Summary
Abstract
Exemestane (Aromasin®) is an orally active steroidal irreversible inactivator of the aromatase enzyme indicated as an adjuvant treatment in postmenopausal women with estrogen receptor-positive early-stage breast cancer following 2–3 years of adjuvant treatment with tamoxifen, and for the treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen or other antiestrogen therapy.
Exemestane is effective for the treatment of postmenopausal women with early-stage or advanced breast cancer. In early-stage disease, switching to exemestane for 2–3 years after 2–3 years of adjuvant tamoxifen treatment was more effective in prolonging disease-free survival than continuing tamoxifen therapy, although it was not associated with an overall survival benefit, except in those with estrogen receptor-positive or unknown receptor status disease when nodal status, hormone replacement therapy (HRT) and chemotherapy use were adjusted for. Moreover, preliminary data suggest that the efficacy of exemestane is generally no different to that of tamoxifen in the primary adjuvant treatment of early-stage breast cancer, although exemestane may be better in prolonging the time to distant recurrence. In advanced disease, exemestane showed equivalent efficacy to megestrol in patients with disease refractory to tamoxifen and an efficacy not significantly different from that of fulvestrant in those refractory to a nonsteroidal aromatase inhibitor. Available data, some of which are limited, suggest exemestane is also effective in the first-line hormonal treatment of advanced breast cancer in postmenopausal women. Exemestane is generally well tolerated, although the potential bone fracture risk of the drug requires further investigation. Results from directly comparative trials indicating the efficacy, tolerability and bone fracture risk of exemestane relative to third-generation aromatase inhibitors and other agents in both early-stage and advanced disease, as well as the optimal sequence of endocrine therapies, are awaited with interest. In the meantime, switching to exemestane should be considered in postmenopausal women who have received 2–3 years of adjuvant tamoxifen treatment for early-stage breast cancer, and is an emerging treatment option for postmenopausal women with advanced breast cancer refractory to one or more antiestrogen therapies.
Pharmacological Properties
Exemestane acts by irreversibly binding to the aromatase active site, resulting in potent suppression of whole body aromatization and estrogen synthesis, without any marked or clinically relevant effect on levels of other steroid hormones. The drug may affect lipid levels and increase levels of homocysteine, but does not appear to have long-term effects on coagulation parameters. A switch from tamoxifen to exemestane may also reverse the endometrial thickening associated with tamoxifen. Exemestane is generally associated with a loss of bone mineral density and increases in serum or urinary levels of markers of bone turnover in postmenopausal women.
The absorption of exemestane is rapid after oral administration, with maximum plasma concentrations reached within 2 hours of a single 25 mg dose. The drug is 90% plasma protein bound and is extensively distributed into tissues. Exemestane undergoes extensive metabolism, primarily via cytochrome P450 3A4, as well as via aldoketoreductases, to metabolites that are inactive or have less activity than the parent drug. Exemestane is excreted mainly via urine and faeces and has a mean terminal elimination half-life of ≈24 hours.
Therapeutic Efficacy
Oral exemestane 25 mg/day for 2–3 years after 2–3 years of adjuvant tamoxifen was generally more effective than 5 years of continuous adjuvant tamoxifen in the treatment of postmenopausal women with early-stage estrogen receptor-positive/unknown receptor status breast cancer in a large well designed trial (IES). Switching from tamoxifen to exemestane was consistently associated with a significantly lower risk of experiencing one of the events (recurrence, contralateral breast cancer or death without recurrence) in the primary combined endpoint of disease-free survival than continued tamoxifen at median follow-ups of 30.6 and 55.7 months. The exemestane regimen was also more effective with regard to other endpoints, including the risk of contralateral breast cancer and breast cancer-free survival. There was generally no overall survival benefit in switching to exemestane, except in patients with estrogen receptor-positive or unknown receptor status disease at the median 55.7-month follow-up, when nodal status, HRT and chemotherapy use were adjusted for.
Moreover, preliminary data from the large, randomized, open-label, phase III, TEAM trial comparing exemestane with tamoxifen indicate that exemestane 25 mg/day is also effective in the primary adjuvant treatment of early-stage breast cancer in postmenopausal women. Disease-free survival (primary endpoint) did not significantly differ between exemestane and tamoxifen recipients. There was also no difference between the treatments in terms of relapse-free survival; however, exemestane was more effective in terms of the risk of distant metastases.
Exemestane 25 mg/day has also demonstrated efficacy in the treatment of postmenopausal women with advanced breast cancer refractory to one or more antiestrogen therapies in two large, well designed, phase III studies. In one of these studies, exemestane provided similar efficacy to megestrol in terms of objective response rate (primary endpoint) in postmenopausal women with advanced breast cancer refractory to tamoxifen. Although there was also no significant difference between the treatments in the median duration of objective response or the rate of overall success, exemestane recipients had a significantly longer median duration of overall success, time to disease progression and time to treatment failure than megestrol recipients. In the other trial, exemestane demonstrated efficacy not significantly different from that of intramuscular fulvestrant in patients with advanced breast cancer refractory to a nonsteroidal aromatase inhibitor, with no significant between-group difference in the time to disease progression (primary end-point) or other endpoints, including objective response rate, clinical benefit rate or overall survival.
Data, some of which are limited, from randomized, open-label, phase II or III studies comparing exemestane with anastrozole or tamoxifen, respectively, suggest that exemestane is an effective first-line hormonal treatment in postmenopausal women with advanced breast cancer. There were generally no differences between exemestane and these agents in terms of efficacy, although a significantly higher objective response rate was seen with exemestane than with tamoxifen in the largest phase III study.
Tolerability
Exemestane was generally well tolerated in postmenopausal women with early-stage or advanced breast cancer, with adverse events generally being mild to moderate in severity and of a similar nature irrespective of the stage of disease. Overall, hot flashes were the most common adverse event considered to be drug-related or of indeterminate cause experienced by exemestane recipients in active comparator-controlled trials.
In the large IES, at a median 55.7-month follow-up, patients with early-stage breast cancer switched to exemestane had significantly fewer serious gynaecological events, endometrial hyperplasia, uterine polyps/fibroids, venous thrombolytic events and muscle cramp than those who continued tamoxifen treatment. In contrast, exemestane was associated with a significantly higher incidence of musculoskeletal pain, arthralgia, diarrhoea, carpal tunnel syndrome, paraesthesia and joint stiffness than tamoxifen. There were no between-group differences in the incidences of the most common adverse events: menopausal symptoms (e.g. hot flashes), hypertension and fatigue. Moreover, a switch to exemestane was associated with a significantly lower incidence of abnormal endometrial thickening than continued tamoxifen. Preliminary data from the TEAM study after 2.75 years’ treatment indicated that the tolerability profile of exemestane versus tamoxifen in the primary adjuvant treatment of early-stage breast cancer was generally consistent with the findings of the IES. The incidence of fractures did not significantly differ between exemestane and tamoxifen recipients in the TEAM trial or in an on-treatment analysis of the IES, although it was significantly greater in exemestane than tamoxifen recipients in the IES when post-treatment adverse events were included.
Exemestane also had a generally similar tolerability profile to that of tamoxifen when used as a first-line hormonal treatment in postmenopausal women with advanced breast cancer, although it appeared to be more favourable than tamoxifen in terms of anaemia, thrombocytopenia and vaginal discharge, and less favourable in terms of weight gain, arthralgias, hypertension and diarrhoea (all between-group differences in incidence ≥5%). Postmenopausal women with advanced breast cancer who received exemestane were less likely to experience dyspnoea or moderate to severe gains in body-weight than those who received megestrol, although they were more likely to experience hot flashes, nausea and vomiting. The tolerability profile of exemestane was broadly similar to that of fulvestrant in postmenopausal women with advanced breast cancer.
Pharmacoeconomic Analyses
In modelled cost-utility analyses that incorporated data from the IES and direct medical costs, switching to exemestane after 2–3 years of tamoxifen was generally predicted to be cost effective with regard to the cost per quality-adjusted life-year (QALY) gained relative to continuing tamoxifen treatment in postmenopausal women with early-stage breast cancer in a number of countries over 10-year, 20-year and/or lifetime time horizons. Using data from trials that did not directly compare the regimens, analyses in Canada and Belgium suggested that switching from tamoxifen to exemestane was cost saving in terms of the cost per QALY gained relative to primary adjuvant anastrozole or extended adjuvant therapy with tamoxifen followed by letrozole in postmenopausal women with early-stage breast cancer over a time horizon of 20 years.
Similar content being viewed by others
1. Introduction
Worldwide, breast cancer is the leading cause of death from cancer among women, with more than 464 000 women estimated to have died from the disease in 2007.[1] Although the global incidence of breast cancer is increasing, mortality from the disease has stabilized or declined in some countries in recent years as a result of early detection and better treatments.[1]
Established risk factors for breast cancer include low parity, late age at first pregnancy, obesity and high alcohol consumption.[2] The mechanisms by which such risk factors relate to breast cancer are unclear at present, although may involve alterations in exposure to estrogens. In postmenopausal women, production of estrogens occurs primarily via the aromatization of androgens in the periphery in contrast to the predominantly ovarian production seen in premenopausal women.[3] Increased levels of estrogens have been associated with an increased risk of developing breast cancer in postmenopausal women.[2] Indeed, a large proportion of breast cancers in postmenopausal women express estrogen receptors and are stimulated by estrogens to proliferate.[4] Consequently, estrogen deprivation has proven to be effective in the treatment of breast cancer.[4,5]
Aromatase inactivators/inhibitors are now an established alternative treatment option to tamoxifen for use in postmenopausal women, reducing estrogen production by inhibiting the activity of aromatase, a key enzyme involved in the synthesis of the majority of non-ovarian estrogen.[4,6]
Exemestane (Aromasin®) is a third-generation steroidal aromatase inactivator (often referred to as an aromatase inhibitor) approved in the UK,[7] US[8] and several other countries worldwide as an adjuvant treatment in postmenopausal women with estrogen receptor-positive early-stage breast cancer following 2–3 years of adjuvant tamoxifen treatment, as well as for the treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen or other antiestrogen therapy. This article focuses on the clinical efficacy and tolerability data relevant to the use of oral exemestane in the treatment of breast cancer and discusses the key pharmacological properties of the drug.
