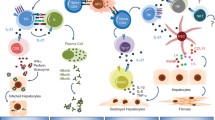
Abstract
Although primary infection with human cytomegalovirus (HCMV), a β-herpesvirus, is widespread and acquired early in life, it rarely causes disease in immune-competent individuals. However, in immune-compromised patients HCMV infection or reactivation invariably leads to serious disease, the effective treatment of which remains a difficult clinical problem. Current antiviral therapy is limited not only by toxicity but also by the continual emergence of drug-resistant viruses. The limitations of these current therapeutics provides a strong impetus to develop novel approaches that will enhance the host's immune responsiveness while at the same time effectively controlling virus replication. Type I interferon (IFN) plays a critical role in initiating innate antiviral defenses and promoting adaptive responses and lymphotoxin (LT)-αβ has recently been identified as an essential effector cytokine regulating the induction of type I IFN during CMV infection. In particular, CMV infection of immune-compromised mice has revealed the immunotherapeutic potential of the lymphotoxin-beta receptor (LTβR) signaling pathway to restore immune function and provide protection from CMV mortality. In this review, we discuss the potential benefits and risks associated with LTβR-directed immunotherapy for CMV disease and other persistent viral infections.
Article PDF
Similar content being viewed by others
References
Locksley RM, Killeen N and Lenardo MJ: The TNF and TNF receptor superfamilies: Integrating mammalian biology. Cell 2001; 104:487–501.
Bodmer JL, Schneider P, Tschopp J: The molecular architecture of the TNF superfamily. Trends Biochem Sci 2002; 27:19–26.
De Togni P, Goellner J, Ruddle NH, et al: Abnormal development of peripheral lymphoid organs in mice deficient in lymphotoxin. Science 1994; 264:703–706.
Ware CF: Network communications: lymphotoxins, LIGHT, and TNF: Annu Rev Immunol 2005; 23:787–819.
Mauri DN, Ebner R, Montgomery RI et al: LIGHT, a new member of the TNF superfamily and lymphotoxin a are ligands for herpesvirus entry mediator. Immunity 1998; 8:21–30.
Sedy JR, Gavrieli M, Potter KG, et al: B and T lymphocyte attenuator regulates T cell activation through interaction with herpesvirus entry mediator. Nat Immunol 2005; 6:90–98.
Cheung TC, Humphreys IR, Potter KG, et al: Evolutionarily divergent herpesviruses modulate T cell activation by targeting the herpesvirus entry mediator cosignaling pathway. Proc Natl Acad Sci USA 2005; 102: 13218–13223.
Ansel KM, Ngo VN, Hyman PL, et al: A chemokine-driven positive feedback loop organizes lymphoid follicles. Nature 2000; 406:309–314.
Luther SA, Bidgol A, Hargreaves DC, et al: Differing activities of homeostatic chemokines CCL19, CCL21, and CXCL12 in lymphocyte and dendritic cell recruitment and lymphoid neogenesis. J Immunol 2002; 169:424–433.
Murphy M, Walter BN, Pike-Nobile L, et al: Expression of the lymphotoxin β-receptor on follicular stromal cells in human lymphoid tissue. Cell Death Differentiation 1998; 5:497–505.
Stopfer P, Obermeier F, Dunger N, et al: Blocking lymphotoxin-beta receptor activation diminishes inflammation via reduced mucosal addressin cell adhesion molecule-1 (MAdCAM-1) expression and leucocyte margination in chronic DSS-induced colitis. Clin Exp Immunol 2004; 136:21–29.
Cohavy O, Zhou J, Granger SW, Ware CF, Targan SR: LIGHT expression by mucosal T cells may regulate IFN-gamma expression in the intestine. J Immunol 2004; 173:251–258.
Morel Y, Schiano de Colella JM, Harrop J, et al: Reciprocal expression of the TNF family receptor herpes virus entry mediator and its ligand LIGHT on activated T cells: LIGHT down-regulates its own receptor. J Immunol 2000; 165:4397–4404.
Kwon BS, Tan KB, Ni J, et al: A newly identified member of the tumor necrosis factor receptor superfamily with a wide tissue distribution and involvement in lymphocyte activation. J Biol Chem 1997; 272:14272–14276.
Wu Q, Sun Y, Wang J, et al: Signal via lymphotoxin-beta R on bone marrow stromal cells is required for an early checkpoint of NK cell development. J Immunol 2001; 166:1684–1689.
Elewaut D, Brossay L, Santee SM, et al: Membrane lymphotoxin is required early in ontogeny for development of different subpopulations of NK T cells. J Immunol 2000; 165:671–679.
Wu Q, Wang Y, Wang J, Hedgeman EO, Browning JL, Fu Y-X: The requirement of membrane lymphotoxin for the presence of dendritic cells in lymphoid tissue. J Exp Med 1999; 190:629–638.
Abe K, Yarovinsky FO, Murakami T, et al: Distinct contributions of TNF and LT cytokines to the development of dendritic cells in vitro and their recruitment in vivo. Blood 2003; 101:1477–1483.
Dejardin E, Droin NM, Delhase M, et al: The lymphotoxin-beta receptor induces different patterns of gene expression via two NF-kappaB pathways. Immunity 2002; 17:525–535.
Anders RA, Subudhi SK, Wang J, Pfeffer K, Fu YX: Contribution of the lymphotoxin beta receptor to liver regeneration. J Immunol 2005; 175:1295–1300.
Benedict CA, Banks TA, Ware CF: Death and survival: viral regulation of TNF signaling pathways. Curr Opin Immunol 2003; 15:59–65.
