized LDL enhances complement activation. Arterioscler Thromb Vasc Biol 1999; 19: 2348–54
Pasceri V, Willerson JT, Yeh ET. Direct proinflammatory effect of C-reactive protein on human endothelial cells. Circulation 2000; 102: 2165–8
Seiler C, Suter TM, Hess OM. Exercise-induced vasomotion of angiographically normal and stenotic coronary arteries improves after cholesterol-lowering drug therapy with bezafibrate. J Am Coll Cardiol 1995; 26: 1615–22
Avogaro A, Miola M, Favaro A, et al. Gemfibrozil improves insulin sensitivity and flow-mediated vasodilatation in type 2 diabetic patients. Eur J Clin Invest 2001; 31: 603–9
Playford DA, Watts GF, Best JD, et al. Effect of fenofibrate on brachial artery flow-mediated dilatation in type 2 diabetes mellitus. Am J Cardiol 2002; 90: 1254–7
Woodman RJ, Mori TA, Burke V, et al. Effects of purified eicosapentaenoic acid and docosahexaenoic acid on platelet, fibrinoloytic and vascular function in hypertensive type 2 diabetic patients. Atherosclerosis 2003; 166: 85–93
McVeigh GE, Brennan GM, Cohn JN, et al. Fish oil improves arterial compliance in non-insulin-dependent diabetes mellitus. Arterioscler Thromb 1994; 14: 1425–9
Wahlqvist ML, Lo CS, Myers KA. Fish intake and arterial wall characteristics in healthy people and diabetic patients. Lancet 1989; II: 944–6
Capell WH, DeSouza CA, Poirier P, et al. Short-term triglyceride lowering with fenofibrate improves vasodilator function in subjects with hypertriglyceridemia. Arterioscler Thromb Vasc Biol 2003; 23: 307–13
Malik J, Melenovsky V, Wichterle D, et al. Both fenofibrate and atorvastatin improve vascular reactivity in combined hyperlipidaemia (fenofibrate versus atorvastatin trial: FAT). Cardiovasc Res 2001; 52: 290–8
Wang TD, Chen WJ, Lin JW, et al. Efficacy of fenofibrate and simvastatin on endothelial function and inflammatory markers in patients with combined hyperlipidemia: relations with baseline lipid profiles. Atherosclerosis 2003; 170: 315–23
Barbier O, Torra IP, Duguay Y, et al. Pleiotropic actions of peroxisome proliferator-activated receptors in lipid metabolism and atherosclerosis. Arterioscler Thromb Vasc Biol 2002; 22: 717–26
Goodfellow J, Bellamy MF, Ramsey MW, et al. Dietary supplementation with marine omega-3 fatty acids improve systemic large artery endothelial function in subjects with hypercholesterolemia. J Am Coll Cardiol 2000; 35: 265–70
Andreassen AK, Hartmann A, Offstad J, et al. Hypertension prophylaxis with omega-3 fatty acids in heart transplant recipients. J Am Coll Cardiol 1997; 29: 1324–31
Fleischhauer FJ, Yan WD, Fischeil TA. Fish oil improves endothelium-dependent coronary vasodilation in heart transplant recipients. J Am Coll Cardiol 1993; 21: 982–9
Mori TA, Watts GF, Burke V, et al. Differential effects of eicosapentaenoic acid and docosahexaenoic acid on vascular reactivity of the forearm microcirculation in hyperlipidemic, overweight men. Circulation 2000; 102: 1264–9
Takase H, Sugiyama M, Nakazawa A, et al. Long-term effect of antihypertensive therapy with calcium antagonist or angiotensin converting enzyme inhibitor on serum nitrite/nitrate levels in human essential hypertension. Arzneimittelforsch 2000; 50: 530–4
Ghiadoni L, Virdis A, Magagna A, et al. Effect of the angiotensin II type 1 receptor blocker candesartan on endothelial function in patients with essential hypertension. Hypertension 2000; 35: 501–6
Mather KJ, Verma S, Anderson TJ. Improved endothelial function with metformin in type 2 diabetes mellitus. J Am Coll Cardiol 2001; 37: 1344–50
Yamamoto H, Yoshimura H, Noma M, et al. Improvement of coronary vasomotion with eicosapentaenoic acid does not inhibit acetylcholine-induced coronary vasospasm in patients with variant angina. Jpn Circ J 1995; 59: 608–16
Mullen MJ, Kharbanda RK, Cross J, et al. Heterogenous nature of flow-mediated dilatation in human conduit arteries in vivo: relevance to endothelial dysfunction in hypercholesterolemia. Circ Res 2001;88: 145–51