2. Pharmacodynamic Profile
The pharmacodynamic properties of exemestane have been reviewed in detail previously.[9,10] Therefore, this section provides an overview of key pharmacodynamic data, focusing on more recent data related to the approved dosage of exemestane 25 mg/day (section 7). Further data from some clinical trials[11–13] are discussed in section 4. Some data are available only as abstracts[14–19] and posters.[14,19]
Exemestane is an irreversible steroidal inactivator of the aromatase enzyme.[7,8] Being structurally related to the aromatase substrate androstenedione,[7,8] exemestane competes with the androgen for binding to the aromatase active site.[8] Upon catalysis, an exemestane intermediate is produced that binds irreversibly to the aromatase active site, thus inactivating the enzyme.[8] In vitro data suggest that exemestane, unlike the aromatase inhibitors anastrozole and letrozole, may destabilize the aromatase protein, leading to its degradation.[20]
Exemestane 25 mg/day for 6–8 weeks almost completely suppressed (≈98%) whole body aromatization in postmenopausal women with advanced breast cancer.[28] Consistent with this finding, exemestane is associated with potent suppression of estrogen levels (table I),[21,27,28] with levels of circulating estrogens maximally suppressed 2–3 days after a single exemestane 25-mg dose.[8,31]
Overview of the general pharmacodynamic properties of exemestane (EXE). Results in postmenopausal women with early-stage[19,21–23] (operable[24] or resected[21–23,25]), invasive[18] or advanced[13,26,27] (locally advanced[28,29] or metastatic[28–30]) breast cancer, healthy postmenopausal women[31,32] (n = 10–340 evaluable) and animal models.[10] The effects of EXE on bone are discussed in table II. Data in postmenopausal women relate to EXE 25 mg/day, unless otherwise noted; the EXE dosage was not specified in one study[18]
In general, exemestane appears to be selective in the suppression of aromatase, generally having no marked affect on plasma levels of steroid hormones other than estrogen (table I).[21,27,29,31] However, studies have produced mixed results with regard to the effect of exemestane 25 mg on levels of sex hormone binding globulin (SHBG) [table I],[27,29] the synthesis of which is stimulated by estrogens and suppressed by androgens. Nevertheless, dose-dependent suppression of plasma SHBG levels was seen with exemestane 5–200 mg/day, possibly due to intrinsic androgenic activity of the exemestane metabolite 17-hydro-exemestane.[29]
Antitumour effects of the drug were initially demonstrated in rat models of postmenopausal breast cancer (table I).[10]
Data concerning the effect of exemestane on lipid levels are variable (table I). Exemestane was generally not associated with any detrimental effects on lipid parameters compared with placebo in postmenopausal women with early-stage breast cancer during 24 months of treatment.[21] Although high-density lipoprotein (HDL) cholesterol and apolipoprotein A1 levels were significantly reduced relative to placebo during exemestane therapy,[21] levels returned to near those at baseline 3 months after drug discontinuation.[33]
The effects of exemestane on several lipid parameters were generally not markedly different from those of tamoxifen,[23,25] anastrozole[32] or letrozole[32] in most studies. However, exemestane had less favourable effects on certain lipids, most notably low-density lipoprotein (LDL) cholesterol, than tamoxifen in some instances in postmenopausal women with breast cancer who switched to exemestane after receiving adjuvant tamoxifen[22,25] or who received exemestane in a primary adjuvant setting[23] (table I). By contrast, exemestane had beneficial effects on triglyceride levels versus tamoxifen in two of these studies[22,23] (table I). In healthy postmenopausal women, exemestane significantly reduced HDL cholesterol levels and increased both LDL : HDL and apolipoprotein B : apolipoprotein A–I ratios relative to anastrozole or letrozole at 24 weeks (table I).[32]
Exemestane recipients had beneficial changes in body fat mass[22,25] and, although the drug had variable effects on coagulation parameters in a short-term study,[18] it did not appear to have long-term effects on such parameters[21] (table I). However, relative to placebo, long-term treatment with exemestane was associated with significant, albeit modest, elevations in plasma levels of total homocysteine,[21] a potential marker of increased vascular disease risk[34,35] (table I), although the between-group difference was not significant 6–12 months after drug discontinuation.[33] Shorter term, exemestane for 5 months had no marked affect on plasma levels of total homocysteine (table I).[26]
With regard to endometrial effects, a switch from tamoxifen to exemestane reduced endometrial thickness and uterine volume relative to continued tamoxifen in postmenopausal women with early-stage breast cancer, according to data from a substudy (n = 183)[19] of the IES (Intergroup Exemestane Study)[11,12] in patients with no prior uterine malignancy, such as endometrial cancer, or undiagnosed vaginal bleeding in the year prior to the trial (table I). The incidence of abnormal endometrial thickness are discussed in section 5.1.1.[19]
Exemestane, as with other aromatase inhibitors, is generally associated with a loss of bone mineral density (BMD).[8] Indeed, the drug significantly increased the mean annual rate of BMD loss at some sites compared with placebo after 2 years in postmenopausal women with early-stage breast cancer (table II),[21] although between-group differences were not significant 1 year after drug discontinuation.[33] A switch to exemestane after 2–3 years of tamoxifen was also associated with significant reductions from baseline in BMD at several sites in postmenopausal women with early-stage or resected breast cancer (table II).[36,37] For example, in a substudy (n = 206)[36] of the IES,[11,12] lumbar spine and total hip BMD were significantly reduced between 12 and 24 months after switching to exemestane, although reductions from baseline predominantly occurred in the first 6 months, perhaps partly due to a loss of the bone protective effects of tamoxifen. Switching to exemestane significantly reduced lumbar spine and total hip BMD relative to continuing tamoxifen therapy,[36] with the changes in BMD at these sites significantly (p < 0.005) differing by 2.8% and 1.4% between treatments at the end of the trial.[17] However, there were no significant between-group differences in BMD 2 years after treatment cessation.[17]
Effects of exemestane (EXE) 25 mg/day on bone in postmenopausal women. Summary of results in studies in postmenopausal women with early-stage,[14,15,21,33,36,38–40] resected[21,33,36,37] or invasive[16] breast cancer,[14–16,21,33,36–40] and in healthy postmenopausal women[32] (n = 51–206 evaluable); the EXE dosage was not reported in one study[16]
The effect of exemestane on BMD has also been compared with that of tamoxifen[14,15,38,40] or anastrozole[14] in a primary adjuvant setting in postmenopausal women with early-stage breast cancer in substudies (n = 51–167 evaluable) of larger clinical trials (some preliminary data available only as abstracts and/or posters[14,15]) [table II]. Where statistical analyses were reported, exemestane generally reduced spine and hip BMD in comparison with tamoxifen after 6–24 months of treatment,[38,40] although no significant difference in BMD was evident among exemestane, tamoxifen or anastrozole recipients after 12 months’ therapy in one study.[14]
Consistent with its effects on BMD, exemestane generally increased serum or urinary levels of bone resorption and bone formation markers relative to baseline, placebo or tamoxifen in postmenopausal women with early-stage,[21,33,36,39] invasive[16] or resected[21,33,36,37] breast cancer and in healthy postmenopausal women,[32] although its effects did not significantly differ from those seen with anastrozole or letrozole[16,32] (table II). Data concerning the incidence of fractures and osteoporosis during exemestane treatment are discussed in section 5.2.
3. Pharmacokinetic Profile
This section provides an overview of the pharmacokinetic profile of exemestane, which has been reviewed previously.[9,10] Pharmacokinetic data for exemestane in postmenopausal women with breast cancer are limited, thus data in this section are mostly from studies in healthy postmenopausal volunteers.[31,41–43] Data from studies evaluating the effect of moderate or severe hepatic (Child-Pugh grade B or C) or renal (creatinine clearance [CLCR] of 30–60 mL/min/1.73 m2 [1.8–3.6 L/h/1.73 m2] or 10–29 mL/min/1.73 m2 [0.6–1.7 L/h/1.73 m2]) impairment on the pharmacokinetics of a single exemestane 25-mg dose are also discussed.[44] Some data are available only as abstracts,[41–43] with additional data obtained from the manufacturer’s prescribing information.[7,8]
The absorption of exemestane is rapid after oral administration,[31] with ≥42% of the dose being absorbed from the gastrointestinal tract.[8] The average maximum plasma concentration (Cmax) of exemestane (17 ng/mL) was reached 2 hours after a single oral 25-mg dose in healthy postmenopausal volunteers.[41] Absorption of exemestane occurred more than twice as rapidly in postmenopausal women with advanced breast cancer than in healthy postmenopausal women (mean time to Cmax 1.2 vs 2.9 hours), with mean area under the plasma concentration-time curve (AUC) values also 82% higher in the women with breast cancer (75.4 vs 41.4 ng ⋅ h/mL).[8]
Plasma levels of the drug were generally similar following administration of single or repeated doses of 25 mg in healthy postmenopausal women,[41] with no unexpected accumulation after repeated administration.[7] Oral exemestane displays dose-proportional pharmacokinetics after administration of single and repeated doses over the range of 10–200 mg and 0.5–50 mg, respectively.[8]
Systemic exposure to exemestane is greater in the fed than the fasted state.[42] The AUC was significantly higher in the presence of food than under fasting conditions after administration of a single dose of exemestane 25 mg in healthy postmenopausal volunteers (41.3 vs 29.7 ng ⋅ h/mL; p < 0.05); corresponding Cmax values were 17.7 versus 11.1 ng/mL.[42] Exemestane should, therefore, be taken after a meal (section 7).[7,8]
Exemestane is 90% plasma protein bound independent of concentration,[7,8] with both α1-acid glycoprotein and albumin contributing to binding.[8] The drug is extensively distributed into tissues,[8] and has a volume of distribution of ≈20 000 L (not corrected for oral bioavailability).[7]
Exemestane undergoes extensive metabolism, with <10% of the dose circulating in plasma unchanged.[8] Metabolism of the drug occurs via oxidation (primarily by the cytochrome P450 [CYP] enzyme CYP3A4) and reduction (by aldoketoreductases), producing metabolites that are inactive or less active than the parent drug.[7,8] The major metabolite, 17-hydro-exemestane, has been shown to reach plasma levels <10% of those of the parent drug.[31]
Elimination of exemestane was mainly via the faeces (42%) and urine (42%) after administration of a radiolabelled dose in healthy postmenopausal volunteers, with <1% of the administered dose excreted via the urine unchanged.[8] The manufacturer’s prescribing information reports a mean terminal half-life for exemestane of ≈24 hours.[7,8] The mean oral plasma clearance of the drug was 517 L/h after a single 25-mg dose and 715 L/h after repeated doses in healthy postmenopausal women.[41] Notably, average exemestane oral clearance was 45% lower in postmenopausal women with advanced breast cancer than in healthy postmenopausal volunteers.[8]
Postmenopausal women with moderate or severe hepatic impairment had a 2- to 3-fold increase in AUC from time zero to infinity (AUC∞) and a 2-fold increase in Cmax relative to women with normal hepatic function.[44] A 2- to 3-fold increase in exemestane AUC∞ was also evident in postmenopausal women with moderate or severe renal impairment relative to women with normal renal function, with a positive correlation demonstrated between CLCR and the apparent oral clearance of the drug.[44] However, exemestane dosage adjustments are not considered necessary in these patient populations (section 7),[7,8] although caution is advised in the UK.[7] The pharmacokinetics of exemestane do not appear to be affected by age.[8]
Coadministration of exemestane and potent CYP3A4 inducers, such as rifampicin (rifampin), may reduce exemestane exposure.[8] In healthy postmenopausal volunteers, mean exemestane Cmax and AUC∞ values were reduced by 41% and 54% after coadministration of exemestane 25 mg and rifampicin 600 mg/day.[8] An increased exemestane dosage of 50 mg/day is recommended in patients receiving potent CYP3A4 inducing agents in the US (section 7).[8]
The clearance of exemestane is not anticipated to be significantly affected by concomitant administration of CYP inhibitors. The pharmacokinetic profile of exemestane 10 mg was not affected when coadministered with the CYP3A4 inhibitor ketoconazole 200 mg/day,[43] although no additional drug interaction studies have been performed.[8] Exemestane does not inhibit the activity of key CYP enzymes, such as CYP1A2, CYP2C9, CYP2D6, CYP2E1 or CYP3A4.[8]
4. Therapeutic Efficacy
The efficacy of exemestane in the treatment of breast cancer in postmenopausal women has been evaluated in numerous clinical studies. This section focuses on data from several randomized, phase II[45] or III[12,13,46–49] trials that compared the efficacy of exemestane with that of a number of active comparators in postmenopausal women with early-stage (section 4.1)[12,46] or advanced (section 4.2)[13,45,47–49] breast cancer. Exemestane was administered orally at a dosage of 25 mg/day in all studies, and patient characteristics at baseline were generally similar between treatment groups. Efficacy assessments were performed in the intent-to-treat (ITT)[12,13,47,48] or modified ITT (exclusive of nine patients who withdrew consent)[46] population, where specified.
4.1 Early-Stage Breast Cancer
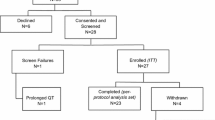
The clinical efficacy of switching to oral exemestane for 2–3 years after receiving 2–3 years of tamoxifen (to complete a total of 5 years of adjuvant therapy) has been compared with that of 5 years of continuous tamoxifen therapy in postmenopausal women with early-stage breast cancer in the large, randomized, double-blind, multicentre, phase III IES (section 4.1.1; see table III for dosage and design details).[12] The efficacy of exemestane has also been compared with that of tamoxifen in the primary adjuvant treatment of early-stage breast cancer in postmenopausal women in the randomized, open-label, multinational, phase III, TEAM (Tamoxifen Exemestane Adjuvant Multinational) trial (section 4.1.2; see figure 1 for dosage and design details).[46]
Efficacy of exemestane (EXE) in postmenopausal women with early-stage breast cancer. Summary of results at median follow-up of 30.6[12] and 55.7[11] mo in patients (pts) who were switched to EXE 25 mg once daily for 2–3 years after 2–3 years of tamoxifen (TAM) 20 mg once daily or continued treatment with TAMa in the randomized, double-blind, multicentre, phase III, IES (total of 5 years of adjuvant endocrine therapy)
Comparative efficacy of exemestane (EXE) versus tamoxifen (TAM) as primary adjuvant treatment for early-stage breast cancer in postmenopausal women. Incidence of combined and individual endpoint events at 2.75 years in the modified intent-to-treat population in the randomized, open-label, multinational, phase III, TEAM study[46] in which patients (pts) received EXE 25 mg/day (n = 4898) or TAM 20 mg/day (n = 4868). The hazard ratio (HR) for the primary endpoint of disease-free survival (based on the combined endpoint events) was 0.89 (95% CI 0.77, 1.03). HRs for individual endpoint events (other than distant metastases; HR 0.81 [95% CI 0.67, 0.98; p < 0.03]) were not reported.
Other studies include the National Surgical Adjuvant Breast and Bowel Project B-33 study,[50] a randomized, double-blind trial that compared the efficacy of 5 years of exemestane with that of placebo following 5 years of adjuvant tamoxifen. However, as the study was statistically underpowered for the primary endpoint analysis because of early termination of patient accrual (in light of efficacy data from another trial), it is not discussed further within this review.
As the preliminary findings of IES have been reviewed previously,[9] this section reviews data from the IES at median follow-up periods of 30.6[12] and 55.7[11] months that are now fully published. Data from the first planned analysis of the TEAM trial at 2.75 years are available as a conference presentation.[46]
Postmenopausal women eligible for inclusion in these studies were aged ≥55 years[12] with histologically[12,46] and/or cytologically[46] confirmed, resected,[12,46] invasive[12,46] breast cancer (unilateral where specified[12]) that was estrogen receptor-[12,46] and/or progesterone receptor-positive[46] or of unknown receptor status.[12]
The primary efficacy variable in both the IES[12] and TEAM study[46] was disease-free survival, which was based on analysis of the combined occurrence of the following events: local recurrence,[12,46] distant recurrence[12] or metastases,[46] primary contralateral breast cancer,[12] new primary breast cancer without metastasis[46] and intercurrent death[12,46] (without recurrence[12]). Among other endpoints were individual events,[46] overall survival,[12] relapse-free survival[46] and breast cancer-free survival (subsidiary endpoint in one study[12]).
