Article contents
Adsorption of Cr(VI) and As(V) on Chitosan-Montmorillonite: Selectivity and pH Dependence
Published online by Cambridge University Press: 01 January 2024
Abstract
Montmorillonite modified with the cationic biopolymer, chitosan, has, in weak acidic solutions, protonated amine groups which act as anion-adsorption sites. Due to the specific surroundings of the adsorption sites and diffusion paths in the interlayer of chitosan-montmorillonite, preferential adsorption of certain anions is likely. In the present study, the adsorption properties for the inorganic anions Cr(VI) and As(V) were determined, taking into account solution pH and competitive adsorption in the presence of Cl− and SO42−${\rm{SO}}_4^{2 - }$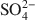 . Chitosan-montmorillonite was prepared by adding an amount of chitosan equivalent to 500% of the cation exchange capacity (CEC) at pH 5 and 75°C. The resulting anion exchange capacity was ∼0.34 molc/kg. The adsorption properties for As(V) and Cr(VI) were determined with the batch technique at pH 3 to 9. Adsorption isotherms were fitted to the Langmuir and Dubinin-Radushkevich equations and judged quantitatively by the correlation coefficient. To describe the competitive adsorption, the selectivity (S) was determined by the ratio of amounts of anions A and B adsorbed (qA/qB) in a binary system. The ionic species adsorbed, i.e. either Cr(VI) or As(V), depended on the pH, as did the degree of protonation of the amine groups, and this played a decisive role in the amount of anions adsorbed. The maximum amount of Cr(VI) adsorbed was 180 mmol/kg at pH 3.5, whereas for As(V) it was 120 mmol/kg at pH 4.0 to 5.0. The adsorption process of Cr(VI) and As(V) fit well to the Langmuir isotherm. By increasing the concentration of the competitive anion, Cl−, in solution, the amount of Cr(VI) and As(V) adsorbed remained almostconstant, whereas SO42−${\rm{SO}}_4^{2 - }$
. Chitosan-montmorillonite was prepared by adding an amount of chitosan equivalent to 500% of the cation exchange capacity (CEC) at pH 5 and 75°C. The resulting anion exchange capacity was ∼0.34 molc/kg. The adsorption properties for As(V) and Cr(VI) were determined with the batch technique at pH 3 to 9. Adsorption isotherms were fitted to the Langmuir and Dubinin-Radushkevich equations and judged quantitatively by the correlation coefficient. To describe the competitive adsorption, the selectivity (S) was determined by the ratio of amounts of anions A and B adsorbed (qA/qB) in a binary system. The ionic species adsorbed, i.e. either Cr(VI) or As(V), depended on the pH, as did the degree of protonation of the amine groups, and this played a decisive role in the amount of anions adsorbed. The maximum amount of Cr(VI) adsorbed was 180 mmol/kg at pH 3.5, whereas for As(V) it was 120 mmol/kg at pH 4.0 to 5.0. The adsorption process of Cr(VI) and As(V) fit well to the Langmuir isotherm. By increasing the concentration of the competitive anion, Cl−, in solution, the amount of Cr(VI) and As(V) adsorbed remained almostconstant, whereas SO42−${\rm{SO}}_4^{2 - }$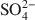 had a more pronounced competitive effect. At concentration ratios of 0.5 and 1 for SO42−${\rm{SO}}_4^{2 - }$
had a more pronounced competitive effect. At concentration ratios of 0.5 and 1 for SO42−${\rm{SO}}_4^{2 - }$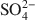 to Cr(VI) and As(V), respectively, the sorption capacity decreased by 10 and 25%, respectively. The sequence of the selectivity was: Cr(VI)>SO42−>As(V)>Cl−${\rm{Cr}}\left( {{\rm{VI}}} \right) > {\rm{SO}}_4^{2 - } > {\rm{As}}\left( {\rm{V}} \right) > {\rm{C}}{{\rm{l}}^ - }$
to Cr(VI) and As(V), respectively, the sorption capacity decreased by 10 and 25%, respectively. The sequence of the selectivity was: Cr(VI)>SO42−>As(V)>Cl−${\rm{Cr}}\left( {{\rm{VI}}} \right) > {\rm{SO}}_4^{2 - } > {\rm{As}}\left( {\rm{V}} \right) > {\rm{C}}{{\rm{l}}^ - }$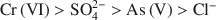 . Chitosan-montmorillonite showed a high selectivity for Cr(VI), which adsorbed chemically. Despite the lower affinity for As(V) and physical adsorption, the adsorption capacity was relatively high.
. Chitosan-montmorillonite showed a high selectivity for Cr(VI), which adsorbed chemically. Despite the lower affinity for As(V) and physical adsorption, the adsorption capacity was relatively high.
- Type
- Article
- Information
- Copyright
- Copyright © The Clay Minerals Society 2009
References
- 30
- Cited by




