Published online Nov 16, 2021. doi: 10.12998/wjcc.v9.i32.9731
Peer-review started: June 30, 2021
First decision: July 26, 2021
Revised: August 27, 2021
Accepted: September 24, 2021
Article in press: September 24, 2021
Published online: November 16, 2021
Acute respiratory distress syndrome (ARDS) is injury of alveolar epithelial cells and capillary endothelial cells caused by various factors, including endogenous and exogenous lung factors, leading to diffuse pulmonary interstitial and alveolar edema, and acute respiratory failure. ARDS involves alveolar epithelial cells and pulmonary interstitial capillary endothelial cells. Circulating endothelial cells (CECs) are the only marker that directly reflects vascular endothelial injury in vivo. There have been few studies on the correlation between peripheral blood CECs and ARDS at home and abroad. The lungs are the organs with the highest capillary density and the most endothelial cells, thus, it is speculated that when ARDS occurs, CECs are stimulated and damaged, and released into the circulatory system.
To explore the correlation between CEC level and severity of ARDS in patients postoperatively.
Blood samples were collected from all patients on day 2 (d2) and day 5 (d5) after surgery. The control group comprised 32 healthy volunteers. Number of CECs was measured by flow cytometry, and operation time was recorded. Changes in various indexes of patients were monitored, and diagnosis of ARDS was determined based on ARDS Berlin definition. We comprised d2 CECs in different groups, correlation between operation time and d2 CECs, ARDS of different severity by d2 CECs, and predictive value of d2 CECs for ARDS in postoperative patients.
The number of d2 CECs in the ARDS group was significantly higher than that in the healthy control group (P < 0.001). The number of d2 CECs in the ARDS group was significantly higher than that in the non-ARDS group (P < 0.001). The number of d2 CECs in the non-ARDS group was significantly higher than that in the healthy control group (P < 0.001). Operation time was positively correlated with number of CECs on d2 (rs = 0.302, P = 0.001). The number of d2 CECs in the deceased group was significantly higher than that in the improved group (P < 0.001). There was no significant difference in number of d2 CECs between patients with mild and moderate ARDS. The number of d2 CECs in patients with severe ARDS was significantly higher than that in patients with mild and moderate ARDS (P = 0.041, P = 0.037). There was no significant difference in number of d5 and d2 CECs in the non-ARDS group after admission to intensive care. The number of d5 CECs was higher than the number of d2 CECs in the ARDS improved group (P < 0.001). The number of d5 CECs was higher than the number of d2 CECs in the ARDS deceased group (P = 0.002). If the number of CECs was > 1351/mL, sensitivity and specificity of predicting ARDS were 80.8% and 78.1%, respectively.
Changes in number of CECs might predict occurrence and adverse outcome of ARDS after surgery, and higher numbers of CECs indicate worse prognosis of ARDS.
Core Tip: This manuscript evaluated the changes in number of circulating endothelial cells (CECs) might predict occurrence and adverse outcome of acute respiratory distress syndrome (ARDS) postoperatively, and higher numbers of CECs are associated with a worse prognosis of ARDS.
- Citation: Peng M, Yan QH, Gao Y, Zhang Z, Zhang Y, Wang YF, Wu HN. Correlation between circulating endothelial cell level and acute respiratory distress syndrome in postoperative patients. World J Clin Cases 2021; 9(32): 9731-9740
- URL: https://www.wjgnet.com/2307-8960/full/v9/i32/9731.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v9.i32.9731
Acute respiratory distress syndrome (ARDS) is injury of alveolar epithelial cells and capillary endothelial cells caused by various factors, including endogenous and exogenous lung factors, leading to diffuse pulmonary interstitial and alveolar edema, and acute respiratory failure[1-3]. The clinical manifestations are progressive respiratory distress and refractory hypoxemia, and the imaging manifestations are heterogeneous exudative changes[4]. The incidence rate of ARDS is 25%-50% when caused by severe infection, 11%-25% when caused by multiple trauma, and the incidence rate can reach 40% when caused by massive blood transfusion. The incidence rate of ARDS may increase further when two or more risk factors are present[5]. The longer the exposure to risk factors, the higher the incidence rate of ARDS. Studies have shown that when exposure to risk factors persisted for 24, 48 and 72 h, the incidence of ARDS was 76%, 85% and 93%, respectively. The incidence rate of ARDS is high, treatment is difficult, and ARDS has a long course and poor prognosis. Many studies have tried to identify the best marker to predict development and prognosis of ARDS at an early stage[6,7]. However, predictive ARDS markers are still in the research stage[3].