Both the IES[51,52] and TEAM study[53] evaluated health-related quality of life (HR-QOL) and the burden of endocrine symptoms in a subgroup of patients (n = 582[51,52] and 543[53]), using a variety of disease-specific questionnaires. Data are available in a separate publication[51] or as an abstract[52,53] and/or a poster.[52]
4.1.1 Adjuvant Treatment After 2–3 Years of Tamoxifen Treatment
Only patients who had received ≥2 years and ≤3 years and 1 month of adjuvant tamoxifen were eligible to participate in the IES.[12] The trial excluded patients who had an estrogen receptor-negative breast tumour, evidence of a distant metastasis or local relapse since diagnosis, a cardiac, skeletal or endocrine disorder of clinical significance or had received hormone replacement therapy (HRT) <4 weeks prior to randomization. At randomization, participants had been treated with tamoxifen for a median of 2.4 years.[12]
In addition to ITT analyses, outcomes for patients whose breast cancer was estrogen receptor-positive (≈86% of patients) or of unknown receptor status (≈12%) were analyzed at the 55.7-month follow-up;[11] this population excluded patients whose estrogen receptor status was unknown at randomization but subsequently identified as being estrogen receptor-negative.
At median follow-ups of 30.6 and 55.7 months, patients with early-stage breast cancer who switched to exemestane consistently had a significantly lower risk of experiencing one of the events in the combined endpoint of recurrence, contralateral breast cancer or death without recurrence than patients who continued treatment with tamoxifen, as indicated by the hazard ratio (HR) for disease-free survival (primary endpoint) [table III].[11,12] This finding was observed in the ITT population[11,12] and in patients whose breast cancer was estrogen receptor-positive or of unknown receptor status.[11] The incidence of disease-free survival 3 years after randomization was 91.5% (95% CI 90.0, 92.7) in recipients of exemestane compared with 86.8% (95% CI 85.1, 88.3) in patients who continued tamoxifen, yielding an absolute difference of 4.7% in the ITT population.[12]
Subgroup analyses for disease-free survival demonstrated that the HRs for this endpoint were not markedly affected by adjusting for prognostic factors, such as estrogen receptor status, nodal status and use of HRT or chemotherapy.[11,12] Moreover, the HRs were generally consistent across subgroups defined by these factors.
A switch to exemestane was also more effective than continued tamoxifen with regard to the risk of contralateral breast cancer in both the ITT population[11,12] and in patients with estrogen receptor-positive or unknown receptor status disease[11] (table III).
There was generally no overall survival benefit in switching to exemestane compared with continuing treatment with tamoxifen (table III).[11,12] However, at the median follow-up of 55.7 months,[11] the risk of death was significantly lower with exemestane than with tamoxifen in patients whose breast cancer was estrogen receptor-positive or of unknown receptor status when adjusted for nodal status and HRT and chemotherapy use (HR 0.83 [95% CI 0.69, 0.99]; p = 0.04). Moreover, exemestane recipients had a significantly lower risk of experiencing death from breast cancer than patients who continued to receive tamoxifen, both in the ITT population and among patients whose disease was estrogen receptor-positive or of unknown status, according to the subsidiary endpoint of breast cancer-free survival (table III).[11,12]
The HR-QOL of patients switched to exemestane generally did not significantly differ from that of patients who continued treatment with tamoxifen, according to data reported in a substudy[51] of the IES.[12] There were generally no significant between-group differences in the primary HR-QOL endpoint of mean change from baseline in FACT-B (Functional Assessment of Cancer Therapy — Breast) Trial Outcome Index score from 3–60 months post-randomization,[51,52] except at the 6-month timepoint (−2.10 with exemestane vs −0.01 with tamoxifen; p = 0.009), although this difference was not considered to be clinically relevant.[51]
The burden of endocrine symptoms was lower at 9, 12, 18, 24[51] and 60[52] months than at baseline in both exemestane and tamoxifen recipients, as indicated by significant (p ≤ 0.01) mean increases in endocrine subscale (ES) scores; no significant between-group differences were evident at any timepoint. In addition, exemestane and tamoxifen recipients did not significantly differ in terms of overall HR-QOL, as measured by combined FACT-B and ES scores, at any timepoint from 3–24 months, despite a significant (p = 0.007) mean reduction from baseline of 3.12 at 6 months in exemestane recipients that was not considered to be clinically relevant.[51]
4.1.2 Primary Adjuvant Treatment
In the TEAM trial,[46] eligible patients were required to receive adjuvant hormonal therapy within 10 weeks of undergoing surgery and/or receiving chemotherapy; exclusion criteria were not reported.
As a primary adjuvant treatment in postmenopausal women with early-stage breast cancer, the efficacy of exemestane was not significantly different to that of tamoxifen in terms of the risk of experiencing one of the events (i.e. local recurrence, distant metastases, new primary breast cancer without metastases or intercurrent death) in the primary combined endpoint of disease-free survival at a follow-up of 2.75 years (HR 0.89 [95% CI 0.77, 1.03]) [figure 1].[46] However, data from an analysis that excluded 96 patients who did not receive either treatment indicated that exemestane (n = 4853) was associated with a significantly (p = 0.02) lower risk of experiencing disease-free survival events than tamoxifen (n = 4817) at this timepoint (HR 0.83; 95% CI 0.71, 0.97).
Moreover, exemestane was more effective than tamoxifen with regard to the risk of distant metastases (HR 0.81 [95% CI 0.67, 0.98]; p < 0.03) [figure 1], although there was no significant between-group difference in terms of relapse-free survival (HR 0.85 [95% CI 0.72, 1.00]).[46]
The HR-QOL of postmenopausal women with early-stage breast cancer who received exemestane therapy generally did not differ from that of those who received tamoxifen, as measured using several questionnaires, including the FACT-ES, European Organisation for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire (QLQ)-C30, and the EORTC-Breast Cancer-Specific QLQ-BR23.[53] After 1 and 2 years of therapy, there were no significant between-group differences with regard to physical, role, cognitive or emotional functioning, breast, arm or endocrine symptoms, or body image. However, exemestane was associated with significantly (p ≤ 0.005) more insomnia and less sexual enjoyment than tamoxifen after 1 year of treatment, and significantly (p < 0.05) more insomnia and worse sexual functioning after 2 years of treatment, although there was no significant between-group difference in sexual enjoyment at the 2-year timepoint.
4.2 Advanced Breast Cancer
4.2.1 Disease Refractory to One or More Antiestrogen Therapies
The results of several noncomparative[54–59] or crossover[45] phase II trials and an exploratory trial[60] (n = 60–241) have demonstrated the clinical efficacy of oral exemestane 25 mg once daily in postmenopausal women with advanced breast cancer[45,54–60] (locally advanced,[54] metastatic[54,57–60] or inoperable,[60] where specified) refractory to tamoxifen,[58] tamoxifen and megestrol[57] or aromatase inhibitors as the most recent hormonal therapy.[45,54–56,59,60] Consequently, the efficacy of exemestane in postmenopausal women with advanced breast cancer refractory to tamoxifen[13] or nonsteroidal aromatase inhibitors[48] has been compared with that of megestrol[13] and fulvestrant[48] in two large, randomized, double-blind, placebo-controlled, multicentre, phase III studies (n = 769[13] and 693[48]) [see table IV for dosage and design details]. Some of these studies[13,55,57–59] have been reviewed previously in Drugs,[10] with discussion in this section focused on the findings of the larger phase III trials.[13,48] Some data are available from the manufacturer’s prescribing information.[8]
Efficacy of exemestane (EXE) in postmenopausal women with advanced breast cancer. Summary of two randomized, double-blind, multicentre, phase III studies of oral EXE versus oral megestrol (MEG)[13] or intramuscular fulvestrant (FUL)[48] in patients (pts) with advanced breast cancer whose disease had progressed or recurred during tamoxifen[13] or nonsteroidal aromatase inhibitor[48] therapy. Values for times are medians; intent-to-treat analyses are presented
In addition, the efficacy of exemestane has been compared with that of anastrozole in postmenopausal women with tamoxifen-refractory advanced breast cancer in a randomized, open-label, multicentre study.[61] However, as the study was prematurely closed because of accrual delays and, therefore, not powered to demonstrate differences between treatment groups, data from the study are not discussed further.
Postmenopausal women included in the phase III trials had advanced breast cancer[13,48] (locally advanced or metastatic, where specified[48]) that had progressed[13,48] or relapsed[13] during tamoxifen[13] or nonsteroidal aromatase inhibitor[48] treatment. Also included in one of the studies[48] were postmenopausal women with locally advanced or metastatic breast cancer who had experienced a disease relapse during, or within 6 months of discontinuing, adjuvant nonsteroidal aromatase inhibitor therapy (≈12% of patients). Patients had to have estrogen and/or progesterone receptor-positive disease[13,48] or disease of unknown receptor status with a previous response to tamoxifen.[13] Other inclusion criteria included a WHO[48] or Eastern Cooperative Oncology Group (ECOG)[13] performance status of 0–2, one or more measurable or evaluable lesions[13,48] and a life expectancy of ≥3 months.[48]
Patients previously treated with up to one chemotherapy regimen for advanced disease were eligible for participation in the trials.[13,48] However, those previously treated with strontium89,[13] high-dose chemotherapy,[13] hormonal agents other than tamoxifen,[13] or extensive cytotoxic or radiation therapy within the 4 weeks prior to the study[48] were excluded. Also excluded were patients with brain metastases,[13,48] leptomeningeal disease[13] or metastases,[48] or massive[13] or life-threatening metastatic[48] visceral disease.
Primary endpoints were objective response rate (defined as the proportion of patients who achieved a complete or partial response, based on modified WHO criteria)[13] and time to disease progression.[48] A number of secondary endpoints related to clinical efficacy (table IV),[13,48] pain,[13] tumour-related signs and symptoms,[13] and HR-QOL[13] were also assessed.
Where reported,[13] the median duration of treatment was ≈17 weeks. The median duration of follow-up was 48.9 weeks[13] and ≈13 months,[48] with an updated analysis of one study[48] at a median follow-up of 20.9 months available as an abstract.[62]
Versus Megestrol
Exemestane provided similar efficacy to megestrol in postmenopausal women with advanced breast cancer refractory to tamoxifen, as determined by prespecified equivalence criteria for the objective response rate (primary endpoint) [table IV].[13] However, although there was also no significant difference between the treatments in the median duration of objective response or the rate of overall success, exemestane recipients had a significantly longer median duration of overall success, time to disease progression and time to treatment failure than megestrol recipients (table IV). In addition, exemestane appeared to be associated with a statistical survival advantage over megestrol (table IV),[13] although there were too few patient deaths during the study to firmly establish between-group differences in overall survival.[8]
At two or more consecutive assessments, 24.7% of exemestane versus 24.2% of megestrol recipients reported an improvement from baseline of >20% in overall pain score (pain severity graded on a 5-point scale from 0 [none] to 5 [intolerable]). Moreover, improvements in overall pain scores were reported in 51.4% and 46.2% of exemestane and megestrol recipients who experienced complete or partial responses.[13] The proportion of patients reporting an improvement in tumour-related signs and symptoms other than pain (graded via the National Cancer Institute Common Toxicity Criteria [CTC]) did not significantly differ between exemestane (12.1%) and megestrol (7.5%) recipients.[13]
Several aspects of HR-QOL were different between exemestane and megestrol recipients.[13] Exemestane significantly (p < 0.01) improved certain scores on the EORTC QLQ-C30 relative to megestrol, including global health, both physical and role functioning, fatigue, dyspnoea and constipation. However, between-group differences were significant in favour of megestrol for improvements in emotional functioning, pain and appetite loss scores (quantitative data and some statistical analyses not reported).
Versus Fulvestrant
Oral exemestane demonstrated clinical efficacy not significantly different from that of intramuscular fulvestrant in postmenopausal women with advanced breast cancer whose disease progressed or recurred during treatment with a nonsteroidal aromatase inhibitor (table IV).[48] There was no significant between-group difference in the median time to disease progression (primary endpoint), the objective response rate or the clinical benefit (i.e. overall success) rate at a median follow-up of ≈13 months (table IV). Moreover, the exemestane and fulvestrant groups did not significantly differ in terms of overall survival in an updated (median 20.9-month follow-up)[62] analysis (available as an abstract) [table IV].
