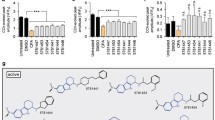
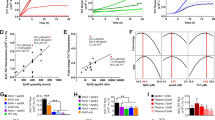
Abstract
The 5′ untranslated region (5′UTR) of the transcript encoding the Alzheimer’s amyloid precursor protein (APP) is a key regulatory sequence that determines the amount of intracellular APP holoprotein present in brain derived cells. Using neuroblastoma cells (SY5Y) we developed a transfection based screen of a library of FDA drugs to identify compounds that limited APP luciferase reporter expression translated from the APP 5′UTR. Paroxetine (Paxil™), dimercaptopropanol, phenserine, desferrioxamine, tetrathiolmobdylate, and azithromycin were six leads that were subsequently found to also suppress APP holoprotein levels or to alter APP cleavage (azithromycin). Since APP holoprotein levels are proportionate to Aβ peptide output in many systems we tested the efficacy of paroxetine and dimercaptopropanol to limit Aβ secretion as measured by ELISA assays. Paroxetine and dimercaptopropanol limited Aβ peptide secretion from lens epithelial cells (B3 cells). Interestingly, paroxetine changed the steady-state levels of transferrin receptor mRNAs. These data suggested that this serotonin reuptake inhibitor (SSRI) provided extra pharmacological action to chelate interacellular iron or change the intracellular iron distribution. An altered iron distribution would be predicted to indirectly limit APP holoprotein expression and Aβ peptide secretion.
Similar content being viewed by others

References
Arjona A., Pooler A., Lee R., and Wurtman R. (2002) Effect of a 5-HT(2C) serotonin agonist, dexnorfenfluramine, on amyloid precursor protein metabolism in guinea pigs. Brain Res. 951, 135.
Chishti M. A., Yang D. S., Janus C., Phinney A. L., Horne P., Pearson J., et al. (2001) Early-onset amyloid deposition and cognitive deficits in transgenic mice expressing a double mutant form of amyloid precursor protein 695. J. Biol. Chem. 276, 21562–21570.
Crapper McLachlan D. R., Dalton A. J., Kruck T. P. A., Bell M. Y., Smith W. L., Kalow W., and Andrews D. F. (1991). Intramuscular desferrioxamine in patients with Alzheimer’s disease. Lancet 337, 1304–1308.
Feng Y., Zhang F., Lokey L. K., Chastain J. L., Lakkis L., Eberhart D., and Warren S. T. (1995) Translational suppression by trinucleotide repeat expansion at FMR1. Science 268, 731–734.
Khan W. A., Sahaa D., Rahmana A., Salama M., Bogaertsa J., and Bennish M. (2002) Comparison of single-dose azithromycin and 12-dose, 3-day erythromycin for childhood cholera: a randomized, double-blind trial. Lancet 360(9374), 1722–1727.
Klausner R., Rouault T. A., and Harford J. B. (1993) Regulating the fate of mRNA: the control of cellular iron metabolism. Cell 72, 19–28.
Lesne S., Docagne F., Gabriel C., Liot G., Lahiri D. K., Buee L., et al. (2003) Transforming growth factor-beta 1 potentiates amyloid-beta generation in astrocytes and in transgenic mice. J. Biol. Chem. 278, 18408–18418.
Lim G. P., Yang F., Chu T., Chen P., Beech W., Teter B., et al. (2000) Ibuprofen suppresses plaque pathology and inflammation in a mouse model for Alzheimer’s disease. J. Neurosci. 20, 5709–5714.
Ly B. T., Williams S. R., and Clark R. F. (2002) Mercuric oxide poisoning treated with whole-bowel irrigation and chelation therapy. Ann Emerg. Med. 39(3), 312–315.
Mandinov L., Mandinova A., Kyurkchiev S., Kyurkchiev D., Kehayov I., Kolev V., et al. (2003) Copper chelation represses the vascular response to injury. Proc. Natl. Acad. Sci. U. S. A. 100, 6700–6705.
Moechars D. G. M., Kuiperi C., Laenen I., and Van Leuven F. (1998) Aggressive behaviour in transgenic mice expressing APP is alleviated by serotonergic drugs. Neuroreport 9, 3561–3564.
Nilsson L., Rogers J., and Potter H. (1998) The essential role of inflammation and induced gene expression in the pathogenic pathway of Alzheimer’s disease. Front. Biosci. 3, 436–446.
Payton S., Cahill C., Randall J., Gullans S., and Rogers J. (2003) Alzheimer’s disease drug discovery targeted to the APP mRNA 5′ untranslated region; paroxetine and dimercaptopropanol are drug hits. J. Mol. Neurosci., in press.
Refolo L. M., Pappolla M. A., LaFrancois J., Malester B., Schmidt S. D., Thomas-Bryant T., et al. (2001) A cholesteol-lowering drug reduces beta-amyloid pathology in a transgenic mouse model of Alzheimer’s disease. Neurobiol Dis. 8, 890–899.
Rogers J. T., Leiter L. M., McPhee J., Cahill C. M., Zhan S. S., Potter H., and Nilsson L. N. (1999) Translation of the Alzheimer amyloid precursor protein mRNA is upregulated by interleukin-1 through 5′-untranslated region sequences. J. Biol. Chem. 274, 6421–6431.
Rogers J. T., Randall J., Cahill C. M., Eder P. S., Huang X., Gunshin H., et al. (2002a) An iron-responsive element type II in the 5′ untranslated region of the Alzheimer’s amyloid precursor protein transcript. J. Biol. Chem. 277, 45518–45528.
Rogers J. T., Randall J., Eder P. S., Huang X., Bush A. I., Tanzi R. E., et al. (2002b) Alzheimer’s disease drug discovery targeted to the APP mRNA 5′ untranslated region. J. Mol. Neurosci. 19, 77–82.
Shaw K. T., Utsuki T., Rogers J., Yu Q. S., Sambamurti K., Brossi A., et al. (2001) Phenserine regulates translation of beta-amyloid precursor protein mRNA by a putative interleukin-1 responsive element, a target for drug development. Proc. Natl. Acad. Sci. U. S. A. 98, 7605–7610.
Svitkin Y. V., Pause A., Haghighat A., Pyronnet S., Witherell G., Belsham G. J., and Sonenberg N. (2001) The requirement for eukaryotic initiation factor 4A (elF4A) in translation is in direct proportion to the degree of mRNA5′ secondary structure. RNA 7, 382–394.
Weggen S., Eriksen J. L., Das P., Sagi S. A., Wang R., Pietrzik C. U., et al. (2001) A subset of NSAIDs lower amyloidogenic Abeta-42 independently of cyclooxygenase activity. Nature 414, 212–216.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morse, L.J., Payton, S.M., Cuny, G.D. et al. FDA-preapproved drugs targeted to the translational regulation and processing of the amyloid precursor protein. J Mol Neurosci 24, 129–136 (2004). https://doi.org/10.1385/JMN:24:1:129
Issue Date:
DOI: https://doi.org/10.1385/JMN:24:1:129