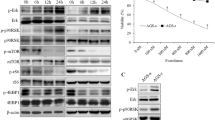
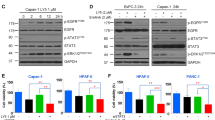
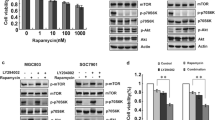
Abstract
Dysregulated crosstalk between different signaling pathways contributes to tumor development, including resistance to cancer therapy. In the present study, we found that the mitogen-activated extracellular signal-regulated kinase (MEK) inhibitor trametinib failed to suppress the proliferation of PANC-1 and MGC803 cells by activating the Janus kinase 2 (JAK2)/signal transducer and activator of transcription 3 (STAT3) signaling pathway, while the JAK2 inhibitor fedratinib failed to inhibit the growth of the PANC-1 cells upon stimulation of extracellular signal-regulated kinase (ERK) signaling. In particular, the most prominent enhancement of the anti-proliferative effect resulted from the concurrent blockage of the JAK2/STAT3 and ERK signaling pathways. Furthermore, the combination of the two inhibitors resulted in a reduced tumor burden in mice. Our evidence suggests novel crosstalk between JAK2/STAT3 and ERK signaling in gastric cancer (GC) and pancreatic ductal adenocarcinoma (PDAC) cells and provides a therapeutic strategy to overcome potential resistance in gastrointestinal cancer.
Abstract
目的
探索在胃肠道肿瘤细胞中的JAK2/STAT3和MEK/ERK信号通路之间的交互作用, 明确两者相互作用在肿瘤细胞增殖和凋亡的作用。
创新点
发现JAK2/STAT3和ERK信号在胃癌 (GC) 和胰腺导管腺癌 (PDAC) 细胞内的相互作用, 为克服胃肠道肿瘤的化疗耐药提供新的治疗策略。
方法
在GC和PDAC细胞系中, 应用MEK抑制剂——曲美替尼 (Trametinib) 和JAK2抑制剂——费德拉替尼 (Fedratinib) 单独或共同抑制MEK和JAK2/STAT3信号通路, 利用CCK-8和蛋白质印迹 (western blot) 方法检测细胞增殖和凋亡, 及相关信号通路的激活情况; 同时利用荷瘤小鼠检测JAK2/STAT3和MEK信号通路的抑制对肿瘤生长的影响。
结论
本研究发现, MEK抑制剂——曲美替尼在抑制MEK信号通路的同时却对JAK2/STAT3信号通路有较强的激活作用, 无法有效抑制胃癌细胞和胰腺癌细胞的增殖; 而JAK2抑制剂——费德拉替尼虽可以有效抑制JAK/STAT3信号通路的激活, 但显著促进ERK信号通路的异常活化, 导致对胃癌细胞和胰腺癌细胞的生长抑制作用失效。而当JAK2/STAT3和ERK信号通路同时被抑制后, 抗增殖效果显著增强, 且该作用效应与促进肿瘤细胞的凋亡进程密切相关。此外, 这两种抑制剂的结合有效阻止了荷瘤小鼠的肿瘤生长, 有很好的抗肿瘤效果, 避免了化疗耐药的发生。
Similar content being viewed by others
References
Anderson JL, Titz B, Akiyama R, et al., 2014. Phosphoproteomic profiling reveals IL6-mediated paracrine signaling within the Ewing sarcoma family of tumors. Mol Cancer Res, 12(12):1740–1754. https://doi.org/10.1158/1541-7786.MCR-14-0159
Andersson E, Kuusanmäki H, Bortoluzzi S, et al., 2016. Activating somatic mutations outside the SH2-domain of STAT3 in LGL leukemia. Leukemia, 30(5):1204–1208. https://doi.org/10.1038/leu.2015.263
Ashrafizadeh M, Zarrabi A, Orouei S, et al., 2020. STAT3 pathway in gastric cancer: signaling, therapeutic targeting and future prospects. Biology (Basel), 9(6): 126. https://doi.org/10.3390/biology9060126
Blair HA, 2019. Fedratinib: first approval. Drugs, 79: 1719–1725 https://doi.org/10.1007/s40265-019-01205-x
Bose P, Verstovsek S, 2017. JAK2 inhibitors for myeloproliferative neoplasms: what is next? Blood, 130(2): 115–125. https://doi.org/10.1182/blood-2017-04-742288
Chatterjee N, Bivona TG, 2019. Polytherapy and targeted cancer drug resistance. Trends Cancer, 5(3): 170–182. https://doi.org/10.1016/j.trecan.2019.02.003
Chen WB, Wu JH, Shi H, et al., 2014. Hepatic stellate cell coculture enables sorafenib resistance in Huh7 cells through HGF/c-Met/Akt and Jak2/Stat3 pathways. BioMed Res Int, 2014:764981. https://doi.org/10.1155/2014/764981
Chen YC, Chien LH, Huang BM, et al., 2016. Aqueous extracts of Toona sinensis leaves inhibit renal carcinoma cell growth and migration through JAK2/stat3, Akt, MEK/ERK, and mTOR/HIF-2α pathways. Nutr Cancer, 68(4):654–666. https://doi.org/10.1080/01635581.2016.1158292
Colmegna B, Morosi L, D’Incalci M, 2018. Molecular and pharmacological mechanisms of drug resistance: an evolving paradigm. Handb Exp Pharmacol, 249:1–12. https://doi.org/10.1007/164_2017_20
Deer EL, González-Hernández J, Coursen JD, et al., 2010. Phenotype and genotype of pancreatic cancer cell lines. Pancreas, 39(4):425–435. https://doi.org/10.1097/MPA.0b013e3181c15963
Degirmenci U, Wang M, Hu JC, 2020. Targeting aberrant RAS/RAF/MEK/ERK signaling for cancer therapy. Cells, 9(1):198. https://doi.org/10.3390/cells9010198
Ezerskyte M, Paredes JA, Malvezzi S, et al., 2018. O6-methylguanine-induced transcriptional mutagenesis reduces p53 tumor-suppressor function. Proc Natl Acad Sci USA, 115(18):4731–4736. https://doi.org/10.1073/pnas.1721764115
Gerber DE, 2008. Targeted therapies: a new generation of cancer treatments. Am Fam Physician, 77(3):311–319.
