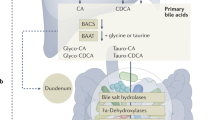
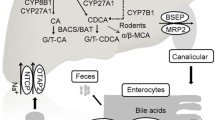
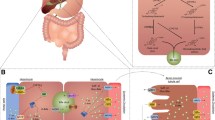
Abstract
Over the past decade, there has been increasing attention on the interaction between microbiota and bile acid metabolism. Bile acids are not only involved in the metabolism of nutrients, but are also important in signal transduction for the regulation of host physiological activities. Microbial-regulated bile acid metabolism has been proven to affect many diseases, but there have not been many studies of disease regulation by microbial receptor signaling pathways. This review considers findings of recent research on the core roles of farnesoid X receptor (FXR), G protein-coupled bile acid receptor (TGR5), and vitamin D receptor (VDR) signaling pathways in microbial–host interactions in health and disease. Studying the relationship between these pathways can help us understand the pathogenesis of human diseases, and lead to new solutions for their treatments.
概要
近十年来, 微生物与胆汁酸代谢的相互作用越来越受到关注。 胆汁酸不仅参与营养物质的代谢, 而且在调节宿主生理活动的信号转导中也起着重要作用。 已有研究表明, 微生物调控的胆汁酸代谢对许多疾病都有显著的影响, 但对微生物受体信号通路调控疾病的相关研究并不多。 本文综述了近年来有关法尼醇受体 (FXR)、 G 蛋白偶联胆汁酸受体 (TGR5) 和维生素 D 受体 (VDR) 信号通路在健康和疾病的微生物-宿主相互作用中的核心作用。 研究这些信号通路之间的关系, 有助于我们了解人类疾病的发病机制, 为人类疾病的治疗提供新的解决方案。
Similar content being viewed by others
References
Alexeev EE, Lanis JM, Kao DJ, et al., 2018. Microbiota-derived indole metabolites promote human and murine intestinal homeostasis through regulation of interleukin-10 receptor. Am J Pathol, 188(5):1183–1194. https://doi.org/10.1016/j.ajpath.2018.01.011
Alnouti Y, 2009. Bile acid sulfation: a pathway of bile acid elimination and detoxification. Toxicol Sci, 108(2):225–246. https://doi.org/10.1093/toxsci/kfn268
Ananthanarayanan M, Balasubramanian N, Makishima M, et al., 2001. Human bile salt export pump promoter is transactivated by the farnesoid X receptor/bile acid receptor. J Biol Chem, 276(31):28857–28865. https://doi.org/10.1074/jbc.M011610200
Baggio LL, Drucker DJ, 2007. Biology of incretins: GLP-1 and GIP. Gastroenterology, 132(6):2131–2157. https://doi.org/10.1053/j.gastro.2007.03.054
Begley M, Gahan CGM, Hill C, 2005. The interaction between bacteria and bile. FEMS Microbiol Rev, 29(4):625–651. https://doi.org/10.1016/j.femsre.2004.09.003
Bustos AY, de Valdez GF, Fadda S, et al., 2018. New insights into bacterial bile resistance mechanisms: the role of bile salt hydrolase and its impact on human health. Food Res Int, 112:250–262. https://doi.org/10.1016/j.foodres.2018.06.035
Cariello M, Piccinin E, Garcia-Irigoyen O, et al., 2017. Nuclear receptor FXR, bile acids and liver damage: introducing the progressive familial intrahepatic cholestasis with FXR mutations. Biochim Biophys Acta Mol Basis Dis, 1864(4): 1308–1318. https://doi.org/10.1016/j.bbadis.2017.09.019