Purasiri P, Murray A, Richardson S, et al. Modulation of cytokine production in vivo by dietary essential fatty acids in patients with colorectal cancer. Clin Sci 1994; 87: 711–7
Mori TA, Dunstan DW, Burke V, et al. Effect of dietary fish and exercise training on urinary F-2-isoprostane excretion in non-insulin-dependent diabetic patients. Metabolism 1999; 48: 1402–8
Guyton JR. Effect of niacin on atherosclerotic cardiovascular disease. Am J Cardiol 1998; 82: 18–23U
Kuvin JT, Ramet ME, Patel AR, et al. A novel mechanism for the beneficial vascular effects of high-density lipoprotein cholesterol: enhanced vasorelaxation and increased endothelial nitric oxide synthase expression. Am Heart J 2002; 144: 165–72
Andrews TC, Whitney EJ, Green G, et al. Effect of gemfibrozil +/−niacin +/−cholestyramine on endothelial function in patients with serum low-density lipoprotein cholesterol levels <160 mg/dl and high-density lipoprotein cholesterol levels <40 mg/dl. Am J Cardiol 1997; 80: 831–5
Evans M, Anderson RA, Smith JC, et al. Effects of insulin lispro and chronic vitamin C therapy on postprandial lipaemia, oxidative stress and endothelial function in patients with type 2 diabetes mellitus. Eur J Clin Invest 2003; 33: 231–8
Caballero AE, Saouaf R, Lim SC, et al. The effects of troglitazone, an insulin-sensitizing agent, on the endothelial function in early and late type 2 diabetes: a placebo-controlled randomized clinical trial. Metabolism 2003; 52: 173–80
van Etten RW, de Koning EJ, Verhaar MC, et al. Impaired NO-dependent vasodilation in patients with type II (non-insulin-dependent) diabetes mellitus is restored by acute administration of folate. Diabetologia 2002; 45: 1004–10
Watts GF, Playford DA, Croft KD, et al. Coenzyme Q(10) improves endothelial dysfunction of the brachial artery in type II diabetes mellitus. Diabetologia 2002; 45: 420–6
Gaenzer H, Neumayr G, Marschang P, et al. Effect of insulin therapy on endothelium-dependent dilation in type 2 diabetes mellitus. Am J Cardiol 2002; 89: 431–4
Vehkavaara S, Makimattila S, Schlenzka A, et al. Insulin therapy improves endothelial function in type 2 diabetes. Arterioscler Thromb Vasc Biol 2000; 20: 545–50
Cheetham C, O’Driscoll G, Stanton K, et al. Losartan, an angiotensin type I receptor antagonist, improves conduit vessel endothelial function in type II diabetes. Clin Sci 2001; 100: 13–7
Cheetham C, Collis J, O’Driscoll G, et al. Losartan, an angiotensin type 1 receptor antagonist, improves endothelial function in non-insulin-dependent diabetes. J Am Coll Cardiol 2000; 36: 1461–6
Maiorana A, O’Driscoll G, Cheetham C, et al. The effect of combined aerobic and resistance exercise training on vascular function in type 2 diabetes. J Am Coll Cardiol 2001; 38: 860–6
Butler R, Morris AD, Belch JJ, et al. Allopurinol normalizes endothelial dysfunction in type 2 diabetics with mild hypertension. Hypertension 2000; 35: 746–51
Chowienczyk PJ, Brett SE, Gopaul NK, et al. Oral treatment with an antioxidant (raxofelast) reduces oxidative stress and improves endothelial function in men with type II diabetes. Diabetologia 2000; 43: 974–7
Heitzer T, Krohn K, Albers S, et al. Tetrahydrobiopterin improves endothelium-dependent vasodilation by increasing nitric oxide activity in patients with type II diabetes mellitus. Diabetologia 2000; 43: 1435–8
Heitzer T, Finckh B, Albers S, et al. Beneficial effects of alpha-lipoic acid and ascorbic acid on endothelium-dependent, nitric oxide-mediated vasodilation in diabetic patients: relation to parameters of oxidative stress. Free Radie Biol Med 2001; 31: 53–61
O’Driscoll G, Green D, Maiorana A, et al. Improvement in endothelial function by angiotensin-converting enzyme inhibition in non-insulin-dependent diabetes mellitus. J Am Coll Cardiol 1999; 33: 1506–11
Ting HH, Timimi FK, Boles KS, et al. Vitamin C improves endothelium-dependent vasodilation in patients with non-insulin-dependent diabetes mellitus. J Clin Invest 1996; 97: 22–8
Asplund K. Antioxidant vitamins in the prevention of cardiovascular disease: a systematic review. J Intern Med 2002; 251: 372–92
