Abstract
Gastric cancer (GC) remains one of the world’s most common and fatal malignant tumors. With a refined understanding of molecular typing in recent years, microsatellite instability (MSI) has become a major molecular typing approach for gastric cancer. MSI is well recognized for its important role during the immunotherapy of advanced GC. However, its value remains unclear in resectable gastric cancer. The reported incidence of microsatellite instability-high (MSI-H)/deficient mismatch repair (dMMR) in resectable gastric cancer varies widely, with no consensus reached on the value of postoperative adjuvant therapy in patients with MSI-H/dMMR resectable GC. It has been established that MSI-H/dMMR tumor cells can elicit an endogenous immune antitumor response and ubiquitously express immune checkpoint ligands such as PD-1 or PD-L1. On the basis of these considerations, MSI-H/dMMR resectable GCs are responsive to adjuvant immunotherapy, although limited research has hitherto been conducted. In this review, we comprehensively describe the differences in geographic distribution and pathological stages in patients with MSI-H/dMMR with resectable gastric cancer and explore the value of adjuvant chemotherapy and immunotherapy on MSI-H/dMMR to provide a foothold for the individualized treatment of this patient population.
Similar content being viewed by others
Gastric cancer (GC) is an aggressive and heterogeneous malignancy with limited therapeutic options available, especially in locally advanced and metastatic stages, accounting for the poor prognosis of affected patients.1 Less than 50% of patients are diagnosed with early stage disease, and surgical resection plus lymphadenectomy remains the mainstay of curative treatment. Surgical resection with subsequent adjuvant chemotherapy has been established as the standard-of-care treatment for patients with stages II and III gastric cancer, which is more beneficial than surgery alone in terms of overall survival (OS).2 Unfortunately, approximately 40–60% of patients who undergo resection relapse and die from cancer.3,4,5
With significant progress achieved in molecular research on gastric cancer, many researchers have attempted to explore the underlying mechanisms and inherent characteristics of the genome, transcriptome, and protein expression of gastric carcinomas, and have successfully proposed several molecular subtyping systems.5,6,7 According to the Cancer Genome Atlas (TCGA) and Asian Cancer Research Group (ACRG), the microsatellite instability (MSI) subtype represents an important molecular subtype of gastric cancer. Microsatellites (MS) are DNA regions prone to mutations, usually assessed by polymerase chain reaction (PCR)/next-generation sequencing (NGS).8 An increasing body of evidence suggests that the mismatch repair (MMR) system measured by immunohistochemistry (IHC) is responsible for monitoring and correcting errors in DNA replication.9,10,11 Studies by Kim et al.12 and Smyth et al.13 evaluated the consistency of microsatellite status and MMR protein expression in gastric cancer and concluded that microsatellite instability-high (MSI-H) was generally strongly correlated with deficient mismatch repair (dMMR).
The proportion of MSI-H/dMMR in resectable gastric cancer exhibits significant heterogeneity across different studies. The prevalence of MSI-H in patients with gastric cancer was 23.5% (n = 111/472) in Italy14 but only 8.5% (n = 170/1990) in South Korea.15 In recent years, the value of adjuvant therapy in MSI-H/dMMR colon cancers has become increasingly well established. In this respect, the National Comprehensive Cancer Network (NCCN) guidelines for colon cancer16 classify patients with MSI-H/dMMR as low risk and have recommended that patients with stage II disease and MSI-H do not require adjuvant therapy since 2010. In the context of MSI-H/dMMR gastric cancer, the existing data in the adjuvant setting are still pauce, and the existing guidelines from leading organizations such as NCCN, CSCO, and ESMO do not offer definitive recommendations regarding the appropriate selection of adjuvant treatment. Although the potential of adjuvant therapy in MSI-H/dMMR resectable gastric cancer has been poorly explored, the effectiveness of immunotherapy has been demonstrated in MSI-H/dMMR advanced gastric cancer. Immune checkpoint inhibitors (ICIs) have been integrated into the NCCN gastric cancer guidelines as a second-line or subsequent treatment for MSI-H/dMMR advanced gastric cancer and have even been suggested as the first-line therapy by the Chinese Society of Clinical Oncology (CSCO) guidelines,17 although their value in the setting of MSI-H/dMMR resectable gastric cancer has not been confirmed. Several small-scale clinical trials are currently underway to predict the value of immunotherapy in MSI-H/dMMR resectable gastric cancer, but no results have been reported yet.
In this review, we focus on the geographical variations and differences in pathological stages of MSI-H/dMMR resectable gastric cancer on the basis of the latest literature, and highlight the potential of adjuvant chemotherapy and immunotherapy for patients with MSI-H/dMMR resectable GC.