Cariou B, van Harmelen K, Duran-Sandoval D, et al., 2006. The farnesoid X receptor modulates adiposity and peripheral insulin sensitivity in mice. J Biol Chem, 281(16):11039–11049. https://doi.org/10.1074/jbc.M510258200
Carr RM, Reid AE, 2015. FXR agonists as therapeutic agents for non-alcoholic fatty liver disease. Curr Atheroscler Rep, 17(4):16. https://doi.org/10.1007/s11883-015-0500-2
Cheng J, Fang ZZ, Kim JH, et al., 2014. Intestinal CYP3A4 protects against lithocholic acid-induced hepatotoxicity in intestine-specific VDR-deficient mice. J Lipid Res, 55(3): 455–465. https://doi.org/10.1194/jlr.M044420
Degirolamo C, Modica S, Palasciano G, et al., 2011. Bile acids and colon cancer: solving the puzzle with nuclear receptors. Trends Mol Med, 17(10):564–572. https://doi.org/10.1016/j.molmed.2011.05.010
Degirolamo C, Rainaldi S, Bovenga F, et al., 2014. Microbiota modification with probiotics induces hepatic bile acid synthesis via downregulation of the FXR-FGF15 axis in mice. Cell Rep, 7(1):12–18. https://doi.org/10.1016/j.celrep.2014.02.032
Devkota S, Wang YW, Musch MW, et al., 2012. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in IL10-/- mice. Nature, 487(7405):104–108. https://doi.org/10.1038/nature11225
Duran-Sandoval D, Cariou B, Percevault F, et al., 2005. The farnesoid X receptor modulates hepatic carbohydrate metabolism during the fasting-refeeding transition. J Biol Chem, 280(33):29971–29979. https://doi.org/10.1074/jbc.M501931200
Enright EF, Joyce SA, Gahan CG, et al., 2017. Impact of gut microbiota-mediated bile acid metabolism on the solubilization capacity of bile salt micelles and drug solubility. Mol Pharm, 14(4):1251–1263. https://doi.org/10.1021/acs.molpharmaceut.6b01155
Espinosa A, Dardalhon V, Brauner S, et al., 2009. Loss of the lupus autoantigen Ro52/Trim21 induces tissue inflammation and systemic autoimmunity by disregulating the IL-23-Th17 pathway. J Exp Med, 206(8):1661–1671. https://doi.org/10.1084/jem.20090585
Gadaleta RM, Oldenburg B, Willemsen EC, et al., 2011. Activation of bile salt nuclear receptor FXR is repressed by pro-inflammatory cytokines activating NF-κB signaling in the intestine. Biochim Biophys Acta, 1812(8):851–858. https://doi.org/10.1016/j.bbadis.2011.04.005
Giorgetti G, Brandimarte G, Fabiocchi F, et al., 2015. Interactions between innate immunity, microbiota, and probiotics. J Immunol Res, 2015:501361. https://doi.org/10.1155/2015/501361
Gonzalez FJ, Jiang CT, Patterson AD, 2016. An intestinal microbiota-farnesoid X receptor axis modulates metabolic disease. Gastroenterology, 151(5):845–859. https://doi.org/10.1053/j.gastro.2016.08.057
Hofmann AF, 1999. The continuing importance of bile acids in liver and intestinal disease. Arch Intern Med, 159(22): 2647–2658. https://doi.org/10.1001/archinte.159.22.2647
Hofmann AF, 2004. Detoxification of lithocholic acid, a toxic bile acid: relevance to drug hepatotoxicity. Drug Metab Rev, 36(3-4):703–722. https://doi.org/10.1081/DMR-200033475.