ARDS involves alveolar epithelial cells and pulmonary interstitial capillary endothelial cells. Current research on the markers related to capillary endothelial cells is mainly focused on angiotensin-2, selectin, von Willebrand factor antigen, intercellular adhesion molecule-1, vascular endothelial growth factor, insulin-like growth factor binding protein-3, apolipoprotein A1, S-100, PPFIA1 gene and other aspects[8], although the sensitivity and specificity of these markers are low. Compared with above indicators, Eizawa et al[9] have considered that circulating endothelial cells (CECs) are the only marker that directly reflects vascular endothelial injury in vivo. There have been few studies on the correlation between peripheral blood CECs and ARDS at home and abroad. The lungs are the organs with the highest capillary density and the most endothelial cells, thus, it is speculated that when ARDS occurs, CECs are stimulated and damaged, and released into the circulatory system.
The present study aimed to investigate the correlation between number of CECs and incidence of ARDS, and to explore whether CECs can be used as a predictive biomarker for incidence and adverse clinical outcomes of ARDS.
During 2012–2014, 125 surgical patients were admitted postoperatively to Tianjin Medical University General Hospital. Blood samples were collected on day 2 (d2) and day 5 (d5) after surgery.
Inclusion criteria were: (1) patients after general anesthesia; and (2) operation time ≥ 4 h. Exclusion criteria were: (1) thoracic surgery, including thoracotomy, thoracoscopic surgery and chest wall mass operation; (2) vascular diseases, such as arteriosclerosis (including hypertensive arteriosclerosis and atherosclerosis), systemic vasculitis, and arteriovenous thrombosis; (3) patients with malignant tumors; (4) patients with chronic organ dysfunction, such as chronic obstructive pulmonary disease, chronic liver failure, or chronic renal insufficiency; (5) head trauma; and (6) non-ARDS related death.