MSI-H/dMMR GCs: Geographical Differences
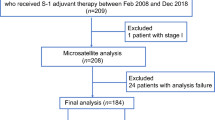
Gastric cancer (GC) accounts for the second highest mortality among cancers worldwide, with higher prevalence in East Asia, especially in Japan, China, South Korea, and some developing countries.7,18 To identify relevant studies, a comprehensive literature search was conducted in PubMed and Web of Science databases (Supplementary Methods, Fig. S1). Given the high concordance with MSI-H, dMMR was also included at retrieval. To enhance the accuracy of our review, we restricted our analysis to studies with a sample size greater than 50. As a result, we identified 31 records that reported the frequencies of MSI-H or dMMR in gastric cancer. Our retrospective analysis of studies conducted over the past few years indicated that the incidence of MSI-H/dMMR varied among countries, ranging from 5.0 to 23.5%.9,14,15,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46 (Table 1 and Fig. 1a). It has been established that the proportion of MSI-H/dMMR gastric cancer in Asia is lower than in European countries (Fig. 1b). In this respect, the median proportion of MSI-H/dMMR GC is 9.3% and 13.8% in Asian and European countries, respectively. Meanwhile, the proportion of MSI-H/dMMR in different nations was similar but differed considerably in America (Fig. S2). A study from Canada reported that the prevalence of MSI-H/dMMR was 5% (n = 7/139),20 while in other research from North America, MSI-H/dMMR was found in 18.7% (n = 52/278) of patients.45 This disparity may be attributed to the relatively small number of patients enrolled in each study. Moreover, considering that the frequency of MSI-H/dMMR may vary by stage, which was scientifically valid in the case of colorectal cancer,47,48 we adjusted for any such stage differences when examining regional differences. The proportion difference in each stage between Asian and European populations is plotted in Fig. 2a–c, and stage I and stage II were combined as the early stage of gastric cancer because of the small amount of literature. Interestingly, the proportion of MSI-H/dMMR in Europe was significantly higher than in Asia, irrespective of the cancer stage (P < 0.05).
Regional differences of MSI-H/dMMR proportion in each stage. a Proportion of MSI-H/dMMR in Asia vs. Europe in stages I–II (*P = 0.0128, unpaired t-test). b Proportion of MSI-H/dMMR in Asia vs. Europe in stage III (*P = 0.0253, unpaired t-test). c Proportion of MSI-H/dMMR in Asia vs. Europe in stage IV (**P = 0.0092, unpaired t-test)
MSI-H/dMMR GCs: Differences in Pathological Stage
Our literature review showed that 14 of 31 studies further staged patients with MSI-H/dMMR according to the American Joint Committee on Cancer (AJCC) staging system (Table 1). Although MSI-H/dMMR can be observed in all gastric cancer stages, the proportion of MSI-H/dMMR in resectable gastric cancer was generally higher than in stage IV disease (Table 2, Fig. 3a). Interestingly, in approximately 80% of included studies, the prevalence of MSI-H/dMMR in stage II was the highest (Fig. 3b). For instance, in a large-scale study published in 2012, tissue specimens from 1990 patients with gastric cancer were collected and analyzed using PCR amplification.15 The prevalence of MSI-H/dMMR was the highest in stage II GC (12.8%), followed by stages I (9.1%), IV (5.3%), and III (4.5%). An increasing body of evidence from recently published studies29,36 suggests that the incidence of MSI-H/dMMR disease in patients with gastric cancer is higher in stage II than in other stages. Although the proportion of MSI-H/dMMR in resectable gastric cancers was higher than in stage IV, the effect of postoperative adjuvant therapy in resectable gastric cancer remains unclear, warranting further exploration.
Adjuvant Chemotherapy of MSI-H/dMMR Resectable GCs: Opportunity or Hindrance?
With adjuvant chemotherapy based on fluorouracil being guideline-endorsed for stage II/III resectable GCs, researchers have been increasingly interested in the sensitivity of chemotherapy drugs to MSI-H/dMMR gastric cancer (Table 3). The MAGIC trial13 conducted in 2017 (ECF neoadjuvant scheme: epirubicin + cisplatin + fluorouracil) and the CLASSIC trial24 in 2019 (XELOX adjuvant scheme: capecitabine plus oxaliplatin) both concluded that patients with MSI-H/dMMR gastric cancer do not benefit from perioperative or postoperative chemotherapy. In addition, a meta-analysis involving samples from four large randomized clinical trials (MAGIC, CLASSIC, ARTIST, and ITACA-S) revealed no significant benefit in OS at 5 years (75.4% in the postoperative chemotherapy arm vs. 82.8% in the only-surgery arm).49 The previously mentioned studies advocated that MSI-high gastric cancers were less likely to respond to chemotherapy.