Hunt MC, Yang YZ, Eggertsen G, et al., 2000. The peroxisome proliferator-activated receptor α (PPARα) regulates bile acid biosynthesis. J Biol Chem, 275(37):28947–28953. https://doi.org/10.1074/jbc.M002782200
Inagaki T, Choi M, Moschetta A, et al., 2005. Fibroblast growth factor 15 functions as an enterohepatic signal to regulate bile acid homeostasis. Cell Metab, 2(4):217–225. https://doi.org/10.1016/j.cmet.2005.09.001
Inagaki T, Moschetta A, Lee YK, et al., 2006. Regulation of antibacterial defense in the small intestine by the nuclear bile acid receptor. Proc Natl Acad Sci USA, 103(10): 3920–3925. https://doi.org/10.1073/pnas.0509592103
Islam KBMS, Fukiya S, Hagio M, et al., 2011. Bile acid is a host factor that regulates the composition of the cecal microbiota in rats. Gastroenterology, 141(5):1773–1781. https://doi.org/10.1053/j.gastro.2011.07.046
Jiang CT, Xie C, Li F, et al., 2015. Intestinal farnesoid X receptor signaling promotes nonalcoholic fatty liver disease. J Clin Invest, 125(1):386–402. https://doi.org/10.1172/JCI76738
Jin DP, Wu SP, Zhang YG, et al., 2015. Lack of vitamin D receptor causes dysbiosis and changes the functions of the murine intestinal microbiome. Clin Ther, 37(5):996–1009. https://doi.org/10.1016/j.clinthera.2015.04.004
Jin LH, Fang ZP, Fan MJ, et al., 2019. Bile-ology: from bench to bedside. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 20(5):414–427. https://doi.org/10.1631/jzus.B1900158
Joyce SA, MacSharry J, Casey PG, et al., 2014. Regulation of host weight gain and lipid metabolism by bacterial bile acid modification in the gut. Proc Natl Acad Sci USA, 111(20):7421–7426. https://doi.org/10.1073/pnas.1323599111
Kakiyama G, Pandak WM, Gillevet PM, et al., 2013. Modulation of the fecal bile acid profile by gut microbiota in cirrhosis. J Hepatol, 58(5):949–955. https://doi.org/10.1016/j.jhep.2013.01.003
Kakiyama G, Hylemon PB, Zhou HP, et al., 2014. Colonic inflammation and secondary bile acids in alcoholic cirrhosis. Am J Physiol Gastrointest Liver Physiol, 306(11): G929–G937. https://doi.org/10.1152/ajpgi.00315.2013
Katsuma S, Hirasawa A, Tsujimoto G, 2005. Bile acids promote glucagon-like peptide-1 secretion through TGR5 in a murine enteroendocrine cell line STC-1. Biochem Biophys Res Commun, 329(1):386–390. https://doi.org/10.1016/j.bbrc.2005.01.139
Keitel V, Donner M, Winandy S, et al., 2008. Expression and function of the bile acid receptor TGR5 in Kupffer cells. Biochem Biophys Res Commun, 372(1):78–84. https://doi.org/10.1016/j.bbrc.2008.04.171
Kundu S, Kumar S, Bajaj A, 2015. Cross-talk between bile acids and gastrointestinal tract for progression and development of cancer and its therapeutic implications. IUBMB Life, 67(7):514–523. https://doi.org/10.1002/iub.1399
Laverdure R, Mezouari A, Carson MA, et al., 2018. A role for methanogens and methane in the regulation of GLP-1. Endocrinol Diabetes Metab, 1(1):e00006. https://doi.org/10.1002/edm2.6
Lee WJ, Hase K, 2014. Gut microbiota-generated metabolites in animal health and disease. Nat Chem Biol, 10(6): 416–424. https://doi.org/10.1038/nchembio.1535
Lee YS, Shin S, Shigihara T, et al., 2007. Glucagon-like peptide-1 gene therapy in obese diabetic mice results in long-term cure of diabetes by improving insulin sensitivity and reducing hepatic gluconeogenesis. Diabetes, 56(6): 1671–1679. https://doi.org/10.2337/db06-1182
Li TG, Chiang JY, 2014. Bile acid signaling in metabolic disease and drug therapy. Pharmacol Rev, 66(4):948–983. https://doi.org/10.1124/pr.113.008201