The clinical data of the enrolled patients are shown in Table 1. There were 32 healthy people in the control group, including 19 men and 13 women, with an average age of 53 ± 11 years (range: 33–67 years).
| Clinical data | n (%) |
| Gender | |
| Male | 75 (60) |
| Female | 50 (40) |
| Age | |
| Age distribution | 33-67 |
| Average | 53 ± 11 |
| Mechanical ventilation | 105 (84) |
| ARDS occurrence | 52 (42) |
| Improvement | 39 (31) |
| Death | 13 (10) |
| Average APACHE II score | 11 ± 5 |
| Department type | |
| General surgery | 77 (62) |
| Orthopedics | 25 (20) |
| Other | 23 (18) |
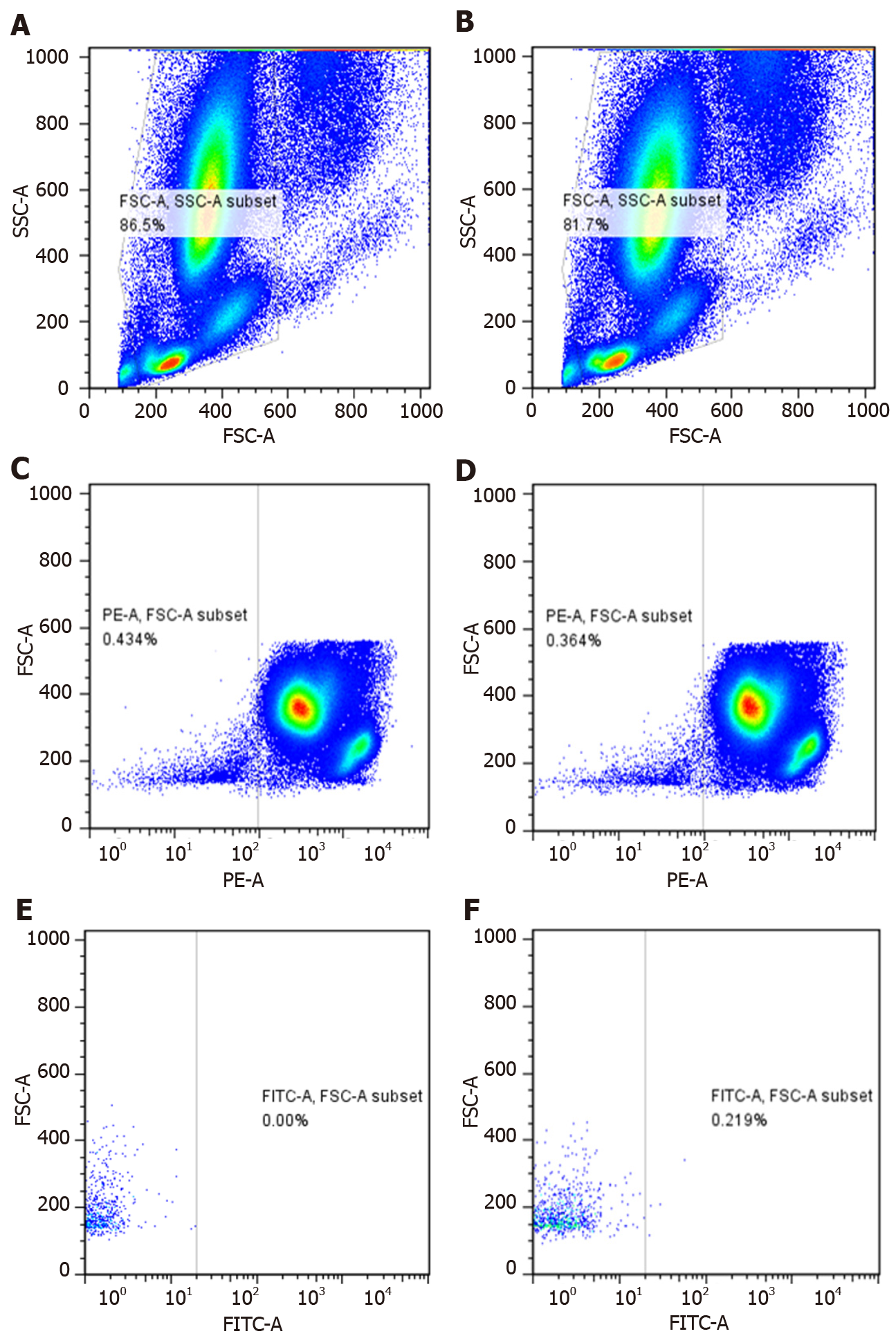
We selected CD146+ CD45--labeled CECs, FITC Mouse anti-Human CD146 (Cat No: 560846; BD Biosciences, Franklin Lakes, NJ, United States), FITC Mouse IgG1, κ Isotype Control (Cat No: 554679; BD Biosciences), PE Mouse anti-Human CD45 (Cat No: 555483; BD Biosciences), PE Mouse IgG1, κ Isotype Control (Cat No: 555489; BD Biosciences). We counted 500000 nucleated cells in each blood sample. The samples were detected by flow cytometry with 488 nm excitation wavelength. The results were analyzed by Flow Cytometry CellQuest software. The percentage of CECs among nuclear cells was calculated.
We collected 5 mL peripheral blood from the median cubital vein of enrolled patients. Two EDTA anticoagulant tubes were used to collect blood samples, with 3 mL for the first tube and 2 mL for the second tube, and placed in ice. The blood (200 mL) was taken from the second tube for detection. We added 4 mL red blood cell lysate; incubated at room temperature for 20 min; centrifuged at 1500 rpm at room temperature for 5 min; discarded supernatant and washed with once or twice with PBS; resuspended the blood cells in PBS to 100 ml; and added antibodies in the following sequence: 10 mL IgG1, κ PE; 5 mL IgG1, κ FITC; 10 mL CD45 PE; 5 mL CD146 FITC; 10 mL CD45 PE; 5 mL IgG1, κ FITC; 10 mL CD45 PE; and 5 mL CD146 FITC. The samples were then incubated in the dark for 20 min, resuspended in 300 mL PBS and subjected to flow cytometry. At the same time, blood samples were removed and analyzed by automatic blood cell analyzer to obtain the white blood cell count.