In recent years, the role of MSI status in predicting the chemotherapy response has been extensively studied. A retrospective study on patients with stage II/III MSI-H/dMMR GC published in 2020 predicted the efficacy of adjuvant therapy by creating a new immune scoring system (ISSGC), suggesting that patients with MSI-H/dMMR GC have longer OS with adjuvant chemotherapy after surgery [hazard ratio, HR 0.59 (0.38–0.93)].50 Notwithstanding that many other studies demonstrated a favorable trend with the implementation of adjuvant chemotherapy on patients with MSI-H/dMMR, the final results remained non-significant.51 For example, in a study performed by Tsai and colleagues,36 although patients who received adjuvant chemotherapy showed better 5 year disease-free survival (DFS) (61.2% vs. 55.9%), no significant benefit was found for postoperative adjuvant chemotherapy of MSI-H/dMMR gastric cancer [DFS, HR 0.64 (0.21–1.94) and OS, HR 0.8 (0.35–1.82)]. In a recent meta-analysis,52 seven studies were included to explore the prognostic impact of adjuvant chemotherapy. By using Cox models or fixed/random effects models to pool HR, it was found that patients with MSI-H/dMMR could benefit from adjuvant chemotherapy, with estimated HRs of 0.56 (95% CI 0.36–0.87; P = 0.010) for DFS and 0.62 (95% CI 0.45–0.83; P = 0.002) for OS.
No consensus has been reached on the value of MSI-H/dMMR as a predictor of the efficacy of adjuvant chemotherapy, mainly due to the low number of patients with MSI-H/dMMR, the non-standardization of MSI detection standards, and the retrospective nature of most studies in the literature. In addition, only one prospective clinical trial (NCT03485196)53 analyzed the relationship between MSI status and efficacy of 5-fluorouracil (5-FU)-based adjuvant chemotherapy by observing the survival period of patients with different MSI statuses. This multicohort trial initiated by the Affiliated Hospital of Qingdao University in China involved 1000 patients with gastric cancer and was completed in August 2022. On the basis of the results, it was concluded that there was no significant difference in 1- and 2-year disease-free survival (DFS) rates after adjuvant chemotherapy among different MSI statuses, including MSI-H and dMMR, regardless of the specific adjuvant chemotherapy regimen used, such as XELOX, SOX, or other adjuvant treatment protocols.
Immunotherapy of MSI-H/dMMR Resectable GCs: A Possible New Approach
The past few years have witnessed a burgeoning interest in immunotherapy, especially gastric cancer research (Table 4). On the basis of the results of the CheckMate 64954 and ORIENT-1655 trials, nivolumab or sintilimab combined with chemotherapy, as the first-line treatment of advanced gastric cancer, is more effective and safer than chemotherapy alone in China and abroad. Meanwhile, pembrolizumab or dostarlimab-gxly is recommended as a second-line or subsequent therapy for MSI-H/dMMR advanced gastric cancer by the 2022 NCCN guidelines.2
Few clinical trials assessing adjuvant immunotherapy for resectable MSI-H/dMMR gastric cancer are currently underway. However, in the phase III IMpower010 study,56 anti-PD-L1 atezolizumab adjuvant treatment was found to improve the progression-free survival (PFS) (HR 0.79) and DFS (HR 0.66) of patients with resectable non-small cell lung cancer (NSCLC). As for postoperative adjuvant immunotherapy of resectable MSI-H/dMMR gastric cancer, we only identified two ongoing clinical trials (Table 5). The NCT05468138 study conducted by Fudan University in Shanghai aims to assess the efficacy of PD-1 antibody as an adjuvant treatment for patients with MSI-H/dMMR gastric cancer after D2 radical surgery. The PD-1 antibody (sintilimab or nivolumab) was set as the experimental group, and the standard chemotherapy regimen (SOX, XELOX) as the control group to compare the 3-year DFS of different adjuvant chemotherapy schemes. Another trial from Shanghai Tongji University (NCT04152889) is being carried out to evaluate the efficacy of camrelizumab in combination with docetaxel + S-1 sequenced by camrelizumab + S-1 in patients with stage III gastric cancer. Patients with stage III PD-L1+/MSI-H/EBV+/dMMR gastric cancer were included to explore the efficacy of combined immunochemotherapy. Although immunoadjuvant therapy can significantly prolong the survival of patients with NSCLC, further data on whether patients with MSI-H/dMMR resectable gastric cancer could benefit from immunoadjuvant treatment is required.
