Abstract
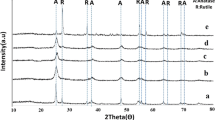
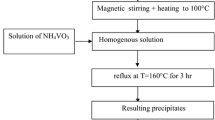
Titanium oxide (TiO2) nanoparticles (NPs) were doped with vanadium using a novel, facile, and inexpensive method. The TiO2 NPs were dispersed in a vanadyl oxalate solution prepared by dissolving vanadium pentoxide (V2O5) in oxalic acid. A short heat treatment at 400 °C applied to the dried mixture resulted in the doping of TiO2 with a net measured decrease of its band gap by about 0.5 eV, making this important semiconductor material usable in the visible light spectrum.




Similar content being viewed by others
References
A. Fujishima and K. Honda: Electrochemical photolysis of water at a semiconductor electrode. Nature 238, 37–38 (1972).
J.C.S. Wu and C.H. Chen: A visible-light response vanadium-doped titania nanocatalyst by sol-gel method. J. Photochem. Photobiol. A: Chem. 163, 509–515 (2004).
S. Klosek and D. Raftery: Visible light driven V-doped TiO2 photocatalyst and its photooxidation of ethanol. J. Phys. Chem. B 105, 2815–2819 (2001).
W. Zhou, Q. Liu, Z. Zhu, and J. Zhang: Preparation and properties of vanadium-doped TiO2 photocatalysts. J. Phys. D: Appl. Phys. 43, 035301 (2010).
J.J. Sene, W.A. Zeltner, and M.A. Anderson: Fundamental photoelectrocatalytic and electrophoretic mobility studies of TiO2 and V-doped TiO2 thinfilm electrode materials. J. Phys. Chem. B 107, 1597–1603 (2003).
D.E. Gu, B.C. Yang, and Y.D. Hu: V and N co-doped nanocrystal anatase TiO2 photocatalysts with enhanced photocatalytic activity under visible light irradiation. J. Catal. Commun. 9, 1472–1476 (2008).
B. Ohtani, O.O. Prieto-Mahaney, D. Li, and R. Abe: What is Degussa (Evonik) P25- crystalline composition analysis, reconstruction from isolated pure particles and photocatalytic activity test. J. Photochem. Photobiol. A: Chem. 216, 179–182 (2010).
D.W. McComb: Bonding and electronic structure in zirconia pseudopolymorphs investigated by electron energy-loss spectroscopy. Phys. Rev. B 54, 7094–7120 (1996).
A. Pan, J.G. Zhang, Z. Nie, G. Cao, B.W. Arey, G. Li, S. Liang, and J. Liu: Facile synthesized nanorod structured vanadium pentoxide for high-rate lithium batteries. J. Mater. Chem. 20, 9193–9199 (2010).
D.W. Fischer: X-ray band spectra and molecular-orbital structure of rutile TiO2. Phys. Rev. B 5, 4219–4226 (1972).
J. Pascual, J. Camassel, and H. Mathieu: Fine structure in the intrinsic absorption edge of TiO2. Phys. Rev. B 18, 5605–5614 (1978).
R. Sanjines, H. Tang, H. Berger, F. Gozzo, G. Margaritondo, and F. Levy: Electronic structure of anatase TiO2 oxide. J. Appl. Phys. 75, 2945–2951 (1994).
Acknowledgments
The partial support from the NSEC Center for Hierarchical Manufacturing at the University of Massachusetts (National Science Foundation award no. 1025020) is gratefully acknowledged. We also acknowledge the NSF for its support (award no. 0701525) to the Nanoscopy Facility, an electron microscopy facility at UPR.
Author information
Authors and Affiliations
Corresponding author
Supplementary materials
Supplementary materials
For supplementary material for this article, please visit http:// dx.doi.org/10.1557/mrc.2014.15
Rights and permissions
About this article
Cite this article
Ahmadi, M., Guinel, M.J.F. Doping of TiO2 nanopowders with vanadium for the reduction of its band gap reaching the visible light spectrum region. MRS Communications 4, 73–76 (2014). https://doi.org/10.1557/mrc.2014.15
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/mrc.2014.15