4.2.2 First-Line Treatment
The efficacy of exemestane in the first-line hormonal treatment of postmenopausal women with advanced breast cancer (i.e. metastatic or locally recurrent inoperable disease) was initially demonstrated in a randomized, open-label, multicentre, phase II study,[63] in which patients received exemestane 25 mg (n = 61) or tamoxifen 20 mg (n = 59) orally once daily. However, as the study was not powered to detect differences between treatment groups, data from the study are not discussed further. Subsequently, two randomized, open-label, multicentre, phase III studies[47,49] were conducted to compare the efficacy of exemestane and tamoxifen in this setting. Results of a randomized, open-label, multicentre, phase II, crossover study comparing the efficacy of exemestane with that of anastrozole in the first-line hormonal treatment of advanced breast cancer in postmenopausal women are also discussed.[45] One study is fully published,[47] whilst other data are available only as abstracts;[45,49] additional data for one study[45] are available from other sources.[64]
Eligible patients had metastatic[47,49,64] or locally advanced[64] or recurrent[47,49] inoperable breast cancer that was hormone receptor-positive[47,49] (estrogen receptor and/or progesterone receptor-positive, where specified[64]) or of unknown receptor status with a disease-free interval of ≥2 years.[47] Other eligibility criteria included measurable disease,[64] one or more measurable lesions[47,49] and an ECOG performance status of 0–2.[64] Where stated, patients who had received prior hormone therapy for metastatic disease were excluded;[47,64] however, patients previously treated with adjuvant tamoxifen with a recurrence-free interval of ≥6 months[47] or with disease progression ≥2 years since such adjuvant treatment[64] were eligible, as were patients previously treated with one chemotherapy regimen for advanced[64] or metastatic[47] disease.
Patients were randomized to receive oral exemestane 25 mg/day,[45,47,49] tamoxifen 20 mg/day[47,49] or anastrozole 1 mg/day[45] until disease progression[45,47,49] or unacceptable toxicity.[47,49] Where stated,[47] the median follow-up was 29 months, with overall survival data available from an updated 49-month analysis.
Primary efficacy measures included progression-free survival[47] and objective response rate.[49,64] A number of secondary endpoints were also reported. In one study,[47] the ITT analysis included all randomized patients with the exception of 11 patients from two sites that were excluded because of inadequate documentation.
Versus Tamoxifen
In the largest phase III trial, the efficacy of exemestane (n = 182) did not significantly differ from that of tamoxifen (n = 189) as first-line hormonal treatment in postmenopausal women with advanced breast cancer, with a median progression-free survival of 9.9 and 5.8 months (primary endpoint) after a median follow-up of 29 months.[47] However, exemestane was associated with a significantly higher objective response rate than tamoxifen at this timepoint (46% vs 31% of patients; odds ratio [OR] 1.85 [95% CI 1.21, 2.82]; p = 0.005), although there was no significant between-group difference with regard to overall survival at 49 months’ follow-up (45% vs 43% of patients had died; HR 1.13; 95% CI 0.85, 1.50).
These data are supported by a smaller study in 167 postmenopausal women with advanced breast cancer.[49] Objective response rates in the exemestane (n = 83) and tamoxifen (n = 84) groups were 37.4% and 29.8% (primary endpoint), median durations of objective response were 59 and 39 weeks, clinical benefit rates were 79.5% and 78.6% and median times to disease progression were 52 and 36 weeks (no statistical analyses reported).
Versus Anastrozole
Exemestane demonstrated efficacy not significantly different from that of anastrozole in the first-line hormonal treatment of 103 postmenopausal women with advanced breast cancer.[45] There were no significant differences between exemestane and anastrozole recipients in the objective response rate (primary endpoint) [37% vs 52% of patients], clinical benefit rate (47% vs 60%) or mean time to disease progression (8.1 vs 12.1 months).
5. Tolerability
The tolerability profile of exemestane in postmenopausal women with early-stage[9] or advanced[10] breast cancer has been previously reviewed. This section provides key tolerability data related to administration of exemestane 25 mg once daily in patients with early-stage[11,46] or advanced[13,47,48] breast cancer available from large phase III clinical studies discussed in sections 4.1 and 4.2, with additional data obtained from a substudy (n = 1614)[65] of the TEAM trial[46] and substudies (n = 183[19] and 206[36]) of the IES.[11] Given that provisional data from the IES at a median 30.6-month follow-up comparing a switch to exemestane versus continued tamoxifen treatment in patients with early-stage breast cancer[12] have been discussed in detail previously,[9] this section reviews the longer-term tolerability data from a median 55.7-month follow-up,[11] focusing mainly on the on-treatment adverse events (i.e. those occurring between randomization and 30 days after treatment discontinuation). Adverse events were graded according to the National Cancer Institute CTC, where specified.[11] Some data are available only as a conference presentation[46] or as an abstract plus poster.[19]
5.1 General Tolerability
Exemestane 25 mg once daily was generally well tolerated in postmenopausal women with early-stage[11,46] or advanced[13,47,48] breast cancer, with adverse events being mostly of mild or moderate severity, where specified.[11,47] The nature of adverse events associated with exemestane treatment was generally similar irrespective of whether the drug was being administered for early-stage or advanced disease. Hot flashes were the most common adverse event considered to be treatment-related[13,48] or of indeterminate cause[13] in exemestane recipients with advanced breast cancer (occurred in 12.6%[13] and 11.5%[48] of patients), with menopausal symptoms (46.3% of patients[11]), such as hot flashes (41.3%[11] and 28.5%[46]), the most commonly reported treatment-emergent adverse events associated with exemestane in patients with early-stage breast cancer.
5.1.1 Versus Tamoxifen
The vast majority of postmenopausal women with early-stage breast cancer who switched to exemestane or continued tamoxifen in the IES experienced a treatment-emergent adverse event of any grade (93% in both groups).[11] Serious treatment-emergent adverse events (grade 3 or 4 in severity) occurred in 18.4% of patients in the exemestane group and 17.6% of patients in the tamoxifen group. The most common treatment-emergent adverse events (i.e. incidence ≥10% in either group) are summarized in figure 2, with menopausal symptoms, such as hot flashes, and hypertension being the most frequent. Notably, there was a significantly (p < 0.01) lower incidence of serious gynaecological events (5.9% vs 9.0% of patients), such as vaginal bleeding (4.6% vs 6.5%) and uterine dilation and curettage (0.6% vs 1.4%), as well as endometrial hyperplasia (0.1% vs 1.0%), uterine polyps/fibroids (1.2% vs 3.2%), venous thromboembolytic events (1.2% vs 2.3%) and muscle cramp (2.3% vs 4.2%) in the exemestane than in the tamoxifen group, whereas musculoskeletal pain (figure 2), arthralgia (figure 2), diarrhoea (4.2% vs 2.2% of patients), carpal tunnel syndrome (2.8% vs 0.3%), paraesthesia (2.8% vs 1.0%) and joint stiffness (1.9% vs 1.0%) occurred with a significantly higher frequency with exemestane therapy. There was no significant between-group difference in the incidence of cardiovascular events (excluding venous thromboembolism) [figure 2], ischaemic cardiovascular disease (8.0% of exemestane vs 6.9% of tamoxifen recipients) or myocardial infarction (1.3% vs 0.8%).
Comparative tolerability of exemestane (EXE) and tamoxifen (TAM) in postmenopausal women with early-stage breast cancer. Adverse events (graded by the Common Toxicity Criteria of the National Cancer Institute) that occurred on-treatment with an incidence of ≥10% in either treatment group in the randomized, double-blind, multicentre, phase III, IES (median follow-up 55.7 months).[11] Patients (pts) who had already received 2–3 years of oral TAM 20 mg/day were randomized to receive oral EXE 25 mg/day (n = 2320 modified intent-to-treat pts) or to continue receiving TAM 20 mg/day (n = 2338) for 2–3 years, thus completing a total of 5 years of adjuvant endocrine therapy. The prespecified level for statistical significance was p < 0.01. Menopausal symptoms included hot flashes. CV = cardiovascular; MS = musculoskeletal; VT = venous thromboembolism. * p < 0.0001 vs EXE.
In a substudy[19] of the IES in patients with no prior uterine malignancy, such as endometrial cancer, or undiagnosed vaginal bleeding in the year prior to the trial, significantly fewer exemestane recipients than continued tamoxifen recipients had abnormal endometrial thickness (≥5 mm) after 24 months of treatment (35.5% vs 61.8%; p = 0.004), with significant between-group differences in favour of exemestane also seen after 6 (43.5% vs 65.2%; p = 0.01) and 12 (31.2% vs 57.4%; p = 0.004) months (see also section 2).
The tolerability profile of exemestane when used as a primary adjuvant treatment in postmenopausal women with early-stage breast cancer was generally consistent with that seen in the IES switching trial. In the TEAM study,[46] several selected treatment-emergent adverse events occurred in significantly (p ≤ 0.001) fewer exemestane than tamoxifen recipients, including hot flashes (28.5% vs 33.3% of patients), thrombolytic events (0.9% vs 2.3%), vaginal discharge (2.3% vs 6.8%), vaginal haemorrhage (1.6% vs 3.1%), vaginal infection (0.7% vs 2.2%), uterine polyps (0.1% vs 0.5%) and endometrial hyperplasia (0% vs 2.0%), whereas the incidence of arthralgia (17.9% vs 9.2%), arthritis (3.0% vs 1.7%) and hypertension (3.3% vs 2.1%) was significantly (p ≤ 0.001) higher with exemestane.[46] The incidence of cardiac events, such as myocardial ischaemia/infarction (0.8% of exemestane vs 0.6% of tamoxifen recipients) and cardiac death (0.4% vs 0.2%), was low and did not significantly differ between the treatment groups.
In an analysis of ten common menopausal symptoms (assessed by self-report questionnaire) during the first 12 months’ treatment in the TEAM study,[65] significantly (p < 0.05) more exemestane than tamoxifen recipients had bone/muscle aches (77% vs 70% of patients), difficulty sleeping (59% vs 56%; estimated from a graph), decreased libido (58% vs 54%) and vaginal dryness (50% vs 42%), whereas tamoxifen was associated with a significantly (p < 0.0001) higher incidence of vaginal discharge (32% vs 12% of exemestane recipients) and a significantly (p = 0.03) higher mean hot flash score (3.6 vs 2.9). However, there was no significant between-group difference in terms of vaginal bleeding, mood alteration, impaired word finding or low energy.
In postmenopausal women with advanced breast cancer, the most common treatment-emergent adverse events (of those with an incidence >30%) in exemestane or tamoxifen recipients included fatigue/malaise/lethargy (37% vs 37% of patients), hot flashes (35% vs 38%), pain other than in bone (35% vs 31%), bone pain (33% vs 35%) and anaemia (29% vs 36%).[47] Changes (all grades) in levels of ALT (64% of exemestane vs 50% of tamoxifen recipients) and AST (54% vs 56%) were also commonly reported in each of the treatment groups. Serious nonhaematological adverse events (41 vs 61 events) and laboratory parameter changes (30 vs 21 events) of grade 3 or 4 severity were documented in both exemestane and tamoxifen recipients.[47] Notably, weight gain (19% vs 13% of patients), arthralgias (12% vs 5%), hypertension (12% vs 6%) and diarrhoea (9% vs 3%) occurred in ≥5% more exemestane than tamoxifen recipients, whereas anaemia (36% vs 29%), thrombocytopenia (12% vs 6%) and vaginal discharge (7% vs 2%) occurred in ≥5% more tamoxifen than exemestane recipients (descriptive analyses only).[47]
5.1.2 Versus Other Agents
The nature of selected adverse events considered to be drug-related or of indeterminate cause was similar in the exemestane and megestrol groups, with 39.1% and 45.8% of patients experiencing such events.[13] However, fewer exemestane than megestrol recipients withdrew from treatment because of adverse events (1.7% vs 5.0%; p = 0.011). The most commonly reported (i.e. an incidence >10% in either treatment group) adverse events were hot flashes (12.6% vs 5.0% of patients) and fatigue (7.5% vs 10.3%). Exemestane treatment was associated with a greater risk of hot flashes (OR 2.73 [95% CI 1.58, 4.72]), nausea (9.2% vs 5.0% of megestrol recipients; OR 1.93 [95% CI 1.09, 3.43]) and vomiting (2.8% vs 0.8%; OR 3.80 [95% CI 1.04, 13.93]), but a lower risk of dyspnoea (0.3% vs 3.0%; OR 0.09 [95% CI 0.01, 0.70]) than megestrol. In addition, moderate to severe (grade 3 or 4) gains in bodyweight occurred in significantly (p = 0.001) fewer exemestane than megestrol recipients (7.6% vs 17.1%), with bodyweight increases of ≥10% reported in 4% and 21.3% of patients in the respective treatment groups who were overweight at baseline.
Exemestane demonstrated a tolerability profile broadly similar to that of fulvestrant in postmenopausal women with advanced breast cancer (figure 3).[48] The most common treatment-related adverse events that occurred in >5% of patients in either treatment group (other than injection-site pain) were hot flashes, fatigue, nausea and arthralgia. Few exemestane or fulvestrant recipients experienced venous thrombolytic events (0.9% vs 1.1%), serious drug-related adverse events occurred in 0.6% and 1.1% of recipients, and 2.6% and 2% of patients withdrew from therapy because of an adverse event.