SPSS version 20.0 software was used for data processing (IBM Corp., Armonk, NY, United States). The measurement data were expressed as mean ± SD. We compared by independent sample t test the numbers of CECs between the ARDS and healthy control groups, and ARDS and non-ARDS groups, the number of CECs between groups with different severity of ARDS, and the number of CECs between the improved and dead groups. The change in number of CECs after surgery was analyzed by paired t test. Spearman’s correlation was used to analyze the relationship between d2 CECs and operation time. Receiver operating characteristic (ROC) curve was used to detect the number of d2 CECs as best cut-off value to predict ARDS occurrence (obtained by calculating the maximum sensitivity and specificity), and defined the number of d2 CECs higher than cut-off value as Hd2. A binary regression model was used to detect the predictive value on the occurrence of ARDS. All tests were bilateral, and P < 0.05 was considered to be statistically significant.
Among the 125 patients enrolled, 105 patients received ventilator support. If their condition progressed to ARDS, a protective lung ventilation strategy was implemented[10,11]. Comprehensive parameters were monitored including vital signs, blood gas analysis, central venous pressure, and pulse contour cardiac output monitoring was carried out if necessary, and all patients were given bundle treatment in the intensive care unit. We enrolled 52 patients with ARDS; 13 died and the others showed eventual improvement; 73 patients did not develop ARDS and improved (Table 1). CECs were measured by flow cytometry (Figure 1). A1, B1 and C1 were isotype control groups, and A2, B2 and C2 were experimental groups.
D2 CECs were significantly higher in patients with ARDS compared with the healthy control group (P < 0.001) (Table 2). D2 CECs of the ARDS group were significantly higher in the ARDS group than in the non-ARDS group (P < 0.001). D2 CECs in the non-ARDS group were significantly higher than in the healthy control group (P < 0.001).
| Group | Number | Count of CECs, in mL |
| ARDS Group | 52 | 2064 ± 892 |
| Non-ARDS Group | 73 | 1038 ± 1371 |
| Healthy control group | 32 | 167 ± 148 |
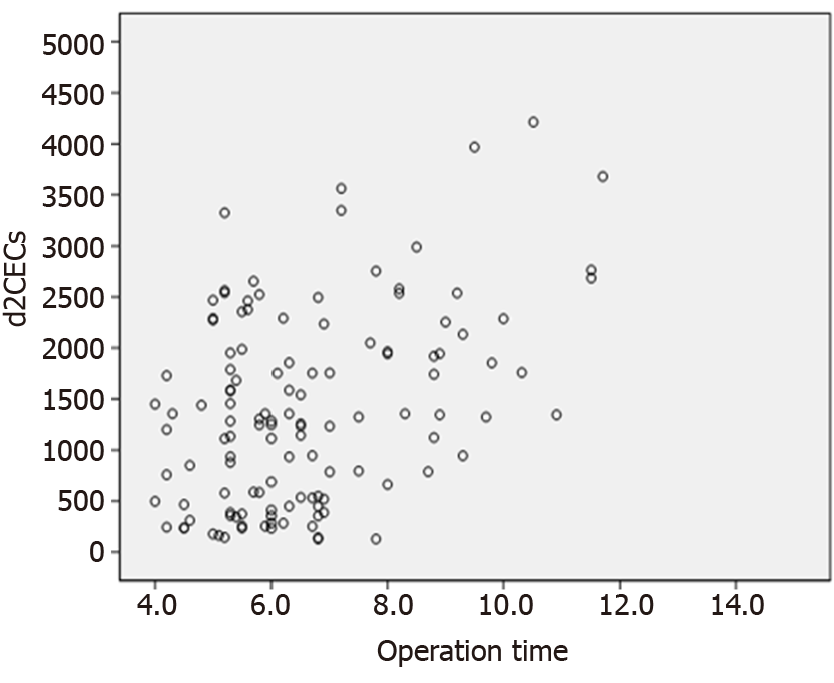
Spearman’s correlation analysis showed a significantly positive correlation between operation time and d2 CECs (rs = 0.302, P = 0.001) (Figure 2).