Grimaldi AM, Simeone E, Festino L, et al., 2017. MEK inhibitors in the treatment of metastatic melanoma and solid tumors. Am J Clin Dermatol, 18(6):745–754. https://doi.org/10.1007/s40257-017-0292-y
Groner B, von Manstein V, 2017. Jak Stat signaling and cancer: opportunities, benefits and side effects of targeted inhibition. Mol Cell Endocrinol, 451:1–14. https://doi.org/10.1016/j.mce.2017.05.033
Guo YJ, Pan WW, Liu SB, et al., 2020. ERK/MAPK signalling pathway and tumorigenesis (Review). Exp Ther Med, 19(3):1997–2007. https://doi.org/10.3892/etm.2020.8454
Hirata E, Sahai E, 2017. Tumor microenvironment and differential responses to therapy. Cold Spring Harb Perspect Med, 7(7):a026781. https://doi.org/10.1101/cshperspect.a026781
Holohan C, van Schaeybroeck S, Longley DB, et al., 2013. Cancer drug resistance: an evolving paradigm. Nat Rev Cancer, 13(10):714–726. https://doi.org/10.1038/nrc3599
Hua F, Li K, Shang S, et al., 2019. Immune signaling and autophagy regulation. Adv Exp Med Biol, 1206:551–593. https://doi.org/10.1007/978-981-15-0602-4_26
Liu QQ, Zeng XL, Guan YL, et al., 2020. Verticillin A inhibits colon cancer cell migration and invasion by targeting c-Met. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 21(10):779–795. https://doi.org/10.1631/jzus.B2000190
Meierjohann S, 2017. Crosstalk signaling in targeted melanoma therapy. Cancer Metastasis Rev, 36(1):23–33. https://doi.org/10.1007/s10555-017-9659-z
Meyer SC, 2017. Mechanisms of resistance to JAK2 inhibitors in myeloproliferative neoplasms. Hematol Oncol Clin North Am, 31(4):627–642. https://doi.org/10.1016/j.hoc.2017.04.003
Mizukami T, Togashi Y, Sogabe S, et al., 2015. EGFR and HER2 signals play a salvage role in MEK1-mutated gastric cancer after MEK inhibition. Int J Oncol, 47(2):499–505. https://doi.org/10.3892/ijo.2015.3050
Mokhtari RB, Homayouni TS, Baluch N, et al., 2017. Combination therapy in combating cancer. Oncotarget, 8(23): 38022–38043. https://doi.org/10.18632/oncotarget.16723
Nie YZ, Wu KC, Yu J, et al., 2017. A global burden of gastric cancer: the major impact of China. Expert Rev Gastroenterol Hepatol, 11(7):651–661. https://doi.org/10.1080/17474124.2017.1312342
Pardanani A, Harrison C, Cortes JE, et al., 2015. Safety and efficacy of fedratinib in patients with primary or secondary myelofibrosis: a randomized clinical trial. JAMA Oncol, 1(5):643–651. https://doi.org/10.1001/jamaoncol.2015.1590
Pencik J, Pham HTT, Schmoellerl J, et al., 2016. JAK-STAT signaling in cancer: from cytokines to non-coding genome. Cytokine, 87:26–36. https://doi.org/10.1016/j.cyto.2016.06.017
Pourhoseingholi MA, Vahedi M, Baghestani AR, 2015. Burden of gastrointestinal cancer in Asia; an overview. Gastroenterol Hepatol Bed Bench, 8(1):19–27.