Comparative tolerability of exemestane (EXE) and fulvestrant (FUL) in postmenopausal women with advanced breast cancer. Descriptive incidence of drug-related adverse events that occurred in >2% of patients (pts) who received oral EXE 25 mg/day (n = 340 evaluable patients) or intramuscular FUL 250 mg every 4 weeks (n = 351) in a randomized, double-blind, multicentre, phase III study (median follow-up of 13 months) conducted in postmenopausal women with advanced breast cancer whose disease had progressed or recurred during nonsteroidal aromatase inhibitor therapy.[48]
5.2 Osteoporosis and Bone Fractures
As with other aromatase inhibitors, exemestane is associated with loss of BMD (section 2), a risk factor for osteoporosis and fractures. Indeed, when used as a primary adjuvant treatment in postmenopausal women with early-stage breast cancer, exemestane was associated with a significantly (p ≤ 0.001) greater incidence of osteoporosis than tamoxifen after 2.75 years of treatment in the TEAM trial (4.7% vs 2.1%).[46] However, switching to exemestane was not (based on a prespecified p-value of <0.01 being significant) associated with a higher incidence of osteoporosis than continued treatment with tamoxifen in the on-treatment analysis of the IES at a median 55.7-month follow-up (7.3% vs 5.5% of patients) or the corresponding on-treatment plus post-treatment analysis (9.2% vs 7.2%).[11] Moreover, in a substudy[36] of the IES in patients who were not osteoporotic at randomization, 5% of exemestane recipients developed osteoporosis over a 24-month period, although all had been osteopenic at baseline. No patients treated with exemestane who had a healthy BMD at baseline and no patients treated with tamoxifen had developed osteoporosis at this timepoint.
The incidence of fractures was not significantly different between exemestane and tamoxifen recipients in the TEAM trial (2.7% vs 2.3% of patients)[46] or in the on-treatment analysis of the IES (4.3% vs 3.1%).[11] However, significantly more exemestane than tamoxifen recipients had experienced fractures in the IES when analyses included both on-treatment and post-treatment adverse events (7.0% vs 4.9%; OR 1.45 [95% CI 1.13, 1.87]; p = 0.003),[11,36] although most fractures occurred at sites other than the hip, spine or wrist.[11]
6. Pharmacoeconomic Analyses in Early-Stage Breast Cancer
This section reviews recent fully published pharmacoeconomic analyses that used efficacy data from the IES (discussed in section 4.1) to estimate the cost utility of switching to exemestane after 2–3 years of adjuvant tamoxifen relative to 5 years of continued tamoxifen,[66–70] 5 years of anastrozole[68,69] or extended therapy with tamoxifen for 5 years then letrozole for 3 years,[69] in postmenopausal women with early-stage[67–70] or operable[66] breast cancer in an adjuvant treatment setting in a number of countries (table V).
Cost effectiveness of exemestane (EXE) in the treatment of early-stage[67–70] or operable[66] breast cancer in postmenopausal women. Summary of cost-utility analyses of a switch to EXE after 2–3 years of adjuvant tamoxifen (TAM) relative to 5 years of continued treatment with TAM, 5 years of primary adjuvant treatment with anastrozole (ANA) or extended adjuvant treatment with TAM for 5 years then letrozole (LET) for 3 years. Patients (pts) had hormone receptor-positive,[68,69] estrogen receptor-positive (ER+)[66,70] or unknown ER[70] status breast cancer and were aged 60,[68,69] 63[66] or 64[67,70] years, where specified. All analyses used Markov models (5[66,68–70] or 6[67] major health states; 3-,[67] 6-[70] or 12-month[66] cycles, where specified) and incorporated efficacy data from the IES; annual discounts (costs and benefits) were 3%[67–70] or 3.5%[66]
The studies used Markov models to estimate the long-term cost utility of a switch to exemestane relative to other adjuvant therapies. Analyses were stated[66,68,69] or assumed[67,70] to be from a healthcare payer perspective as they included only direct medical costs (drug and other resource costs, including those related to adverse events or complications, such as recurrence or relapse); although the US analysis[70] stated it was from a societal perspective, it did not directly include loss-of-productivity costs. Costs were based on a variety of sources, including healthcare databases,[66,70] expert opinion[67] and literature.[67–70] Utility values were based on literature, and transition values were from the IES and other trials, literature and other sources.
Switching to exemestane after 2–3 years of tamoxifen generally appeared to be cost effective with regard to the cost per quality-adjusted life-years (QALYs) gained compared with continuing tamoxifen in postmenopausal women with early-stage breast cancer in several countries.[67–70] Relative to continued treatment with tamoxifen, switching to exemestane increased costs, but also increased QALYs, resulting in incremental costs per QALY gained that were below widely accepted cost-effectiveness thresholds (e.g. $US50 000 or €30 000 per QALY gained),[67–70] with the exception of 10- and 20-year horizon results in postmenopausal women with operable breast cancer in a Spanish analysis[66] (table V). As might be predicted, the incremental costs per QALY gained appeared to be greater (i.e. less favourable) when shorter time horizons were modelled (table V).[66,68]
In a US analysis,[70] the cost effectiveness of the exemestane regimen relative to 5 years of continued tamoxifen appeared to be marginally greater in patients with known estrogen receptor-positive disease than in patients whose disease was estrogen receptor-positive or of unknown receptor status. A cost-effectiveness acceptability curve demonstrated that the probability of a switch from tamoxifen to exemestane being cost effective relative to continued tamoxifen was 70.5% and 96.4% at acceptability thresholds of $US25 000 and $US50 000 per QALY gained in postmenopausal women with early-stage estrogen receptor-positive or unknown receptor status breast cancer.[70]
When consequences of coronary heart disease were included in a Swedish analysis,[67] a switch to exemestane still appeared to be cost effective relative to continued tamoxifen, with an incremental cost per QALY gained of €31 434 (year of costing 2005). Tamoxifen was assumed to be associated with a reduced risk of coronary heart disease relative to exemestane for a total of 11 years post-treatment initiation in this analysis. The incremental cost per QALY gained for the exemestane regimen versus tamoxifen remained below €50 000 when the period of reduced risk associated with tamoxifen was increased to 14 and 18 years, but not when increased to 24 years.[67]
Analyses from Belgian[69] and Canadian[68] healthpayer perspectives over 20-year time horizons predicted that a switch to exemestane after 2.5 years of adjuvant tamoxifen was dominant (i.e. less costly and more effective) over both first-line treatment with adjuvant anastrozole (5 years)[68,69] and extended adjuvant therapy with tamoxifen (5 years) followed by letrozole (3 years)[69] with regard to the cost per QALY gained (table V). Furthermore, estimates over a 10-year horizon in Canada suggested that first-line treatment with anastrozole was not a cost-effective option relative to a switch from tamoxifen to exemestane, as the incremental costs per QALY gained were well in excess of the accepted cost-effectiveness threshold of $Can50 000.[68] Moreover, data from a Spanish analysis[66] suggested that switching to exemestane after 2–3 years of tamoxifen instead of continuing tamoxifen was associated with incremental costs per QALY gained that were lower than those associated with comparisons of other regimens (table V). These data should be interpreted with caution owing to the marked differences in the regimens compared and the indirect nature of the comparisons.
Sensitivity analyses conducted in the studies demonstrated that overall findings were generally robust to plausible variations in most key assumptions and input parameters.[66–70]
However, as with all pharmacoeconomic studies, pharmacoeconomic analyses of exemestane are subject to several inherent limitations. Pharmacoeconomic analyses based on clinical trials extrapolate the results of such trials to the general population; however, patient populations, rates of compliance and major outcomes in clinical trials may differ from those observed in real-life practice. Modelled analyses, such as those presented in this section,[66–70] rely on a number of assumptions and use data from a variety of sources. The studies were generally well designed, although some analyses[66,68,69] incorporated data from trials that did not directly compare the various regimens owing to a lack of relevant head-to-head clinical trial data. Results of pharmacoeconomic analyses may not be applicable to other geographical regions because of differences in healthcare systems, medical practice and unit costs.
7. Dosage and Administration
Exemestane is approved in the UK,[7] US[8] and several other countries worldwide as an adjuvant treatment in postmenopausal women with estrogen receptor-positive early-stage (invasive[7]) breast cancer following 2–3 years of adjuvant tamoxifen treatment, as well as for the treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen or other antiestrogen therapy.
The recommended dosage is exemestane 25 mg administered orally once daily after a meal.[7,8] In patients with early-stage disease who have received 2–3 years of tamoxifen treatment, exemestane should be administered until a total of 5 years of adjuvant endocrine therapy has been received, or until tumour relapse[7]/recurrence[8] or contralateral breast cancer[8] occurs. In patients with advanced disease, exemestane treatment should continue until there is evidence of tumour progression.
Dosage adjustments are not required in patients with hepatic or renal impairment (section 3).[7,8] However, in patients receiving drugs that are potent inducers of CYP3A4 enzymes, such as phenytoin or rifampicin, an increased exemestane dosage of 50 mg/day is recommended in the US.[8] Local prescribing information should be consulted for further information regarding contraindications, warnings, drug interactions and other precautions.
8. Place of Exemestane in the Management of Breast Cancer in Postmenopausal Women
The benefits of endocrine therapy in the treatment of breast cancer have been known for over 100 years, and is today the cornerstone therapy in management of the disease.[77] The selective estrogen receptor modulator tamoxifen, which blocks estrogen activity at the receptor level, was the gold standard endocrine therapy for the treatment of both early-stage and advanced hormone receptor-positive breast cancer for many years. However, safety concerns relating to certain estrogenic effects, including an increased risk of endometrial cancer and thromboembolytic disease, as well as the problem of developing resistance to the drug, prompted development of alternative antiestrogen agents.[78]
The aromatase inactivators/inhibitors are one such group of agents, inhibiting the peripheral production of estrogens and thus lacking any estrogenic activity. Currently available third-generation aromatase inactivators/inhibitors include the steroidal aromatase inactivator exemestane and the nonsteroidal inhibitors anastrozole and letrozole.[79] Fadrozole, an earlier second-generation nonsteroidal aromatase inhibitor, is available only in Japan. Other endocrine agents, available for the treatment of advanced breast cancer, include progestins (e.g. megestrol), androgens and high-dose estrogen,[79] although their mechanisms of action are not clearly understood. More recently, the estrogen receptor down-regulator fulvestrant has become available for use in this patient population.[79]
In early-stage breast cancer, the overall aim of treatment is to increase the time to tumour progression and improve overall patient survival.[80] The mainstay endocrine treatment for postmenopausal women with early-stage hormone receptor-positive breast cancer has historically been 5 years of adjuvant tamoxifen.[81,82] However, data from recent clinical trials using third-generation aromatase inactivators/inhibitors instead of, or in sequence with, tamoxifen indicated that these agents were more effective in preventing disease recurrence than tamoxifen in this patient population.[81,82] As a result, aromatase inactivators/inhibitors presently challenge tamoxifen as the standard of care for the treatment of early-stage hormone receptor-positive breast cancer in postmenopausal women.[82]
According to current treatment guidelines[79,83] and the 10th St Gallen expert consensus meeting on the primary therapy of early breast cancer,[84] postmenopausal women with hormone receptor-positive disease can be given aromatase inactivator/inhibitor therapy upfront for 5 years or following 2–3 or 5 years of tamoxifen therapy. The treatment guidelines[79,83] recommend that aromatase inactivators/inhibitors are used at some stage during the treatment of all postmenopausal women for whom endocrine therapy is appropriate. However, in the St Gallen consensus meeting,[84] a switch to an aromatase inactivator/inhibitor after 2–3 years of tamoxifen was the preferred aromatase inactivator/inhibitor treatment strategy, with the majority of the panel considering 5 years of tamoxifen to be a viable treatment option in some patients.
Exemestane was approved in the UK,[7] the US[8] and several other countries for use as an adjuvant treatment in postmenopausal women with estrogen receptor-positive early-stage breast cancer after 2–3 years of adjuvant tamoxifen treatment based on the results of the large well designed IES (section 4.1.1). According to data from this study, exemestane for 2–3 years after 2–3 years of adjuvant tamoxifen is generally more effective than 5 years of continuous adjuvant tamoxifen in the treatment of postmenopausal women with early-stage breast cancer (section 4.1.1). Switching from tamoxifen to exemestane was consistently associated with a significantly lower risk of experiencing one of the events (recurrence, contralateral breast cancer or death without recurrence) in the primary combined endpoint of disease-free survival than continued tamoxifen at median follow-ups of 30.6 and 55.7 months. The exemestane regimen was also more effective with regard to other endpoints, including the risk of contralateral breast cancer and breast cancer-free survival. There was generally no overall survival benefit in switching to exemestane, except in patients with estrogen receptor-positive or unknown receptor status disease at the median 55.7-month follow-up, when nodal status, HRT and chemotherapy use were adjusted for. Whether switching from tamoxifen to an aromatase inhibitor/inactivator provides an overall survival benefit over continued tamoxifen in postmenopausal women with early-stage breast cancer is unclear at present, with variable results reported in the analyses of three large clinical studies that compared a switch to anastrozole with continued tamoxifen therapy.[85]
Anastrozole is the only third-generation aromatase inhibitor to have been approved for use in this indication (in the UK[86] and a number of other countries), based on the findings of several studies.[81,85] However, the efficacy of letrozole in sequence with tamoxifen (2 years of tamoxifen followed by 3 years of letrozole; 2 years of letrozole followed by 3 years of tamoxifen) in the adjuvant setting was not significantly different from that of primary adjuvant treatment with letrozole, according to limited data from a trial available as an abstract.[87]
Although, at present, exemestane is not approved for use as a primary adjuvant treatment in postmenopausal women with hormone receptor-positive early-stage breast cancer, preliminary data from the large, randomized, open-label, phase III, TEAM trial comparing exemestane with tamoxifen indicate that exemestane 25 mg/day is effective in this setting (section 4.1.2). After 2.75 years, disease-free (primary endpoint) or relapse-free survival did not significantly differ between exemestane and tamoxifen recipients, although exemestane was more effective with regard to the risk of distant metastases. However, the TEAM trial has since been revised to compare the efficacy of 5 years of exemestane therapy with that of sequential treatment with tamoxifen for 2–3 years followed by exemestane for 2–3 years based on the findings of the IES.[12] In contrast to exemestane, both anastrozole and letrozole are approved for use as primary adjuvant treatments in this patient population in the UK,[86,88] the US[89,90] and other countries worldwide on the basis of the findings of clinical studies.[81]
For women who have received 5 years of hormonal therapy there remains a persistent risk of relapse.[82] Benefits of extending adjuvant treatment with an aromatase inhibitor following standard adjuvant treatment with tamoxifen have been demonstrated for both letrozole and anastrozole in postmenopausal women with early-stage breast cancer.[81] Currently, only letrozole is approved for such an indication (in several countries worldwide, including the UK[88] and US[90]).