The number of d2 CECs in the dead group was significantly higher than in the improved group (P < 0.001) (Table 3).
| Group | Number | Count of CECs, mL | P value |
| Deceased group | 13 | 2863 ± 651 | < 0.001 |
| Improvement group | 39 | 1799 ± 801 |
There was no significant difference in number of CECs in patients with mild compared with moderate ARDS (P = 0.924). The number of CECs in patients with severe ARDS was significantly greater than in patients with moderate ARDS (P = 0.037). The number of CECs in patients with severe ARDS was significantly greater than in patients with mild ARDS (P = 0.041) (Table 4).
| Severity of ARDS | Number | Count of CECs, mL |
| Mild | 19 | 1924 ± 872 |
| Moderate | 20 | 1897 ± 890 |
| Severe | 13 | 2580 ± 791 |
There was no significant difference between the number of d5 and d2 CECs in the non-ARDS group after admission (P = 0273). The number of d5 CECs was significantly higher than the number of d2 CECs in the improved group (P < 0.001). The number of d5 CECs was significantly higher than the number of d2 in the deceased group (P = 0.002) (Table 5).
| Group | Number | Count of CECs, mL | P value | |
| Count of d2 CECs | Count of d5 CECs | |||
| Non-ARDS group | 73 | 1038 ± 1371 | 854 ± 648 | 0.273 |
| Improvement group | 39 | 1799 ± 801 | 2274 ± 1017 | < 0.001 |
| Death group | 13 | 2863 ± 652 | 3548 ± 1035 | 0.002 |
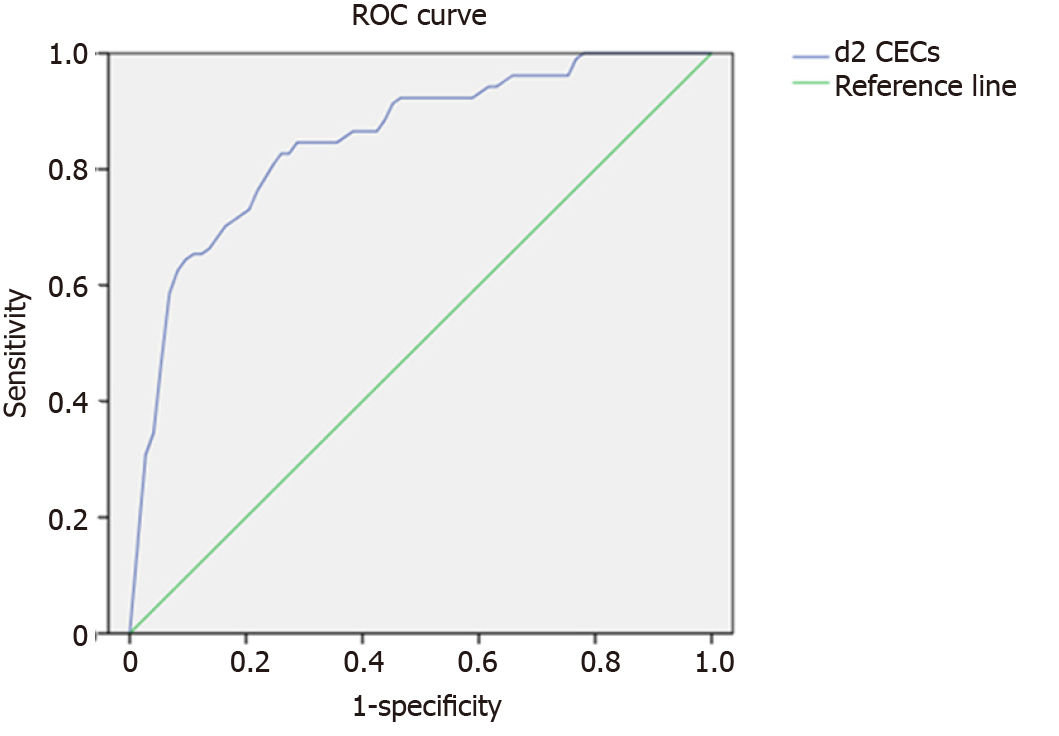
The Youden index of the ROC curve was used to calculate the best cut-off value of d2 CECs at 1351/mL (area under the curve = 0.85, 95%CI: 0.781–0.919, P < 0.001), sensitivity to predict occurrence of ARDS was 80.8%, and specificity was 78.1%. Hd2 was an independent predictor of ARDS (odds ratio = 14.96, 95%CI: 6.18–36.25, P < 0.002) (Figure 3).