Spahn TW, Eugster HP, Fontana A, Domschke W, Kucharzik T: Role of lymphotoxin in experimental models of infectious diseases: potential benefits and risks of a therapeutic inhibition of the lymphotoxin-beta receptor pathway. Infect Immun 2005; 73:7077–7088.
Benedict CA, Banks TA, Senderowicz L, et al: Lymphotoxins and cytomegalovirus cooperatively induce interferon-β, establishing host-virus détente. Immunity 2001; 15:617–626.
Banks TA, Rickert S, Benedict CA, et al: A lymphotoxin-IFN-beta axis essential for lymphocyte survival revealed during cytomegalovirus infection. J Immunol 2005; 174:7217–7225.
Williams-Abbott L, Walter BN, Cheung T, Goh CR, Porter AG, Ware CF: The lymphotoxin-α (LTα) subunit is essential for the assembly, but not receptor specificity, of the membrane-anchored LTα1β2 heterotrimeric ligand. J Biol Chem 1997; 272: 19451–19456.
Rooney IA, Butrovich KD, Glass AA, et al: The lymphotoxin-beta receptor is necessary and sufficient for LIGHT-mediated apoptosis of tumor cells. J Biol Chem 2000; 275:14307–14315.
Singh N: Cytomegalovirus infection in solid organ transplant recipients: new challenges and their implications for preventive strategies. J Clin Virol 2006; 35:474–477.
Vancikova Z, Dvorak P: Cytomegalovirus infection in immunocompetent and immunocompromised individuals—a review. Curr Drug Targets Immune Endocr Metabol Disord 2001; 1:179–187.
Grahame-Clarke C: Human cytomegalovirus, endothelial function and atherosclerosis. Herpes 2005; 12:42–45.
Revello MG, Gerna G: Pathogenesis and prenatal diagnosis of human cytomegalovirus infection. J Clin Virol 2004; 29:71–83.
Gilbert C, Boivin G: Human cytomegalovirus resistance to antiviral drugs. Antimicrob Agents Chemother 2005; 49:873–883.
Baldanti F, Lurain N, Gerna G: Clinical and biologic aspects of human cytomegalovirus resistance to antiviral drugs. Hum Immunol 2004; 65:403–409.
Anderson KP, Fox MC, Brown-Driver V, Martin MJ, Azad RF: Inhibition of human cytomegalovirus immediate-early gene expression by an antisense oligonucleotide complementary to immediate-early RNA. Antimicrob Agents Chemother 1996; 40:2004–2011.
Schleiss MR, Heineman TC: Progress toward an elusive goal: current status of cytomegalovirus vaccines. Expert Rev Vaccines 2005; 4:381–406.
Hakki M, Riddell SR, Storek J, et al: Immune reconstitution to cytomegalovirus after allogeneic hematopoietic stem cell transplantation: impact of host factors, drug therapy, and subclinical reactivation. Blood 2003; 102:3060–3067.
Fries BC, Riddell SR, Kim HW, et al: Cytomegalovirus disease before hematopoietic cell transplantation as a risk for complications after transplantation. Biol Blood Marrow Transplant 2005; 11:136–148.
Tartour E, Mathiot C, Fridman WH: Current status of interleukin-2 therapy in cancer. Biomed Pharmacother 1992; 46:473–484.
de Vries EG, Timmer T, Mulder NH, et al: Modulation of death receptor pathways in oncology. Drugs Today (Barc) 2003; 39 Suppl C:95–109.
Santoro A, Canova C: Anemia and erythropoietin treatment in chronic kidney diseases. Minerva Urol Nefrol 2005; 57:23–31.
Lyman GH: Guidelines of the National Comprehensive Cancer Network on the use of myeloid growth factors with cancer chemotherapy: a review of the evidence. J Natl Compr Canc Netw 2005; 3:557–571.
Courtney LP, Phelps JL, Karavodin LM: An anti-IL-2 antibody increases serum half-life and improves antitumor efficacy of human recombinant interleukin-2. Immunopharmacology 1994; 28:223–232.
Bonnet D, Schmaltz AA, Feltes TF: Infection by the respiratory syncytial virus in infants and young children at high risk. Cardiol Young 2005; 15:256–265.
Knight DM, Trinh H, Le J, et al: Construction and initial characterization of a mouse-human chimeric anti-TNF antibody. Mol Immunol 1993; 30:1443–1453.
Mohler KM, Torrance DS, Smith CA, et al: Soluble tumor necrosis factor (TNF) receptors are effective therapeutic agents in lethal endotoxemia and function simultaneously as both TNF carriers and TNF antagonists. J Immunol 1993; 151:1548–1561.
Sarawar SR, Lee BJ, Reiter SK, Schoenberger SP: Stimulation via CD40 can substitute for CD4 T cell function in preventing reactivation of a latent herpesvirus. Proc Natl Acad Sci USA 2001; 98:6325–6329.
Browning JL, Miatkowski K, Sizing I, et al: Signalling through the lymphotoxin-b receptor induces the death of some adenocarcinoma tumor lines. J Exp Med 1996; 183:867–878.
Soriano V, Martin-Carbonero L, Maida I, Garcia-Samaniego J, Nunez M: New paradigms in the management of HIV and hepatitis C virus coinfection. Curr Opin Infect Dis 2005; 18:550–560.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Banks, T.A., Rickert, S. & Ware, C.F. Restoring immune defenses via lymphotoxin signaling. Immunol Res 34, 243–253 (2006). https://doi.org/10.1385/IR:34:3:243
Issue Date:
DOI: https://doi.org/10.1385/IR:34:3:243