Lonn E, Yusuf S, Hoogwerf B, et al. Effects of vitamin E on cardiovascular and microvascular outcomes in high-risk patients with diabetes: results of the HOPE study and MICRO-HOPE substudy. Diabetes Care 2002; 25: 1919–27
Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of antioxidant vitamin supplementation in 20,536 high-risk individuals: a randomised placebo-controlled trial. Lancet 2002; 360: 23–33
Stephens NG, Parsons A, Schofield PM, et al. Randomised controlled trial of vitamin E in patients with coronary disease: Cambridge Heart Antioxidant Study (CHAOS). Lancet 1996; 347: 781–6
Gazis A, White DJ, Page SR, et al. Effect of oral vitamin E (alpha-tocopherol) supplementation on vascular endothelial function in type 2 diabetes mellitus. Diabet Med 1999; 16: 304–11
Darko D, Dornhorst A, Kelly FJ, et al. Lack of effect of oral vitamin C on blood pressure, oxidative stress and endothelial function in type II diabetes. Clin Sci 2002; 103: 339–44
Perticone F, Ceravolo R, Maio R, et al. Effects of atorvastatin and vitamin C on endothelial function of hypercholesterolemic patients. Atherosclerosis 2000; 152: 511–8
Watts GF, Castelluccio C, Rice-Evans C, et al. Plasma coenzyme Q (ubiquinone) concentrations in patients treated with simvastatin. J Clin Pathol 1993; 46: 1055–7
Hodgson JM, Watts GF, Playford DA, et al. Coenzyme Q10 improves blood pressure and glycaemic control: a controlled trial in subjects with type 2 diabetes. Eur J Clin Nutr 2002; 56: 1137–42
Mullan BA, Young IS, Fee H, et al. Ascorbic acid reduces blood pressure and arterial stiffness in type 2 diabetes. Hypertension 2002; 40: 804–9
De Mattia G, Ferri C, Laurenti O, et al. Circulating catecholamines and metabolic effects of captopril in NIDDM patients. Diabetes Care 1996; 19: 226–30
de Gasparo M. Angiotensin II and nitric oxide interaction. Heart Fail Rev 2002; 7: 347–58
Asmar R, Gosse P, Topouchian J, et al. Effects of telmisartan on arterial stiffness in type 2 diabetes patients with essential hypertension. J Renin Angiotensin Aldosterone Syst 2002; 3: 176–80
Warnholtz A, Nickenig G, Schulz E, et al. Increased NADH-oxidase-mediated Superoxide production in the early stages of atherosclerosis: evidence for involvement of the renin-angiotensin system. Circulation 1999; 99: 2027–33
Wolffenbuttel BH, Boulanger CM, Crijns FR, et al. Breakers of advanced glycation end products restore large artery properties in experimental diabetes. Proc Natl Acad Sci U S A 1998; 95: 4630–4
Kass DA, Shapiro EP, Kawaguchi M, et al. Improved arterial compliance by a novel advanced glycation end-product crosslink breaker. Circulation 2001; 104: 1464–70
Stout RW. Insulin as a mitogenic factor: role in the pathogenesis of cardiovascular disease. Am J Med 1991; 90: 62S–5S
Sowers JR. Insulin resistance, hyperinsulinemia, dyslipidemia, hypertension, and accelerated atherosclerosis. J Clin Pharmacol 1992; 32: 529–35
Landin K, Stigendal L, Eriksson E, et al. Abdominal obesity is associated with an impaired fibrinolytic activity and elevated plasminogen activator inhibitor-1. Metabolism 1990; 39: 1044–8
Juhan-Vague I, Alessi MC, Vague P. Increased plasma plasminogen activator inhibitor 1 levels: a possible link between insulin resistance and atherothrombosis. Diabetologia 1991: 34: 457–62
Festa A, D’Agostino RJ, Mykkanen L, et al. Relative contribution of insulin and its precursors to fibrinogen and PAI-1 in a large population with different states of glucose tolerance: the Insulin Resistance Atherosclerosis Study (IRAS). Arterioscler Thromb Vasc Biol 1999; 19: 562–8
Westerbacka J, Wilkinson I, Cockcroft J, et al. Diminished wave reflection in the aorta: a novel physiological action of insulin on large blood vessels. Hypertension 1999; 33: 1118–22
Arcaro G, Cretti A, Balzano S, et al. Insulin causes endothelial dysfunction in humans: sites and mechanisms. Circulation 2002; 105: 576–82
Yudkin JS, Panahloo A, Stehouwer C, et al. The influence of improved glycaemic control with insulin and sulphonylureas on acute phase and endothelial markers in type II diabetic subjects. Diabetologia 2000; 43: 1099–106