CECs refer to vascular endothelial cells (VECs) obtained from circulating blood under physiological or pathological conditions[12]. VECs are large cells that are present in large numbers throughout the pulmonary circulation, and have active metabolism and complex function. Pulmonary VECs are important target and effector cells in ARDS, and are damaged earliest in the disease course[13]. Under pathological conditions, the number of CECs often increases significantly. At present, although research on CECs is common, most of it is limited to studies of infection, cardiovascular disease and oxidative stress. There are few clinical studies on the changes in CECs and their predictive potency in patients with ARDS. Recently, a relationship between endothelial damage, CECs and ARDS has been seen in the study of ARDS induced by coronavirus disease 2019[14].
The cellular immunophenotype of CECs is currently not clear[15], and most endothelial progenitor cells and partially activated T lymphocytes also express part of the same phenotype as CECs. Therefore, it is necessary to label multiple phenotypic antibodies at the same time to identify CECs more accurately by flow cytometry. In this study, we defined CECs as CD146+ CD45- cells.
This is believed to be the first study to compare numbers of CECs in patients with ARDS. In patients with ARDS postoperatively, the number of d2 CECs in the deceased group was higher than in the improved group, so increased number of CECs might predict increased mortality at an early stage. Number of CECs on d2 was correlated with whether patients developed ARDS, severity of ARDS and clinical outcome of ARDS, which indicated that degree of endothelial injury represented by CECs had a predictive and warning effect on the clinical outcome of ARDS. Most ARDS patients are have respiratory dysfunction, but not all of them die of respiratory failure. A large number of etiological studies have shown that multiple organ dysfunction is an independent risk factor for poor prognosis of ARDS[16]. Therefore, extensive endothelial injury and massive platelet thrombosis lead to tissue hypoxia and multiple organ dysfunction. Therefore, changes in CECs, which are a marker of endothelial cell injury, might predict mortality of ARDS patients. Moussa et al[17] have suggested that activation and dysfunction of endothelial cells are involved in the pathophysiological process of ARDS. CECs might be a useful biomarker of endothelial dysfunction and injury. Therefore, they conducted a study on sepsis-related ARDS. The median CECs count in moderate and severe ARDS on d1 was significantly higher than that in patients with sepsis and mild ARDS and sepsis control patients, and all sepsis patients (with or without ARDS) had higher CECs counts on d1 than patients without ARDS. Therefore, CECs count on d1 of ARDS could be used as a useful indicator of severity of ARDS.
In our study, the number of CECs in patients with ARDS on d2, postoperatively, was significantly higher than that in the control group. Moreover, ROC curve analysis showed that the best cut-off value of d2 CECs was 1351 cells/mL. Therefore, number of CECs was an effective marker to predict development and outcome of ARDS. The cut-off value showed that when CECs were > 1351 cells/mL, patients were more likely to have ARDS, and sensitivity and specificity were 80.8% and 78.1%, respectively. For patients with ARDS, there was no significant difference in number of d2 CECs between mild and moderate disease, while the number of d2 CECs in severe ARDS was significantly higher than that in mild and moderate ARDS. Therefore, it could be inferred that number of CECs in peripheral blood is significantly increased in patients with severe ARDS, which reflected severity of ARDS. We need to expand sample size and investigate specific changes in CECs in peripheral blood of patients with ARDS of different severity.
According to the current consensus, the time course of ARDS is: corresponding respiratory symptoms appear, or respiratory symptoms are aggravated, or new respiratory symptoms appear after an external stress within 1 wk[18,19]. Therefore, in our study, the number of CECs was detected on d5 for patients enrolled postoperatively. We found that there was no significant change in CECs during recovery of non-ARDS patients, suggesting that there was no further damage to the vascular endothelial system; however, the number of CECs increased in the improved and deceased groups of ARDS patients, which was thought to be related to the combined effects of many factors such as shock and infection postoperatively. We found that there was a positive correlation between operation time and number of CECs, suggesting that longer operation time resulted in greater trauma. The effects of the operation itself and postoperative inflammatory reaction could both lead to damage of the vascular endothelial system. This mechanism is consistent with pathogenesis of extrapulmonary ARDS[20].
Changes in number of CECs might predict occurrence and adverse outcome of ARDS postoperatively, and higher numbers of CECs are associated with worse prognosis of ARDS.
Acute respiratory distress syndrome (ARDS) involves alveolar epithelial cells and pulmonary interstitial capillary endothelial cells. Circulating endothelial cells (CECs) are the only marker that directly reflects vascular endothelial injury in vivo. There have been few studies on the correlation between peripheral blood CECs and ARDS at home and abroad.