Li-Hawkins J, Gåfvels M, Olin M, et al., 2002. Cholic acid mediates negative feedback regulation of bile acid synthesis in mice. J Clin Invest, 110(8):1191–1200. https://doi.org/10.1172/JCI16309
Liu Q, Shao WT, Zhang CL, et al., 2017. Organochloride pesticides modulated gut microbiota and influenced bile acid metabolism in mice. Environ Pollut, 226:268–276. https://doi.org/10.1016/j.envpol.2017.03.068
Lu TT, Makishima M, Repa JJ, et al., 2000. Molecular basis for feedback regulation of bile acid synthesis by nuclear receptors. Mol Cell, 6(3):507–515. https://doi.org/10.1016/S1097-2765(00)00050-2
Ma YJ, Huang YX, Yan LN, et al., 2013. Synthetic FXR agonist GW4064 prevents diet-induced hepatic steatosis and insulin resistance. Pharm Res, 30(5):1447–1457. https://doi.org/10.1007/s11095-013-0986-7
Masubuchi N, Sugihara M, Sugita T, et al., 2016. Oxidative stress markers, secondary bile acids and sulfated bile acids classify the clinical liver injury type: promising diagnostic biomarkers for cholestasis. Chem Biol Interact, 255:83–91. https://doi.org/10.1016/j.cbi.2015.08.016
Matsubara T, Li F, Gonzalez FJ, 2013. FXR signaling in the enterohepatic system. Mol Cell Endocrinol, 368(1-2):17–29. https://doi.org/10.1016/j.mce.2012.05.004
Modica S, Murzilli S, Salvatore L, et al., 2008. Nuclear bile acid receptor FXR protects against intestinal tumorigenesis. Cancer Res, 68(23):9589–9594. https://doi.org/10.1158/0008-5472.CAN-08-1791
Mullish BH, Pechlivanis A, Barker GF, et al., 2018. Functional microbiomics: evaluation of gut microbiota-bile acid metabolism interactions in health and disease. Methods, 149:49–58. https://doi.org/10.1016/j.ymeth.2018.04.028
Neish AS, 2009. Microbes in gastrointestinal health and disease. Gastroenterology, 136(1):65–80. https://doi.org/10.1053/j.gastro.2008.10.080
Nie YF, Jun HU, Yan XH, 2015. Cross-talk between bile acids and intestinal microbiota in host metabolism and health. J Zhejiang Univ-Sci B (Biomed & Biotechnol), 16(6):436–446. https://doi.org/10.1631/jzus.B1400327
Out C, Patankar JV, Doktorova M, et al., 2015. Gut microbiota inhibit Asbt-dependent intestinal bile acid reabsorption via Gata4. J Hepatol, 63(3):697–704. https://doi.org/10.1016/j.jhep.2015.04.030
Pabst O, 2012. New concepts in the generation and functions of IgA. Nat Rev Immunol, 12(12):821–832. https://doi.org/10.1038/nri3322
Pathak P, Xie C, Nichols RG, et al., 2018. Intestine farnesoid X receptor agonist and the gut microbiota activate G-protein bile acid receptor-1 signaling to improve metabolism. Hepatology, 68(4):1574–1588. https://doi.org/10.1002/hep.29857
Peng L, Piekos S, Guo GL, et al., 2016. Role of farnesoid X receptor in establishment of ontogeny of phase-I drug metabolizing enzyme genes in mouse liver. Acta Pharm Sin B, 6(5):453–459. https://doi.org/10.1016/j.apsb.2016.07.015
Pols TWH, Nomura M, Harach T, et al., 2011. TGR5 activation inhibits atherosclerosis by reducing macrophage inflammation and lipid loading. Cell Metab, 14(6):747–757. https://doi.org/10.1016/j.cmet.2011.11.006
Reijnders D, Goossens GH, Hermes GDA, et al., 2016. Effects of gut microbiota manipulation by antibiotics on host metabolism in obese humans: a randomized double-blind placebo-controlled trial. Cell Metab, 24(1):63–74. https://doi.org/10.1016/j.cmet.2016.06.016
Ridlon JM, Kang DJ, Hylemon PB, 2006. Bile salt biotransformations by human intestinal bacteria. J Lipid Res, 47(2):241–259. https://doi.org/10.1194/jlr.R500013-JLR200
Russell DW, 2003. The enzymes, regulation, and genetics of bile acid synthesis. Annu Rev Biochem, 72:137–174. https://doi.org/10.1146/annurev.biochem.72.121801.161712