Sundaresan P, Lykos D, Daher A, et al. Acute effects of oral glibenclamide on blood pressure and forearm vascular resistance in diabetics. Clin Exp Pharmacol Physiol 1998; 25: 170–4
Pinkney JH, Stehouwer CD, Coppack SW, et al. Endothelial dysfunction: cause of the insulin resistance syndrome. Diabetes 1997; 46 Suppl. 2: S9-13
Tack CJ, Ong MK, Lutterman JA, et al. Insulin-induced vasodilatation and endothelial function in obesity/insulin resistance: effects of troglitazone. Diabetologia 1998; 41: 569–76
Natali A, Baldeweg S, Toshi E, et al. Rosiglitazone directly improves endothelial function in type 2 diabetic patients [abstract]. Diabetes 2002; 51: A142
Minamikawa J, Tanaka S, Yamauchi M, et al. Potent inhibitory effect of troglitazone on carotid arterial wall thickness in type 2 diabetes. J Clin Endocrinol Metab 1998; 83: 1818–20
Koshiyama H, Shimono D, Kuwamura N, et al. Rapid communication: inhibitory effect of pioglitazone on carotid arterial wall thickness in type 2 diabetes. J Clin Endocrinol Metab 2001; 86: 3452–6
Asagami T, Abbasi F, Stuelinger M, et al. Metformin treatment lowers asymmetric dimethylarginine concentrations in patients with type 2 diabetes. Metabolism 2002; 51: 843–6
Preli RB, Klein KP, Herrington DM. Vascular effects of dietary L-arginine supplementation. Atherosclerosis 2002; 162: 1–15
Hirooka Y, Egashira K, Imaizumi T, et al. Effect of L-arginine on acetylcholine-induced endothelium-dependent vasodilation differs between the coronary and forearm vasculatures in humans. J Am Coll Cardiol 1994; 24: 948–55
Abbasi F, Asagmi T, Cooke JP, et al. Plasma concentrations of asymmetric dimethylarginine are increased in patients with type 2 diabetes mellitus. Am J Cardiol 2001; 88: 1201–3
Regensteiner JG, Popylisen S, Bauer TA, et al. Oral L-arginine and vitamins E and C improve endothelial function in women with type 2 diabetes. Vasc Me. 2003; 8: 169–75
Doshi SN, McDowell IF, Moat SJ, et al. Folie acid improves endothelial function in coronary artery disease via mechanisms largely independent of homocysteine lowering. Circulation 2002; 105: 22–6
Lee SJ, Lee DW, Kim KS, et al. Effect of estrogen on endothelial dysfunction in postmenopausal women with diabetes. Diabetes Res Clin Pract 2001; 54 Suppl. 2: S81-92
Perera M, Petrie JR, Hillier C, et al. Hormone replacement therapy can augment vascular relaxation in post-menopausal women with type 2 diabetes. Hum Reprod 2002; 17: 497–502
Guetta V, Quyyumi AA, Prasad A, et al. The role of nitric oxide in coronary vascular effects of estrogen in postmenopausal women. Circulation 1997; 96: 2795–801
American Diabetes Association. Aspirin therapy in diabetes. Diabetes Care 1997; 20: 1772–3
Moller DE. Potential role of TNF-alpha in the pathogenesis of insulin resistance and type 2 diabetes. Trends Endocrinol Metab 2000; 11: 212–7
Woodman RJ, Watts GF, Puddey IB, et al. Leukocyte count and vascular function in type 2 diabetic subjects with treated hypertension. Atherosclerosis 2002; 163: 175–81
Kharbanda RK, Walton B, Allen M, et al. Prevention of inflammation-induced endothelial dysfunction: a novel vasculo-protective action of aspirin. Circulation 2002; 105: 2600–4
Husain S, Andrews NP, Mulcahy D, et al. Aspirin improves endothelial dysfunction in atherosclerosis. Circulation 1998; 97: 716–20
Chenevard R, Hurlimann D, Bechir M, et al. Selective COX-2 inhibition improves endothelial function in coronary artery disease. Circulation 2003; 107: 405–9
Pan XR, Li GW, Hu YH, et al. Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance: the Da Qing IGT and Diabetes Study. Diabetes Care 1997; 20: 537–44
Tuomilehto J, Lindstrom J, Eriksson JG, et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. Finnish Diabetes Prevention Study Group. N Engl J Med 2001; 344: 1343–50
Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. Diabetes Prevention Program Research Group. N Engl J Med 2002; 346: 393–403