Several questions still surround the use of exemestane and the aromatase inhibitors in postmenopausal women with early-stage breast cancer. For instance, because of the lack of comparative trials, it is not clear whether there are differences in efficacy between the agents. It is also not yet clear what the optimal duration of therapy is, whether it is beneficial to include a period of tamoxifen treatment before treatment with an aromatase inactivator/inhibitor and which patients may benefit the most from therapy. However, data from ongoing head-to-head trials[64] and other studies may help to provide answers to some of these questions.
Advanced breast cancer is incurable, with most patients dying due to progression of the disease.[78] As treatment in this patient population is predominantly palliative, factors affecting HR-QOL, such as toxicity, are important. Thus, endocrine therapy is generally preferred over chemotherapy in patients with advanced hormone receptor-positive breast cancer, unless the disease is rapidly progressive in nature.[78]
Current treatment guidelines commonly recommend third-generation aromatase inactivators/inhibitors as first-line treatment options for postmenopausal women with advanced hormone receptor-positive breast cancer,[79,91,92] with tamoxifen also considered an acceptable first-line agent in certain instances in some guidelines[79,91] (e.g. in patients naive to antiestrogen therapy or who received antiestrogen treatment >1 year previously[79]).
For patients who experience progression during treatment with an antiestrogen, such as tamoxifen, the previous standard of care was treatment with the progestin megestrol or the non-selective first-generation aromatase inhibitor aminoglutethimide.[93] However, these agents tend to be poorly tolerated, with adverse effects commonly leading to discontinuation of therapy.[93] For instance, progestins are associated with shortness of breath, gains in bodyweight and thromboembolytic disease, whereas aminoglutethimide is associated with drowsiness, vertigo, skin rashes and blood dyscrasias and requires concomitant corticosteroid replacement therapy.[94] Important advances over these agents have been seen with third-generation aromatase inactivators/inhibitors, which are now generally the preferred second-line endocrine therapy option for the treatment of advanced hormone receptor-positive breast cancer in postmenopausal women.[95]
Exemestane is approved for the treatment of advanced breast cancer in postmenopausal women whose disease has progressed following tamoxifen or other antiestrogen therapy in several countries, including the UK[7] and the US;[8] anastrozole and letrozole are also approved for similar indications.[88–90]
In a large, well designed, phase III study, exemestane provided similar efficacy to megestrol in terms of objective response rate (primary endpoint) in postmenopausal women with advanced breast cancer refractory to tamoxifen (section 4.2.1). Although there was also no significant difference between the treatments in the median duration of objective response or the rate of overall success, exemestane recipients had a significantly longer median duration of overall success, time to disease progression and time to treatment failure than megestrol recipients.
In light of the increasing use of aromatase inactivators/inhibitors in the adjuvant treatment of breast cancer, it is ever more likely that patients presenting with advanced disease will already have been treated with such agents, and some patients will have progressed after receiving aromatase inactivator/inhibitor therapy for advanced disease. Results from a large, well designed, phase III study indicate that the efficacy of exemestane is not significantly different from that of intramuscular fulvestrant in patients with advanced breast cancer refractory to a nonsteroidal aromatase inhibitor, with regard to time to disease progression (primary endpoint) and other endpoints, including objective response rate, clinical benefit rate or overall survival (section 4.2.1).
Unlike exemestane and the third-generation aromatase inhibitors, which are administered orally daily, fulvestrant is administered intramuscularly once monthly, which some patients may find inconvenient. Fulvestrant has recently been approved for the treatment of postmenopausal women with hormone receptor-positive locally advanced[96] or metastatic[96,97] breast cancer with disease progression on (EU)[96] or following (US)[97] antiestrogen therapy and for relapse on/after adjuvant antiestrogen treatment (EU).[96]
Although exemestane is not currently approved for use in this setting, data, some of which are preliminary, suggest that exemestane is an effective first-line hormonal treatment in postmenopausal women with hormone receptor-positive advanced breast cancer (section 4.2.2). There were generally no differences between exemestane and tamoxifen or anastrozole in terms of efficacy, although a significantly higher objective response rate was seen with exemestane than with tamoxifen in the largest phase III study. However, the widely available third-generation aromatase inhibitors, anastrozole and letrozole, have also demonstrated efficacy relative to tamoxifen in the first-line treatment of such patients in large well designed studies,[6] and are approved for use as first-line treatment options in postmenopausal women with locally advanced or metastatic breast cancer that is hormone receptor-positive or of unknown receptor status in the US[89,90] and for similar indications in other countries, including the UK.[88]
As yet, there are no direct comparisons of exemestane and the aromatase inhibitors in patients refractory to antiestrogen therapy, and additional robust head-to-head clinical trials are required to evaluate the efficacy of these agents relative to fulvestrant in this setting. Further head-to-head trials are also warranted to evaluate the efficacy of exemestane with respect to the aromatase inhibitors in the first-line treatment setting and to establish the optimal sequence of endocrine therapies.
Exemestane is generally well tolerated in postmenopausal women with early-stage or advanced breast cancer, with most adverse events being mild to moderate in severity and of a similar nature irrespective of the stage of disease (section 5). As with tamoxifen and the aromatase inhibitors,[98] exemestane is commonly associated with adverse events typical of the menopause, such as hot flashes (section 5). The tolerability profile of exemestane is similar to that of fulvestrant in women with advanced breast cancer (section 5.1.2). However, the tolerability profiles of the aromatase inactivators/inhibitors are different to, and for some tolerability issues better than, those of traditional endocrine therapies.
Tamoxifen has estrogenic effects on some tissues and is consequently associated with gynaecological symptoms, such as vaginal bleeding, an increased risk of endometrial cancer, as well as thromboembolic events, including deep vein thrombosis, stroke and pulmonary embolism.[99] Some of these tolerability issues, such as increased endometrial cancer risk, are of particular relevance to patients with early-stage breast cancer who may receive tamoxifen therapy for longer than those with advanced disease. Relative to tamoxifen, exemestane was associated with a significantly lower incidence of certain gynaecological and thrombolytic events in the IES and TEAM studies (section 5.1.1). Moreover, substudy data from the IES suggest that endometrial abnormalities associated with tamoxifen may be reversed after switching to exemestane in the adjuvant setting (sections 2 and 5.1.1).
Exemestane also has some tolerability advantages over megestrol in postmenopausal women with advanced breast cancer, with exemestane recipients less likely to experience dyspnoea or moderate to severe gains in bodyweight; however, exemestane recipients are more likely to experience hot flashes, nausea and vomiting (section 5.1.2).
As is typical of the aromatase inhibitors, exemestane is more commonly associated with musculoskeletal adverse events, such as arthralgias, than tamoxifen (section 5), although determining the precise origin of arthralgia can be difficult because of the joint pain often associated with the degenerative changes that typically occur in menopausal women.[80] However, arthralgias can often be managed with lifestyle modifications (e.g. weight-bearing exercise), physiotherapy and analgesics, such as paracetamol (acetaminophen) or NSAIDs.[80]
The main concern with aromatase inactivator/inhibitor treatment, particularly in the adjuvant setting, is the risk of skeletal events, such as osteoporosis and fractures, due to estrogen depletion and bone demineralization (section 2).[99] Tamoxifen has estrogenic effects on bone that help to protect against menopause-associated bone loss,[99] and was associated with a significantly lower incidence of osteoporosis or fractures than exemestane in some, but not all, trials or substudies (section 5.2). The UK prescribing information for exemestane recommend that BMD is monitored when commencing adjuvant treatment in patients with, or at risk of, osteoporosis, and that treatment for osteoporosis is given to patients at risk of the disease.[7] Similar recommendations apply to both anastrozole and letrozole.[86,88] It may be possible to minimize bone loss during aromatase inactivator/inhibitor therapy with the use of bisphosphonates.[100]
Anastrozole and letrozole regimens have been associated with an increased incidence of fractures relative to tamoxifen in some studies,[99] with more letrozole than placebo recipients reporting osteoporosis in an extended adjuvant trial.[99] The effect of exemestane on biochemical bone marker levels did not significantly differ from those seen with anastrozole or letrozole in healthy postmenopausal women (section 2); however, the agents have yet to be directly compared in terms of osteoporosis and fracture risk in robust clinical trials in patients with breast cancer.
Unlike tamoxifen, which may improve lipid parameters, there is some concern regarding the effect that aromatase inactivators/inhibitors may have on the lipid profile, and thus, a patient’s cardiovascular risk.[99] At present, the effect of exemestane (section 2) and the aromatase inhibitors on lipid levels are not clear, with the findings of some adjuvant studies comparing these agents with tamoxifen considered possibly misleading because of the potentially beneficial effects of tamoxifen.[99] However, the incidence of various cardiovascular adverse events was not significantly different between exemestane and tamoxifen recipients in the IES and TEAM studies (section 5.1.1). The effect of agents such as exemestane on bone and lipid parameters has not been as intensively studied in patients with advanced breast cancer, as data interpretation is limited by the bone metastases and metabolic disturbances often present in such patients.[101]
Given that breast cancer is the leading cause of death from cancer among women worldwide,[1] it follows that treatment choices may be determined in part by pharmacoeconomic factors. Fully published pharmacoeconomic model analyses that included direct medical costs have indicated that switching to exemestane after 2–3 years of tamoxifen is generally cost effective with regard to the cost per QALY gained relative to continuing tamoxifen treatment in postmenopausal women with early-stage breast cancer (section 6). Using data from trials that did not directly compare therapeutic regimens, some analyses predicted that switching from tamoxifen to exemestane was generally cost saving relative to primary adjuvant treatment with anastrozole or extended adjuvant therapy with tamoxifen followed by letrozole with regard to the cost per QALY gained in postmenopausal women with early-stage breast cancer when costs and benefits were modelled over a 20-year time horizon (section 6). However, the findings of such indirect analyses should be interpreted with caution. Pharmacoeconomic studies evaluating whether there are any pharmacoeconomic differences between primary adjuvant treatment with exemestane and sequential treatment with tamoxifen and exemestane would be of interest, as would pharmacoeconomic studies in advanced breast cancer.
In conclusion, exemestane is effective for the treatment of postmenopausal women with early-stage or advanced breast cancer. In early-stage disease, switching to exemestane for 2–3 years after 2–3 years of adjuvant tamoxifen treatment was more effective in prolonging disease-free survival than continuing tamoxifen therapy, although it was not associated with an overall survival benefit, except in those with estrogen receptor-positive or unknown receptor status disease when nodal status, HRT and chemotherapy use were adjusted for. Moreover, preliminary data suggest that the efficacy of exemestane is generally no different to that of tamoxifen in the primary adjuvant treatment of early-stage breast cancer, although exemestane may be better in prolonging the time to distant metastases. In advanced disease, exemestane showed equivalent efficacy to megestrol in patients with disease refractory to tamoxifen and an efficacy not significantly different from that of fulvestrant in those refractory to a nonsteroidal aromatase inhibitor. Available data, some of which are limited, suggest exemestane is also effective in the first-line hormonal treatment of advanced breast cancer in postmenopausal women. Exemestane is generally well tolerated, although the potential bone fracture risk of the drug requires further investigation and comparison with that of other aromatase inhibitors. Additional comparative data are required to definitively position exemestane with respect to other agents, in both early-stage and advanced disease, and to determine the optimal sequence of endocrine therapies. Data from ongoing trials in patients with early-stage breast cancer directly comparing exemestane with the third-generation aromatase inhibitors may provide an indication as to the relative efficacy and tolerability of these agents, at least in the adjuvant setting. In the meantime, switching to exemestane should be considered in postmenopausal women who have received 2–3 years of adjuvant tamoxifen treatment for early-stage breast cancer and is an emerging treatment option for postmenopausal women with advanced breast cancer refractory to one or more antiestrogen therapies.
Disclosure
The preparation of this review was not supported by any external funding. During the peer review process, the manufacturer of the agent under review was offered an opportunity to comment on this article. Changes resulting from comments received were made on the basis of scientific and editorial merit.