Ramamonjisoa N, Ackerstaff E, 2017. Characterization of the tumor microenvironment and tumor-stroma interaction by non-invasive preclinical imaging. Front Oncol, 7:3. https://doi.org/10.3389/fonc.2017.00003
Samatar AA, Poulikakos PI, 2014. Targeting RAS-ERK signalling in cancer: promises and challenges. Nat Rev Drug Discov, 13(12):928–942. https://doi.org/10.1038/nrd4281
Schizas D, Charalampakis N, Kole C, et al., 2020. Immunotherapy for pancreatic cancer: a 2020 update. Cancer Treat Rev, 86:102016. https://doi.org/10.1016/j.ctrv.2020.102016
Thambi P, Sausville EA, 2002. STI571 (imatinib mesylate): the tale of a targeted therapy. Anticancer Drugs, 13(2):111–114. https://doi.org/10.1097/00001813-200202000-00001
Verstovsek S, Gotlib J, Mesa RA, et al., 2017. Long-term survival in patients treated with ruxolitinib for myelofibrosis: COMFORT-I and -II pooled analyses. J Hematol Oncol, 10:156. https://doi.org/10.1186/s13045-017-0527-7
Vert G, Chory J, 2011. Crosstalk in cellular signaling: background noise or the real thing?. Dev Cell, 21(6):985–991. https://doi.org/10.1016/j.devcel.2011.11.006
Wang LH, Wu CF, Rajasekaran N, et al., 2018. Loss of tumor suppressor gene function in human cancer: an overview. Cell Physiol Biochem, 51(6):2647–2693. https://doi.org/10.1159/000495956
Wu QY, Ma MM, Fu L, et al., 2018. Roles of germline JAK2 activation mutation JAK2 V625F in the pathology of myeloproliferative neoplasms. Int J Biol Macromol, 116: 1064–1073. https://doi.org/10.1016/j.ijbiomac.2018.05.120
Wu QY, Ma MM, Zhang S, et al., 2019. Disruption of R867 and Y613 interaction plays key roles in JAK2 R867Q mutation caused acute leukemia. Int J Biol Macromol, 136:209–219. https://doi.org/10.1016/j.ijbiomac.2019.06.068
Xin P, Xu XY, Deng CJ, et al., 2020. The role of JAK/STAT signaling pathway and its inhibitors in diseases. Int Immunopharmacol, 80:106210. https://doi.org/10.1016/j.intimp.2020.106210
Zhang JY, Tian XJ, Xing JH, 2016. Signal transduction pathways of EMT induced by TGF-β, SHH, and WNT and their crosstalks. J Clin Med, 5(4):41. https://doi.org/10.3390/jcm5040041
Zhang Q, Zhang Y, Diamond S, et al., 2016. The Janus kinase 2 inhibitor fedratinib inhibits thiamine uptake: a putative mechanism for the onset of Wernicke’s encephalopathy. Drug Metabo Dispos, 42(10):1656–1662. https://doi.org/10.1124/dmd.114.058883
Acknowledgments
This study was supported by the Zhejiang Provincial Natural Science Foundation of China (Nos. LQ18H280005 and LY21H030002), the National Natural Science Foundation of China (Nos. 81770535, 81600595, 81503297, 81603340, 81773945, and 81803775), the Medical Health Science and Technology Project of Zhejiang Provincial Health Commission (Nos. 2019RC228, 2019RC229, and 2019RC113), and the Open Foundation from Chinese and Western Integrative Medicine in the Most Important Subjects of Zhejiang (No. ZXYJH2018002), China.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Author contributions
Xi WANG, Chunyan DAI, Yifei YIN, and Yufei FU performed the experimental research and data analysis. Xi WANG, Chunyan DAI, and Zhe CHEN wrote and edited the manuscript. Lin WU and Weiyang JIN performed the establishment of animal models. Ke HAO and Bin LU contributed to the study design, data analysis, writing and editing of the manuscript. All authors have read and approved the final manuscript and, therefore, have full access to all the data in the study and take responsibility for the integrity and security of the data.
Compliance with ethics guidelines
Xi WANG, Chunyan DAI, Yifei YIN, Lin WU, Weiyang JIN, Yufei FU, Zhe CHEN, Ke HAO, and Bin LU declare that they have no conflict of interest.
All institutional and national guidelines for the care and use of laboratory animals were followed. Approval for the animal experiments was obtained from the Experimental Animal Committee of the Provincial Government of Zhejiang (China), and the animal protocols fulfilled the institutional guidelines on the Protection of Animals used for Scientific or Educational Purposes.
Supplementary information
Tables S1–S6; Fig. S1
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, X., Dai, C., Yin, Y. et al. Blocking the JAK2/STAT3 and ERK pathways suppresses the proliferation of gastrointestinal cancers by inducing apoptosis. J. Zhejiang Univ. Sci. B 22, 492–503 (2021). https://doi.org/10.1631/jzus.B2000842
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B2000842