Abstract
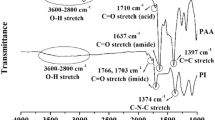
The novel diamine containing bulky tert-butyl substituent, 1,4-bis(4-aminophenoxy)-2-tert-butylbenzene (BATB) was used as monomer with various aromatic tetracarboxylic dianhydrides to synthesize polyimides via a conventional two-stage procedure that included ring-opening polyaddition in a polar solvent such as N,N-dimethylacetamide (DMAc) to give poly(amic acid)s, followed by cyclodehydration to polyimides. BATB(II) was prepared through the nucleophilic displacement of tert-butylhydroquinone with p-chloronitrobenzene in the presence of K2CO3, followed by catalytic reduction. Depending on the dianhydride used, the poly(amic acid)s obtained had inherent viscosities of 0.76–1.79 dL g−1. All poly(amic acid)s could be cast from the DMAc solutions and thermally converted to transparent, fiexible, and tough polyimide films. The polyimide films had high tensile strength of 122–208 MPa, an elongation at breaks of 3–9%, and a tensile modulus of 1.92–2.68 GPa. Polyimides from 4,4′-sulfonyldiphthalic anhydride and 4,4′-hexafluoro-isopropylidenediphathalic anhydride had better solubility than the other polyimides. These polyimides had glass transition temperatures between 240–268°C and 10% mass loss temperatures were recorded at 479–522°C in nitrogen.
Similar content being viewed by others
Article PDF
References
P. E. Cassidy, “Thermally Stable Polymers,” Dekker, New York., N.Y., 1980.
M. I. Bessonov, M. M. Koton, V. V. Kudryavtsev, and L. A. Laius, Ed., “Polyimides,” Consultants Bureau, New York, N.Y., 1987.
C. E. Sroog, J. Polym. Sci., Macromol. Rev., 11, 161 (1976).
W. A. Feld, B. Ramalingam, and F. W. Harris, J. Polym. Sci., Polym. Chem. Ed., 21, 319 (1983).
Y. Imai, N. N. Maldar, and M. Kakimoto, J. Polym. Sci., Part A: Polym. Chem., 22, 2189 (1989).
N. D. Ghatge, B. M. Shinde, and U. P. Mulik, J. Polym. Sci., Polym. Chem. Ed., 22, 3359 (1984).
T. Takekoshi, J. G. Wirth, D. R. Heath, J. E. Kochanowski, J. S. Manello, and M. J. Webber, J. Polym. Sci., Polym. Chem. Ed., 18, 3069 (1980).
J. P. Critchley and M. A. White, J. Polym. Sci., Polym. Chem. Ed., 10, 1809 (1972).
T. L. St. Clair, A. K. St. Clair, and E. N. Smith, Polym. Prepr., Am. Chem. Soc., Div. Polym. Chem., 17, 359 (1976).
V. L. Bell, B. L. Stump, and H. Gager, J. Polym. Sci., Polym. Chem. Ed., 14, 2275 (1976).
H. J. Jeong, Y. Oishi, M. Kakimoto, and Y. Imai, J. Polym. Sci., Part A: Polym. Chem., 29, 39 (1991).
H. J. Jeong, M. Kakimoto, and Y. Imai, J. Polym. Sci., Part A: Polym. Chem., 29, 1691 (1991).
Y. Oishi, M. Ishida, M. Kakimoto, and Y. Imai, and T. Kurosaki, J. Polym. Sci., Part A: Polym. Chem., 30, 1027 (1992).
Y. Imai and M. Kakimoto, Polym. Plast. Technol. Eng., 28, 371 (1989).
M. Sato and M. Yokoyama, Eur. Polym. J., 15, 733 (1979).
P. K. Bhowmik, E. D. T. Atkins and H. Han, Polym. Prepr., Am. Chem. Soc., Div. Polym. Chem., 36, 326 (1995).
Z. G. Li, J. E. Mclntyre, and J. G. Tomka, Polymer, 34, 551 (1993).
T. Heitz, P. Rohrbach, and H. Hoecker, Makromol. Chem., 190, 3295 (1989).
W. Heitz and N. Niessner, Makromol. Chem., 191, 225 (1990).
H. W. Schmidt and D. Guo, Makromol. Chem., 189, 2029 (1988).
W. Risse and D. T. Sogah, Macromolecules, 23, 4029 (1990).
D. J. Liaw, J. Polym. Sci., Part A: Polym. Chem., 33, 605, (1995).
K. H. Becker and H. W. Schmidt, Polym. Mater. Sci. Eng., 66, 303 (1992).
C. P. Yang and S. H. Hsiao, J. Appl. Polym. Sci., 30, 2883 (1985).
C. P. Yang and W. T. Chen, Macromolecules, 26, 4865 (1993).
S. E. Delos, R. K. Schellenberg, J. E. Smedley, and D. E. Kranbuehl, J. Appl. Polym. Sci., 27, 4295 (1982).
D. J. Liaw and W. C. Shen, Angew. Makromol. Chem., 199, 171 (1992).
F. Keitoku, M. Kakimoto, Y. Imai, J. Polym. Sci., Part A: Polym. Chem., 32, 317 (1994).
S. H. Hsiao, C. P. Yang, and C. K. Lin, J. Polym. Res., 2, 1 (1995).
C. P. Yang and J. H. Lin, J. Polym. Sci., Part A: Polym. Chem., 30, 1027 (1992).
D. J. Liaw and B. Y. Liaw, J. Polym. Sci., Part A: Polym. Chem., submitted.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liaw, DJ., Liaw, BY. Synthesis and Properties of Polyimides Derived from 1,4-Bis(4-aminophenoxy)-2-tert-butylbenzene. Polym J 28, 970–975 (1996). https://doi.org/10.1295/polymj.28.970
Issue Date:
DOI: https://doi.org/10.1295/polymj.28.970
Keywords
This article is cited by
-
Synthesis and characterization of polyimides from 4,4′-(3-(tert-butyl)-4-aminophenoxy)diphenyl ether
Journal of Polymer Research (2017)
-
Soluble aromatic polyimides with high glass transition temperature from benzidine containing tert-butyl groups
Journal of Polymer Research (2014)
-
Synthesis and characterization of organosoluble aromatic copolyimids
Frontiers of Chemistry in China (2007)