Watts GF, Herrmann S, Riches FM. Effects of diet and serotonergic agonist on hepatic apolipoprotein B-100 secretion and endothelial function in obese men. QJM 2000; 93: 153–61
Sciacqua A, Candigliota M. Weight loss in combination with physical activity improves endothelial dysfunction in human obesity. Diabetes Care 2003; 26: 1673–8
Prins JB. Adipose tissue as an endocrine organ. Best Pract Res Clin Endocrinol Metab 2002; 16: 639–51
Ziccardi P, Nappo F, Giugliano G, et al. Reduction of inflammatory cytokine concentrations and improvement of endothelial functions in obese women after weight loss over one year. Circulation 2002; 105: 804–9
Kumada M, Kihara S, Sumitsuji S, et al. Association of hypoadiponectinemia with coronary artery disease in men. Osaka CAD Study Group. Arterioscler Thromb Vase Biol 2003; 23: 85–9
Hotta K, Funahashi T, Arita Y, et al. Plasma concentrations of a novel, adipose-specific protein, adiponectin, in type 2 diabetic patients. Arterioscler Thromb Vasc Biol 2000; 20: 1595–9
Matsubara M, Maruoka S, Katayose S. Decreased plasma adiponectin concentrations in women with dyslipidemia. J Clin Endocrinol Metab 2002; 87: 2764–9
Ouchi N, Kihara S, Funahashi T, et al. Reciprocal association of C-reactive protein with adiponectin in blood stream and adipose tissue. Circulation 2003; 107: 671–4
Ouchi N, Kihara S, Arita Y, et al. Novel modulator for endothelial adhesion molecules: adipocyte-derived plasma protein adiponectin. Circulation 1999; 100: 2473–6
Ouchi N, Kihara S, Arita Y, et al. Adipocyte-derived plasma protein, adiponectin, suppresses lipid accumulation and class A scavenger receptor expression in human monocyte-derived macrophages. Circulation 2001; 103: 1057–63
Okamoto Y, Arita Y, Nishida M, et al. An adipocyte-derived plasma protein, adiponectin, adheres to injured vascular walls. Horm Metab Res 2000; 32: 47–50
Ouchi N, Ohishi M, Kihara S, et al. Association of hypoadiponectinemia with impaired vasoreactivity. Hypertension 2003; 42: 231–4
Kingwell BA, Berry KL, Cameron JD, et al. Arterial compliance increases after moderate-intensity cycling. Am J Physiol 1997; 273: H2186–91
American College of Sports Medicine and American Diabetes Association joint position statement. Diabetes mellitus and exercise. Med Sci Sport Exerc 1997; 29: i–vi
Foster GD, Wyatt HR, Hill JO, et al. A randomized trial of a low-carbohydrate diet for obesity. N Engl J Med 2003; 348: 2082–90
Samaha FF, Iqbal N, Seshadri P, et al. A low-carbohydrate as compared with a low-fat diet in severe obesity. N Engl J Med 2003; 348: 2074–81
Vogel RA, Corretti MC, Plotnick GD. The postprandial effect of components of the Mediterranean diet on endothelial function. J Am Coll Cardiol 2000; 36: 1455–60
Vogel RA. Alcohol, heart disease, and mortality: a review. Rev Cardiovasc Med 2002; 3: 7–13
Zilkens RR, Rich L, Burke V, et al. Effects of alcohol intake on endothelial function in men: a randomized controlled trial. J Hypertens 2003; 21: 97–103
Nestel PJ, Pomeroy S, Kay S, et al. Isoflavones from red clover improve systemic arterial compliance but not plasma lipids in menopausal women. J Clin Endocrinol Metab 1999; 84: 895–8
Nestel PJ, Yamashita T, Sasahara T, et al. Soy isoflavones improve systemic arterial compliance but not plasma lipids in menopausal and perimenopausal women. Arterioscler Thromb Vasc Biol 1997; 17: 3392–8
Vlassara H, Cai W, Crandall J, et al. Inflammatory mediators are induced by dietary glycotoxins, a major risk factor for diabetic angiopathy. Proc Natl Acad Sci U S A 2002; 99: 15596–601
Acknowledgements
Our work on the effects of pharmacotherapy and nutritional factors on vascular function in diabetes is supported by research grants from The National Health and Medical Research Council of Australia and from Fournier Pharma.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Woodman, R.J., Chew, G.T. & Watts, G.F. Mechanisms, Significance and Treatment of Vascular Dysfunction in Type 2 Diabetes Mellitus. Drugs 65, 31–74 (2005). https://doi.org/10.2165/00003495-200565010-00003
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200565010-00003