References
American Cancer Society. Global cancer facts and figures 2007 [online]. Available from URL: http://www.cancer.org/downloads/STT/Global_Cancer_Facts_and_Figures_2007_rev.pdf [Accessed 2008 Jul 30]
Travis RC, Key TJ. Oestrogen exposure and breast cancer risk. Breast Cancer Res 2003 Jul 28; 5(5): 239–47
Foster PA. Steroid metabolism in breast cancer. Minerva Endocrinol 2008 Mar; 33(1): 27–37
Suzuki T, Miki Y, Nakamura Y, et al. Sex steroid-producing enzymes in human breast cancer. Endocr Relat Cancer 2005 Dec; 12(4): 701–20
Smith IE, Dowsett M. Aromatase inhibitors in breast cancer. N Eng J Med 2003 Jun 12; 348(24): 2431–42
Rugo HS. The breast cancer continuum in hormone-receptor-positive breast cancer in postmenopausal women: evolving management options focusing on aromatase inhibitors. Annal Oncol 2008 Jan; 19(1): 16–27
Pharmacia Ltd. Aromasin® 25 mg coated tablets: summary of product characteristics [online]. Available from URL: http://www.emc.medicines.org.uk [Accessed 2009 Mar 13]
Pharmacia and Upjohn Co. Aromasin® exemestane tablets: US prescribing information [online]. Available from URL: http://www.pfizer.com/files/products/uspi_aromasin.pdf [Accessed 2009 Apr 28]
Moen MD, Wagstaff AJ. Exemestane: a review of its use as adjuvant treatment for early breast cancer in postmenopausal women. Am J Cancer 2006; 5(4): 259–72
Clemett D, Lamb HM. Exemestane: a review of its use in postmenopausal women with advanced breast cancer. Drugs 2000 Jun; 59(6): 1279–96
Coombes RC, Kilburn LS, Snowdon CF, et al. Survival and safety of exemestane versus tamoxifen after 2–3 years’ tamoxifen treatment (Intergroup Exemestane Study): a randomised controlled trial. Lancet 2007 Feb 17; 369(9561): 559–70
Coombes RC, Hall E, Gibson LJ, et al. A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Eng J Med 2004 Mar 11; 350(11): 1081–92
Kaufmann M, Bajetta E, Dirix LY, et al. Exemestane is superior to megestrol acetate after tamoxifen failure in postmenopausal women with advanced breast cancer: results of a phase III randomized double-blind trial. J Clin Oncol 2000 Apr; 18(7): 1399–411
Aihara T, Hozumi Y, Suemasu K, et al. The effects of exemestane, anastrozole and tamoxifen on bone mineral density and bone turnover markers in postmenopausal early breast cancer patients: preliminary results of N-SAS (National Surgical Adjuvant Study) BC04, the TEAM Japan sub-study [abstract no. 2086]. Breast Cancer Res Treat 2007 Dec; 106 Suppl. 1: 114. Plus poster presented at the 30th Annual San Antonio Breast Cancer Symposium; 2007 Dec 13–16; San Antonio (TX)
van Nes JGH, Papapoulos SE, Braun JJ, et al. Effect of exemestane on bone turnover markers and bone mineral density (BMD): 2 year results of the Dutch/Belgian Tamoxifen Exemestane Adjuvant Multicentre (TEAM) trial [abstract no. 1154]. Cancer Res 2009 Jan 15; 69 (2 Suppl.): 152s
McCaig FM, Renshaw L, Williams L, et al. A randomized study of the effects of anastrozole (A), letrozole (L) and exemestane (E) on bone turnover [abstract no. 1145]. Cancer Res 2009 Jan 15; 69 (2 Suppl.): 149s
Coleman RE, Banks LM, Girgis S, et al. Reversal of skeletal effects of endocrine treatments in the intergroup study [abstract no. 1128]. Cancer Res 2009 Jan 15; 69 (2 Suppl.): 144s
McCaig FM, Renshaw L, Williams L, et al. A randomised study comparing the effects of anastrozole (A), letrozole (L), exemestane (E) and tamoxifen (T) on coagulation [abstract no. 1132]. Cancer Res 2009 Jan 15; 69 (2 Suppl.): 145s
Bertelli G, Hall E, Ireland E, et al. Endometrial status in the Intergroup Exemestane Study up to 2 years post-treatment [abstract no. 2078]. Breast Cancer Res Treat 2007 Dec; 106 Suppl. 1: 111. Plus poster presented at the 30th Annual San Antonio Breast Cancer Symposium; 2007 Dec 13–16; San Antonio (TX)
Wang X, Chen S. Aromatase destabilizer: novel action of exemestane, a food and drug administration-approved aromatase inhibitor. Cancer Res 2006 Nov 1; 66(21): 10281–6
Lønning PE, Geisler J, Krag LE, et al. Effects of exemestane administered for 2 years versus placebo on bone mineral density, bone biomarkers, and plasma lipids in patients with surgically resected early breast cancer. J Clin Oncol 2005 Aug 1; 23(22): 5126–37
Montagnani A, Gonnelli S, Cadirni A, et al. The effects on lipid serum levels of a 2-year adjuvant treatment with exemestane after tamoxifen in postmenopausal women with early breast cancer. Eur J Int Med 2008 Dec; 19(8): 592–7
Markopoulos C, Polychronis A, Zobolas V, et al. The effect of exemestane on the lipidemic profile of post-menopausal early breast cancer patients: preliminary results of the TEAM Greek sub-study. Breast Cancer Res Treat 2005 Sep; 93(1): 61–6
Markopoulos C, Chrissochou M, Michailidou A, et al. Effect of exemestane on the lipidemic profile of post-menopausal operable breast cancer patients following 5–7 years of adjuvant tamoxifen: preliminary results of the ATENA sub-study. Anticancer Drugs 2005 Sep; 16(8): 879–83
Francini G, Petrioli R, Montagnani A, et al. Exemestane after tamoxifen as adjuvant hormonal therapy in postmenopausal women with breast cancer: effects on body composition and lipids. Br J Cancer 2006 Jul 17; 95(2): 153–8
Anker GB, Refsum H, Ueland PM, et al. Influence of aromatase inhibitors on plasma total homocysteine in postmenopausal breast cancer patients. Clin Chem 1999 Feb; 45(2): 252–6
Zilembo N, Noberasco C, Bajetta E, et al. Endocrinological and clinical evaluation of exemestane, a new steroidal aromatase inhibitor. Br J Cancer 1995; 72(4): 1007–12
Geisler J, King N, Anker G, et al. In vivo inhibition of aromatization by exemestane, a novel irreversible aromatase inhibitor, in postmenopausal breast cancer patients. Clin Cancer Res 1998 Sep; 4(9): 2089–93
Johannessen DC, Engan T, Di Salle E, et al. Endocrine and clinical effects of exemestane (PNU 155971), a novel steroidal aromatase inhibitor, in postmenopausal breast cancer patients: a phase I study. Clin Cancer Res 1997 Jul; 3(7): 1101–8
Atalay G, Dirix L, Biganzoli L, et al. The effect of exemestane on serum lipid profile in postmenopausal women with metastatic breast cancer: a companion study to EORTC Trial 10951, ’Randomized phase II study in first line hormonal treatment for metastatic breast cancer with exemestane or tamoxifen in postmenopausal patients’. Ann Oncol 2004 Feb; 15(2): 211–7
Evans TR, Di Salle E, Ornati G, et al. Phase I and endocrine study of exemestane (FCE 24304), a new aromatase inhibitor, in postmenopausal women. Cancer Res 1992 Nov 1; 52(21): 5933–9
McClosky EV, Hannon RA, Lakner G, et al. Effects of third generation aromatase inhibitors on bone health and other safety parameters: results of an open, randomised, multi-centre study of letrozole, exemestane and anastrozole in healthy postmenopausal women. Eur J Cancer 2007 Nov; 43(17): 2523–31
Geisler J, Lønning PE, Krag LE, et al. Changes in bone and lipid metabolism in postmenopausal women with early breast cancer after terminating 2-year treatment with exemestane: a randomised, placebo-controlled study. Eur J Cancer 2006 Nov; 42(17): 2968–75
Lonn E. Homocysteine in the prevention of ischemic heart disease, stroke and venous thromboembolism: therapeutic target or just another distraction? Curr Opin Hematol 2007 Sep; 14(5): 481–7
Humphrey LL, Fu R, Rogers K, et al. Homocysteine levels and coronary heart disease incidence: a systemic review and meta-analysis. Mayo Clin Proc 2008 Nov; 83(11): 1203–12
Coleman RE, Banks LM, Girgis SI, et al. Skeletal effects of exemestane on bone-mineral density, bone biomarkers, and fracture incidence in postmenopausal women with early breast cancer participating in the Intergroup Exemestane Study (IES): a randomised controlled study. Lancet Oncol 2007 Feb; 8(2): 119–27
Gonnelli S, Cadirni A, Caffarelli C, et al. Changes in bone turnover and in bone mass in women with breast cancer switched from tamoxifen to exemestane. Bone 2007 Jan; 40(1): 205–10
Hadji P, Ziller M, Kieback DG, et al. Effects of exemestane and tamoxifen on bone health within the Tamoxifen Exemestane Adjuvant Multicentre (TEAM) trial: results of a German, 12-month, prospective, randomised sub-study. Ann Oncol. Epub 2009 Feb 13
Hadji P, Ziller M, Kieback DG, et al. The effect of exemestane or tamoxifen on markers of bone turnover: results of a German sub-study of the Tamoxifen Exemestane Adjuvant Multicentre (TEAM) trial. Breast Epub 2009 Apr 11
Jones S, Stokoe C, Sborov M, et al. The effect of tamoxifen or exemestane on bone mineral density during the first 2 years of adjuvant treatment of postmenopausal women with early breast cancer. Clin Breast Cancer 2008 Dec; 8(6): 527–32
Spinelli R, Jannuzzo MG, Poggesi I, et al. Pharmacokinetics (PK) of Aromasin® (exemestane, EXE) after single and repeated doses in healthy postmenopausal volunteers (HPV) [abstract no. 1185]. Eur J Cancer 1999 Sep; 35 Suppl. 4: S295
Poggesi I, Jannuzzo MG, Di Salle E, et al. Effect of food and formulation on the pharmacokinetics and pharmacodynamics of a single oral dose of exemestane (Aromasin®, EXE) [abstract no. 741]. Proc Am Soc Clin Oncol 1999; 18: 193a
Jannuzzo MG, Spinelli R, Poggesi I, et al. Inhibition of CYP3A4 does not influence Aromasin® (exemestane, EXE) pharmacokinetics (PK) in healthy postmenopausal volunteers (HPV) [abstract no. 1184]. Eur J Cancer 1999 Sep; 35 Suppl. 4: S294
Jannuzzo MG, Poggesi I, Spinelli R, et al. The effects of degree of hepatic or renal impairment on the pharmacokinetics of exemestane in postmenopausal women. Cancer Chemother Pharmacol 2004 Jun; 53(6): 475–81
Mayordomo J, Llombart A, Martin M, et al. Randomized, multicenter, crossover phase II trial to compare exemestane vs. anastrozole in postmenopausal patients with advanced breast cancer and positive hormone receptors. Final efficacy analysis of GEICAM 2001-03 study [abstract no. 638]. J Clin Oncol 2006 Jun 20; 24 (18 Suppl.): 37
Jones SE, Seynaeve C, Hasenburg A, et al. Results of the first planned analysis of the TEAM (tamoxifen exemestane adjuvant multinational) prospective randomized phase III trial in hormone sensitive postmenopausal early breast cancer [abstract no. 15 plus presentation]. San Antonio Breast Cancer Symposium 2008 Dec 10–14; San Antonio (TX)
Paridaens RJ, Dirix LY, Beex LV, et al. Phase III study comparing exemestane with tamoxifen as first-line hormonal treatment of metastatic breast cancer in postmenopausal women: the European Organisation for Research and Treatment of Cancer Breast Cancer Cooperative Group. J Clin Oncol 2008 Oct 20; 26(30): 4883–90
Chia S, Gradishar W, Mauriac L, et al. Double-blind, randomized placebo controlled trial of fulvestrant compared with exemestane after prior nonsteroidal aromatase inhibitor therapy in postmenopausal women with hormone receptor-positive, advanced breast cancer: results from EFECT. J Clin Oncol 2008 Apr 1; 26(10): 1664–70
Chernozemsky I, Kalinov K, Tzekov H, et al. Randomized phase III trial of exemestane or tamoxifen in first-line hormonal treatment of postmenopausal women with metastatic breast cancer [abstract no. 2101]. Breast Cancer Res Treat 2007 Dec; 106 Suppl. 1: 119
Mamounas EP, Jeong JH, Wickerham DL, et al. Benefit from exemestane as extended adjuvant therapy after 5 years of adjuvant tamoxifen: intention-to-treat analysis of the National Surgical Adjuvant Breast and Bowel Project B-33 Trial. J Clin Oncol 2008 Apr 20; 26(12): 1965–71
Fallowfield LJ, Bliss JM, Porter LS, et al. Quality of life in the Intergroup Exemestane Study: a randomized trial of exemestane versus continued tamoxifen after 2 to 3 years of tamoxifen in postmenopausal women with primary breast cancer. J Clin Oncol 2006 Feb 20; 24(6): 910–7
Fallowfield LJ, Langridge CI, Kilburn LS, et al. Quality of life in the Intergroup Exemestane Study 5 years post-randomisation [abstract no. 1091]. Breast Cancer Res Treat 2007 Dec; 106 Suppl. 1: 73. Plus poster presented at the 30th San Antonio Breast Cancer Symposium; 2007 Dec 13–17; San Antonio, (TX)