Sayin SI, Wahlström A, Felin J, et al., 2013. Gut microbiota regulates bile acid metabolism by reducing the levels of tauro-beta-muricholic acid, a naturally occurring FXR antagonist. Cell Metab, 17(2):225–235. https://doi.org/10.1016/j.cmet.2013.01.003
Shehata MG, el Sohaimy SA, El-Sahn MA, et al., 2016. Screening of isolated potential probiotic lactic acid bacteria for cholesterol lowering property and bile salt hydrolase activity. Ann Agric Sci, 61(1):65–75. https://doi.org/10.1016/j.aoas.2016.03.001
Sinclair P, Brennan DJ, le Roux CW, 2018. Gut adaptation after metabolic surgery and its influences on the brain, liver and cancer. Nat Rev Gastroenterol Hepatol, 15(10): 606–624. https://doi.org/10.1038/s41575-018-0057-y
Sonnenburg JL, Bäckhed F, 2016. Diet-microbiota interactions as moderators of human metabolism. Nature, 535(7610): 56–64. https://doi.org/10.1038/nature18846
Sun J, 2016. VDR/vitamin D receptor regulates autophagic activity through ATG16L1. Autophagy, 12(6):1057–1058. https://doi.org/10.1080/15548627.2015.1072670
Sun J, 2017. The role of vitamin D and vitamin D receptors in colon cancer. Clin Transl Gastroenterol, 8(6):e103. https://doi.org/10.1038/ctg.2017.31
Sung J, Kim S, Cabatbat JJT, et al., 2017. Global metabolic interaction network of the human gut microbiota for context-specific community-scale analysis. Nat Commun, 8:15393. https://doi.org/10.1038/ncomms15393
Swann JR, Want EJ, Geier FM, et al., 2011. Systemic gut microbial modulation of bile acid metabolism in host tissue compartments. Proc Natl Acad Sci USA, 108(Suppl 1): 4523–4530. https://doi.org/10.1073/pnas.1006734107
Thaiss CA, Zmora N, Levy M, et al., 2016. The microbiome and innate immunity. Nature, 535(7610):65–74. https://doi.org/10.1038/nature18847
Theriot CM, Bowman AA, Young VB, 2016. Antibiotic-induced alterations of the gut microbiota alter secondary bile acid production and allow for Clostridium difficile spore germination and outgrowth in the large intestine. mSphere, 1(1):e00045-15. https://doi.org/10.1128/mSphere.00045-15
Tremaroli V, Bäckhed F, 2012. Functional interactions between the gut microbiota and host metabolism. Nature, 489(7415): 242–249. https://doi.org/10.1038/nature11552
Vijayvargiya P, Camilleri M, Carlson P, et al., 2017. Performance characteristics of serum C4 and FGF19 measurements to exclude the diagnosis of bile acid diarrhoea in IBS-diarrhoea and functional diarrhoea. Aliment Pharmacol Ther, 46(6):581–588. https://doi.org/10.1111/apt.14214
Vítek L, Haluzík M, 2016. The role of bile acids in metabolic regulation. J Endocrinol, 228(3):R85–R96. https://doi.org/10.1530/JOE-15-0469
Vogtmann E, Goedert JJ, 2016. Epidemiologic studies of the human microbiome and cancer. Br J Cancer, 114(3): 237–242. https://doi.org/10.1038/bjc.2015.465
Vrieze A, van Nood E, Holleman F, et al., 2012. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology, 143(4):913–916. https://doi.org/10.1053/j.gastro.2012.06.031
Vrieze A, Out C, Fuentes S, et al., 2014. Impact of oral vancomycin on gut microbiota, bile acid metabolism, and insulin sensitivity. J Hepatol, 60(4):824–831. https://doi.org/10.1016/j.jhep.2013.11.034
Wada K, Tanaka H, Maeda K, et al., 2009. Vitamin D receptor expression is associated with colon cancer in ulcerative colitis. Oncol Rep, 22(5):1021–1025. https://doi.org/10.3892/or_00000530
Wang J, Thingholm LB, Skiecevičienė J, et al., 2016. Genome-wide association analysis identifies variation in vitamin D receptor and other host factors influencing the gut microbiota. Nat Genet, 48(11):1396–1406. https://doi.org/10.1038/ng.3695