This research studied correlation between level of CECs and severity of ARDS, and preliminarily observed change trend of CECs at different time points. This is believed to be initiated research to compare CECs levels changes in patients with ARDS, and it had value of guiding treatment and evaluating prognosis for ARDS patients.
This study aimed to explore the correlation between CEC level and severity of ARDS in patients postoperatively.
Blood samples were collected from all patients on day 2 (d2) and day 5 (d5) after surgery. Number of CECs was measured by flow cytometry, and operation time was recorded. Changes in various indexes of patients were monitored, and diagnosis of ARDS was determined based on ARDS Berlin definition.
The number of d2 CECs in the ARDS group was significantly higher than that in the healthy control group. The number of d2 CECs in the ARDS group was significantly higher than that in the non-ARDS group. The number of d2 CECs in the non-ARDS group was significantly higher than that in the healthy control group. There was no significant difference in number of d2 CECs between patients with mild and moderate ARDS. The number of d2 CECs in patients with severe ARDS was significantly higher than that in patients with mild and moderate ARDS. The number of d5 CECs was higher than the number of d2 CECs in the ARDS deceased group.
Changes in number of CECs might predict occurrence and adverse outcome of ARDS postoperatively, and higher numbers of CECs are associated with worse prognosis of ARDS.
In future experiments, we need to further expand sample size by collecting more enrolled patients, refining grouping, and conducting hierarchical analysis. If one special group of patients was dynamically tracked for detection, this would increase the refinement of CECs detection time points for better observation of dynamic changes of CECs.
Provenance and peer review: Unsolicited article; Externally peer reviewed
Specialty type: Critical Care Medicine
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Arima A S-Editor: Wang JL L-Editor: Filipodia P-Editor: Wang JL
| 1. | Spinelli E, Mauri T, Beitler JR, Pesenti A, Brodie D. Respiratory drive in the acute respiratory distress syndrome: pathophysiology, monitoring, and therapeutic interventions. Intensive Care Med. 2020;46:606-618. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 87] [Cited by in F6Publishing: 131] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 2. | Yadav H, Thompson BT, Gajic O. Fifty Years of Research in ARDS. Is Acute Respiratory Distress Syndrome a Preventable Disease? Am J Respir Crit Care Med. 2017;195:725-736. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 74] [Cited by in F6Publishing: 103] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 3. | Redant S, Devriendt J, Botta I, Attou R, De Bels D, Honoré PM, Pierrakos C. Diagnosing Acute Respiratory Distress Syndrome with the Berlin Definition: Which Technical Investigations Should be the Best to Confirm it? J Transl Int Med. 2019;7:1-2. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 4. | Rawal G, Yadav S, Kumar R. Acute Respiratory Distress Syndrome: An Update and Review. J Transl Int Med. 2018;6:74-77. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 39] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 5. | Huppert LA, Matthay MA, Ware LB. Pathogenesis of Acute Respiratory Distress Syndrome. Semin Respir Crit Care Med. 2019;40:31-39. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 256] [Cited by in F6Publishing: 234] [Article Influence: 46.8] [Reference Citation Analysis (0)] |
| 6. | Ren R, Ruan Z, Ding H, Du J, Yu W. Phosphoproteome profiling provides insight into the mechanisms of ventilator-induced lung injury. Exp Ther Med. 2020;19:3627-3633. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1] [Cited by in F6Publishing: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 7. | Harris AJ, Mirchandani AS, Lynch RW, Murphy F, Delaney L, Small D, Coelho P, Watts ER, Sadiku P, Griffith D, Dickinson RS, Clark E, Willson JA, Morrison T, Mazzone M, Carmeliet P, Ghesquiere B, O'Kane C, McAuley D, Jenkins SJ, Whyte MKB, Walmsley SR. IL4Rα Signaling Abrogates Hypoxic Neutrophil Survival and Limits Acute Lung Injury Responses In Vivo. Am J Respir Crit Care Med. 2019;200:235-246. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 8. | Janz DR, Ware LB. Biomarkers of ALI/ARDS: pathogenesis, discovery, and relevance to clinical trials. Semin Respir Crit Care Med. 2013;34:537-548. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 48] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 9. | Eizawa T, Ikeda U, Murakami Y, Matsui K, Yoshioka T, Suzuki C, Takahashi M, Muroi K, Kamisawa O, Fuse K, Shimada K. Increase in circulating endothelial progenitor cells after aortic aneurysm repair. Heart Vessels. 2004;19:107-110. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 10. | Derwall M, Martin L, Rossaint R. The acute respiratory distress syndrome: pathophysiology, current clinical practice, and emerging therapies. Expert Rev Respir Med. 2018;12:1021-1029. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 11. | Neto AS, Simonis FD, Barbas CS, Biehl M, Determann RM, Elmer J, Friedman G, Gajic O, Goldstein JN, Linko R, Pinheiro de Oliveira R, Sundar S, Talmor D, Wolthuis EK, Gama de Abreu M, Pelosi P, Schultz MJ; PROtective Ventilation Network Investigators. Lung-Protective Ventilation With Low Tidal Volumes and the Occurrence of Pulmonary Complications in Patients Without Acute Respiratory Distress Syndrome: A Systematic Review and Individual Patient Data Analysis. Crit Care Med. 2015;43:2155-2163. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 150] [Cited by in F6Publishing: 168] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 12. | Mohandas R, Diao Y, Chamarthi G, Krishnan S, Agrawal N, Wen X, Dass B, Shukla AM, Gopal S, Koç M, Segal MS. Circulating endothelial cells as predictor of long-term mortality and adverse cardiovascular outcomes in hemodialysis patients. Semin Dial. 2021;34:163-169. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1] [Cited by in F6Publishing: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 13. | Kawasaki T, Chen W, Htwe YM, Tatsumi K, Dudek SM. DPP4 inhibition by sitagliptin attenuates LPS-induced lung injury in mice. Am J Physiol Lung Cell Mol Physiol. 2018;315:L834-L845. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 67] [Cited by in F6Publishing: 80] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 14. | Rambaldi A, Gritti G, Micò MC, Frigeni M, Borleri G, Salvi A, Landi F, Pavoni C, Sonzogni A, Gianatti A, Binda F, Fagiuoli S, Di Marco F, Lorini L, Remuzzi G, Whitaker S, Demopulos G. Endothelial injury and thrombotic microangiopathy in COVID-19: Treatment with the lectin-pathway inhibitor narsoplimab. Immunobiology. 2020;225:152001. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 83] [Cited by in F6Publishing: 94] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 15. | Danova M, Comolli G, Manzoni M, Torchio M, Mazzini G. Flow cytometric analysis of circulating endothelial cells and endothelial progenitors for clinical purposes in oncology: A critical evaluation. Mol Clin Oncol. 2016;4:909-917. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 16. | Bhatia M, Moochhala S. Role of inflammatory mediators in the pathophysiology of acute respiratory distress syndrome. J Pathol. 2004;202:145-156. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 810] [Cited by in F6Publishing: 846] [Article Influence: 42.3] [Reference Citation Analysis (0)] |
| 17. | Moussa MD, Santonocito C, Fagnoul D, Donadello K, Pradier O, Gaussem P, De Backer D, Vincent JL. Evaluation of endothelial damage in sepsis-related ARDS using circulating endothelial cells. Intensive Care Med. 2015;41:231-238. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 18. | Potey PM, Rossi AG, Lucas CD, Dorward DA. Neutrophils in the initiation and resolution of acute pulmonary inflammation: understanding biological function and therapeutic potential. J Pathol. 2019;247:672-685. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 122] [Cited by in F6Publishing: 136] [Article Influence: 27.2] [Reference Citation Analysis (0)] |
| 19. | Rebetz J, Semple JW, Kapur R. The Pathogenic Involvement of Neutrophils in Acute Respiratory Distress Syndrome and Transfusion-Related Acute Lung Injury. Transfus Med Hemother. 2018;45:290-298. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 66] [Cited by in F6Publishing: 62] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 20. | Xiong S, Hong Z, Huang LS, Tsukasaki Y, Nepal S, Di A, Zhong M, Wu W, Ye Z, Gao X, Rao GN, Mehta D, Rehman J, Malik AB. IL-1β suppression of VE-cadherin transcription underlies sepsis-induced inflammatory lung injury. J Clin Invest. 2020;130:3684-3698. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 107] [Cited by in F6Publishing: 103] [Article Influence: 25.8] [Reference Citation Analysis (0)] |