van Nes JGH, Voskuil DW, van Leeuwen FE, et al. Quality of life in relation to hormonal treatment of postmenopausal women in the Dutch Tamoxifen Exemestane Adjuvant Multicentre (TEAM) trial [abstract no. 21]. Cancer Res 2009 Jan 15; 69 (2 Suppl.): 69s
Laffaioli RV, Formato R, Tortoriello A, et al. Phase II study of sequential hormonal therapy with anastrozole/exemestane in advanced and metastatic breast cancer. Br J Cancer 2005; 92(9): 1621–5
Thürlimann B, Paridaens R, Serin D, et al. Third-line hormonal treatment with exemestane in postmenopausal patients with advanced breast cancer progressing on aminoglutethimide: a phase II multicentre multinational study. Eur J Cancer 1997 Oct; 33(11): 1767–73
Gennatas C, Michalaki V, Carvounis E, et al. Third-line hormonal treatment with exemestane in postmenopausal patients with advanced breast cancer progressing on letrozole or anastrozole. A phase II trial conducted by the Hellenic Group of Oncology (HELGO). Tumori 2006 Jan–Feb; 92(1): 13–7
Jones S, Vogel C, Arkhipov A, et al. Multicenter, phase II trial of exemestane as third-line hormonal therapy of postmenopausal women with metastatic breast cancer. J Clin Oncol 1999 Nov; 17(11): 3418–25
Kvinnsland S, Anker G, Dirix LY, et al. High activity and tolerability demonstrated for exemestane in postmenopausal women with metastatic breast cancer who had previously failed on tamoxifen treatment. Eur J Cancer 2000 May; 36(8): 976–82
Lønning PE, Bajetta E, Murray R, et al. Activity of exemestane in metastatic breast cancer after failure of nonsteroidal aromatase inhibitors: a phase II trial. J Clin Oncol 2000 Jun; 18(11): 2234–44
Bertelli G, Garrone O, Merlano M, et al. Sequential treatment with exemestane and non-steroidal aromatase inhibitors in advanced breast cancer. Oncology 2005; 69(6): 471–7
Campos SM, Guastalla JP, Subar M, et al. A comparative study of exemestane versus anastrozole in patients with postmenopausal breast cancer with visceral metastases. Clin Breast Cancer 2009 Feb; 9(1): 39–44
Chia S, Piccart M, Gradishar W, et al. Fulvestrant vs exemestane following non-steroidal aromatase inhibitor failure: first overall survival data from the EFECT trial [abstract no. 2091]. 30th Annual San Antonio Breast Cancer Symposium; 2007 Dec 13–16; San Antonio (TX)
Paridaens R, Dirix L, Lohrisch C, et al. Mature results of a randomized phase II multicenter study of exemestane versus tamoxifen as first-line hormone therapy for postmenopausal women with metastatic breast cancer. Ann Oncol 2003 Sep; 14(9): 1391–8
US National Institutes of Health. Providing information regarding several clinical trials that are planned/ongoing/completed [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2008 Sep 25]
Jones SE, Cantrell J, Vukelja S, et al. Comparison of menopausal symptoms during the first year of adjuvant therapy with either exemestane or tamoxifen in early breast cancer: report of a Tamoxifen Exemestane Adjuvant Multicenter trial substudy. J Clin Oncol 2007 Oct 20; 25(30): 4765–71
Gil JM, Rubio-Terrés C, Del Castillo A, et al. Pharmacoeconomic analysis of adjuvant therapy with exemestane, anastrozole, letrozole or tamoxifen in postmenopausal women with operable and estrogen receptor-positive breast cancer. Clin Transl Oncol 2006 May; 8(5): 339–48
Lundkvist J, Wilking N, Holmberg S, et al. Cost-effectiveness of exemestane versus tamoxifen as adjuvant therapy for early-stage breast cancer after 2–3 years treatment with tamoxifen in Sweden. Breast Cancer Res Treat 2007 May; 102(3): 289–99
Skedgel C, Rayson D, Dewar R, et al. Cost-utility of adjuvant hormone therapies for breast cancer in postmenopausal women: sequential tamoxifen-exemestane and upfront anastrozole. Breast Cancer Res Treat 2007 Mar; 101(3): 325–33
Skedgel C, Rayson D, Dewar R, et al. Cost-utility of adjuvant hormone therapies with aromatase inhibitors in post-menopausal women with breast cancer: upfront anastrozole, sequential tamoxifen-exemestane and extended tamoxifen-letrozole. Breast 2007 Jun; 16(3): 252–61
Thompson D, Taylor DC, Montoya EL, et al. Cost-effectiveness of switching to exemestane after 2 to 3 years of therapy with tamoxifen in postmenopausal women with early-stage breast cancer. Value Health 2007 Sep–Oct; 10(5): 367–76
Coombes RC, Hall E, Snowdon CF, et al. The Intergroup Exemestane Study: a randomized trial in postmenopausal patients with early breast cancer who remain disease-free after two to three years of tamoxifen — updated survival analysis [abstract no. 3]. Breast Cancer Res Treat 2004; 88 Suppl. 1: 7
Howell A, Cuzick J, Baum M, et al. Results of the ATAC (Arimidex, Tamoxifen, Alone or in Combination) trial after completion of 5 years’ adjuvant treatment for breast cancer. Lancet 2005 Jan; 365(9453): 60–2
Baum M, Buzdar AU, Cuzick J, et al. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: first results of the ATAC randomised trial [published erratum appears in Lancet 2002; 360 (9344): 152]. Lancet 2002; 359(9324): 2131–9
Baum M, Buzdar A, Cuzick J, et al. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early-stage breast cancer: results of the ATAC (Arimidex, Tamoxifen Alone or in Combination) trial efficacy and safety update analyses. Cancer 2003 Nov; 98(9): 1802–10
Goss PE, Ingle JN, Martino S, et al. A randomized trial of letrozole in postmenopausal women after five years of tamoxifen therapy for early-stage breast cancer. N Engl J Med 2003 Nov 6; 349(19): 1793–802
Goss PE, Ingle JN, Martino S, et al. Randomized trial of letrozole following tamoxifen as extended adjuvant therapy in receptor-positive breast cancer: updated findings from NCIC CTG MA. 17. J Natl Cancer Inst 2005 Sep; 97(17): 1262–71
Gould RE, Garcia AA. Update on aromatase inhibitors in breast cancer. Curr Opin Obstet Gynecol 2006 Feb; 18(1): 41–6
Gradishar WJ. Tamoxifen: what next? Oncologist 2004; 9(4): 378–84
National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: breast cancer 2009 [online]. Available from URL: http://misc.medscape.com/images/586/406/Breast-Medscape.pdf [Accessed 2009 Apr 28]
Thorne C. Management of arthralgias associated with aromatase inhibitor therapy. Curr Oncol 2007 Dec; 14 Suppl. 1: S11–9
Buzdar AU, Coombes RC, Goss PE, et al. Summary of aromatase inhibitor clinical trials in postmenopausal women with early breast cancer. Cancer 2008 Dec; 112 Suppl. 3: 700–9
Wheler J, Johnson M, Seidman A. Adjuvant therapy with aromatase inhibitors for postmenopausal women with early breast cancer: evidence and ongoing controversy. Semin Oncol 2006 Dec; 33(6): 672–80
Pestalozzi B, Castiglione M. Primary breast cancer: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol 2008 May; 19 Suppl. 2: ii7–10
Goldhirsch A, Wood WC, Gelber RD, et al. Progress and promise: highlights of the international expert consensus on the primary therapy of early breast cancer 2007. Ann Oncol 2007 Jul; 18(7): 1133–44
Sanford M, Plosker GL. Anastrozole: a review of its use in postmenopausal women with early-stage breast cancer. Drugs 2008; 68(9): 1319–40
AstraZeneca UK Ltd. Arimidex® 1 mg film-coated tablets: summary of product characteristics [online]. Available from URL: http://www.emc.medicines.org.uk [Accessed 2009 Apr 28]
Mouridsen HT, Giobbie-Hurder A, Mauriac L, et al. BIG 1-98: a randomized double-blind phase III study evaluating letrozole and tamoxifen given in sequence as adjuvant endocrine therapy for postmenopausal women with receptor-positive breast cancer [abstract no. 13]. Cancer Res 69 (2 Suppl.): 66s
Novartis Pharmaceuticals UK Ltd. Femara® 2.5mg tablets: summary of product characteristics [online]. Available from URL: http://www.emc.medicines.org.uk [Accessed 2009 Apr 28]
AstraZeneca Pharmaceuticals LP. Arimidex® (anastrozole) tablets: US prescribing information [online]. Available from URL: http://www.fda.gov/cder/foi/label/2008/020541s020s021s023lbl.pdf [Accessed 2009 Mar 13]
Novartis Pharmaceuticals Corporation. Femara® (letrozole) 2.5mg tablets: US prescribing information [online]. Available from URL: http://www.fda.gov/cder/foi/label/2007/020726s014lbl.pdf [Accessed 2008 Sep 22]
Kataja V, Catiglione M. Locally recurrent or metastatic breast cancer: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol 2008 May; 19 Suppl. 2: ii11–3
National Breast and Ovarian Cancer Centre. Recommendations for use of endocrine therapy for the treatment of hormone receptor-positive advanced breast cancer [online]. Available from URL: http://www.nbocc.org.au/bestpractice/resources/ETAG_recommendationsforus.pdf [Accessed 2008 Sep 25]
Goss PE, Strasser K. Tamoxifen resistant and refractory breast cancer. The value of aromatase inhibitors. Drugs 2002; 62(6): 957–66
Lønning PE. Is there a growing role for endocrine therapy in the treatment of breast cancer? Drugs 2000 Jul; 60(1): 11–21
Carlini P, Bria E, Giannarelli D, et al. New aromatase inhibitors as second-line endocrine therapy in postmenopausal patients with metastatic breast carcinoma: a pooled analysis of the randomized trials. Cancer 2005 Oct 1; 104(7): 1335–42
AstraZeneca UK Ltd. Faslodex: summary of product characteristics [online]. Available from URL: http://www.emea.europa.eu/humandocs/PDFs/EPAR/faslodex/H-540-PI-en.pdf [Accessed 2009 Mar 13]
AstraZeneca Pharmaceuticals LP. Faslodex (fulvestrant) injection: US prescribing information [online]. Available from URL: http://www.fda.gov/cder/foi/label/2005/021344s004lbl.pdf [Accessed 2008 Sep 26]
Perez EA. Safety profiles of tamoxifen and the aromatase inhibitors in adjuvant therapy of hormone-responsive early breast cancer. Ann Oncol 2007; 18 Suppl. 8: viii26–35
Conte P, Frassoldati A. Aromatase inhibitors in the adjuvant treatment of postmenopausal women with early breast cancer: putting safety issues into perspective. Breast J 2007 Jan–Feb; 13(1): 28–35
Brufsky A, Harker WG, Beck JT, et al. Zoledronic acid inhibits adjuvant letrozole-induced bone loss in postmenopausal women with early breast cancer. J Clin Oncol 2007 Mar 1; 25(7): 829–36
Lønning PE. Exemestane for breast cancer prevention: a feasible strategy? Clin Cancer Res 2005 Jan 15; 11 (2 Pt 2): 918–24s
Author information
Authors and Affiliations
Corresponding author
Additional information
Various sections of the manuscript reviewed by: A.U. Buzdar, Department of Breast Medical Oncology, M. D. Anderson Cancer Center, University of Texas, Houston, Texas, USA; P. Carlini, Department of Medical Oncology, Regina Elena National Cancer Institute, Rome, Italy; S. Gonnelli, Department of Internal Medicine, University of Siena, Siena, Italy; P.E. Lønning, Institute of Medicine, University of Bergen, and Department of Oncology, Haukeland University Hospital, Bergen, Norway.
Data Selection
Sources: Medical literature published in any language since 1980 on ‘exemestane’, identified using MEDLINE and EMBASE, supplemented by AdisBase (a proprietary database of Wolters Kluwer Health | Adis). Additional references were identified from the reference lists of published articles. Bibliographical information, including contributory unpublished data, was also requested from the company developing the drug.
Search strategy: MEDLINE, EMBASE and AdisBase search terms were ‘exemestane’ and (‘early breast cancer’ or ‘early-stage breast cancer’ or ‘advanced breast cancer’). Searches were last updated 21 April 2009.
Selection: Studies in patients with breast cancer who received exemestane. Inclusion of studies was based mainly on the methods section of the trials. When available, large, well controlled trials with appropriate statistical methodology were preferred. Relevant pharmacodynamic and pharmacokinetic data are also included.
Index terms: Exemestane, aromatase inhibitors, breast cancer, postmenopausal, pharmacodynamics, pharmacokinetics, therapeutic use, tolerability, pharmacoeconomics.
Rights and permissions
About this article
Cite this article
Deeks, E.D., Scott, L.J. Exemestane. Drugs 69, 889–918 (2009). https://doi.org/10.2165/00003495-200969070-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200969070-00007