Wang YD, Chen WD, Wang MH, et al., 2008. Farnesoid X receptor antagonizes nuclear factor κB in hepatic inflammatory response. Hepatology, 48(5):1632–1643. https://doi.org/10.1002/hep.22519
Wang YD, Chen WD, Yu D, et al., 2011. The G-protein-coupled bile acid receptor, Gpbar1 (TGR5), negatively regulates hepatic inflammatory response through antagonizing nuclear factor kappa light-chain enhancer of activated B cells (NF-κB) in mice. Hepatology, 54(4):1421–1432. https://doi.org/10.1002/hep.24525
Watanabe M, Houten SM, Mataki C, et al., 2006. Bile acids induce energy expenditure by promoting intracellular thyroid hormone activation. Nature, 439(7075):484–489. https://doi.org/10.1038/nature04330
Wolfe A, Thomas A, Edwards G, et al., 2011. Increased activation of the Wnt/β-catenin pathway in spontaneous hepatocellular carcinoma observed in farnesoid X receptor knockout mice. J Pharmacol Exp Ther, 338(1):12–21. https://doi.org/10.1124/jpet.111.179390
Woodhouse CA, Patel VC, Singanayagam A, et al., 2018. Review article: the gut microbiome as a therapeutic target in the pathogenesis and treatment of chronic liver disease. Aliment Pharmacol Ther, 47(2):192–202. https://doi.org/10.1111/apt.14397
Wu SP, Liao AP, Xia YL, et al., 2010. Vitamin D receptor negatively regulates bacterial-stimulated NF-κB activity in intestine. Am J Pathol, 177(2):686–697. https://doi.org/10.2353/ajpath.2010.090998
Xie C, Jiang CT, Shi JM, et al., 2017. An intestinal farnesoid X receptor-ceramide signaling axis modulates hepatic gluconeogenesis in mice. Diabetes, 66(3):613–626. https://doi.org/10.2337/db16-0663
Yamada S, Takashina Y, Watanabe M, et al., 2018. Bile acid metabolism regulated by the gut microbiota promotes non-alcoholic steatohepatitis-associated hepatocellular carcinoma in mice. Oncotarget, 9(11):9925–9939. https://doi.org/10.18632/oncotarget.24066
Yamagata K, Daitoku H, Shimamoto Y, et al., 2004. Bile acids regulate gluconeogenic gene expression via small heterodimer partner-mediated repression of hepatocyte nuclear factor 4 and Foxo1. J Biol Chem, 279(22):23158–23165. https://doi.org/10.1074/jbc.M314322200
Yang T, Owen JL, Lightfoot YL, et al., 2013. Microbiota impact on the epigenetic regulation of colorectal cancer. Trends Mol Med, 19(12):714–725. https://doi.org/10.1016/j.molmed.2013.08.005
Zhang YQ, Lee FY, Barrera G, et al., 2006. Activation of the nuclear receptor FXR improves hyperglycemia and hyperlipidemia in diabetic mice. Proc Natl Acad Sci USA, 103(4):1006–1011. https://doi.org/10.1073/pnas.0506982103
Zimmer J, Lange B, Frick JS, et al., 2012. A vegan or vegetarian diet substantially alters the human colonic faecal microbiota. Eur J Clin Nutr, 66(1):53–60. https://doi.org/10.1038/ejcn.2011.141
Author information
Authors and Affiliations
Contributions
Er-teng JIA and Qin-yu GE designed the research. Er-teng JIA wrote the manuscript. Zhi-yu LIU, Min PAN, Jia-feng LU, and Qin-yu GE provided guidance on the writ¬ing of this review. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Er-teng JIA, Zhi-yu LIU, Min PAN, Jia-feng LU, and Qin-yu GE declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Project supported by the National Key Research and Development Program of China (No. 2016YFA0501602) and the National Natural Science Foundation of China (Nos. 61801108 and 81801478)
Rights and permissions
About this article
Cite this article
Jia, Et., Liu, Zy., Pan, M. et al. Regulation of bile acid metabolism-related signaling pathways by gut microbiota in diseases. J. Zhejiang Univ. Sci. B 20, 781–792 (2019). https://doi.org/10.1631/jzus.B1900073
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1900073