Besides research on immunoadjuvant therapy, immunotherapy is now used for neoadjuvant therapy, representing a good choice for patients with resectable MSI-H/dMMR gastric cancer. On the basis of the rationale of neoadjuvant treatment, more benefits could be obtained by immunotherapy. Indeed, patients are usually in a better physical condition before surgery, which can better mobilize immune function, and more antigens are released during an immune attack due to the presence of the primary tumor, leading to increased sensitivity to immunotherapy. A case series was published in 2020,57 including six patients who received neoadjuvant ICIs and surgery for advanced, resectable, and MSI-H gastrointestinal tumors. After radical surgery, pathological responses were observed in all MSI-H/dMMR tumors, with a complete response observed in 83% (n = 5/6) of patients, substantiating the efficacy of neoadjuvant immunotherapy in patients with MSI-H/dMMR gastrointestinal tumors. The American Society of Clinical Oncology (ASCO) annual meeting in 2022 reported the final data of the NICHE study,58 the first neoadjuvant immunotherapy study in colon cancer. It was found that 100% of dMMR patients responded to the combination of neoadjuvant nivolumab plus ipilimumab, and 97% of dMMR patients achieved a major pathologic response (MPR). Substantial benefits were observed with the double-blocking effect of PD-1 and CTLA4. According to the NCCN guidelines published in 2022,16 nivolumab ± ipilimumab or pembrolizumab (preferred) is recommended as a neoadjuvant treatment option for resectable MSI-H/dMMR metastatic colorectal cancer (mCRC). During the GERCOR NEONIPIGA phase II study (NCT04006262),59 nivolumab- and ipilimumab-based neoadjuvant therapy was also indicated for MSI-H/dMMR gastric/gastroesophageal junction cancer (G/GEJ) adenocarcinoma, with a pathologic complete response (pCR) of 59%. The feasibility of double-blocking neoadjuvant treatment was demonstrated and associated with a high pCR rate in gastric cancer, suggesting that some patients with MSI-H/dMMR GC may be protected from surgery. The INFINITY study,60 an ongoing phase II, multicenter, single-arm, multicohort trial from Italy, was designed to assess the activity and safety of the combination of anti-CTLA4 tremelimumab and the anti-PD-L1 durvalumab as a neoadjuvant treatment for patients with resectable MSI-H/dMMR G/GEJ cancer. Approximately 310 patients underwent the molecular prescreening test. Ultimately, 31 patients were enrolled and classified in cohorts 1 (n = 18) and 2 (n = 13). Patients in cohort 1 received a 12 week treatment with a single high dose of tremelimumab 300 mg and durvalumab 1500 mg for 4 weeks (T300/D) for three cycles followed by surgery, and cohort 2 investigated non-operative management after the same treatment regimen. The primary endpoint of cohort 1 was pCR rate (ypT0N0) with negative circulating-tumor DNA (ctDNA) status after T300/D neoadjuvant immunotherapy in the resectable MSI-H/dMMR G/GEJ cancer population. The preliminary results have been published in ASCO 2023,61 showing that among 15 evaluable patients, the pCR rate was 60% (9/15) and the major-complete pathological response (< 10% viable cells) was 80%. Overall, preoperative T300/D was safe and provided promising proof of biological clinical evidence on the neoadjuvant immunotherapy schedule with ICIs of patients with resectable MSI-H/dMMR G/GEJ cancer. As previously mentioned, neoadjuvant immunotherapy for patients with resectable MSI-H/dMMR GC has also been actively implemented in clinical practice. A case report by Chubenko et al.62 illustrated the favorable effects of neoadjuvant chemotherapy on a patient with MSI-H/dMMR GC. Partial remission was observed by computed tomography (CT) examination after four cycles of neoadjuvant therapy with nivolumab. After six cycles of neoadjuvant nivolumab treatment, endoscopic surgery was performed, and postoperative pathology showed no residual tumor cells, achieving pCR. In addition, four prospective clinical trials are currently underway with pembrolizumab used in two experimental groups (Table 5). In the experimental group of the two remaining trials, AK104 (a PD-1/CTLA-4 bispecific antibody) and JS001 are being assessed, respectively. JS001 is a Chinese anti-PD-1 monoclonal injection antibody approved for melanoma. It is highly conceivable that the preoperative treatment paradigm will evolve in the near future as results from these clinical trials evaluating the inclusion of immunodrugs in neoadjuvant therapy become available.
Discussion
To our knowledge, this is the first review to investigate the heterogeneity and adjuvant therapy regimes in resectable GC with MSI-H/dMMR. We explored the heterogeneity of MSI-H/dMMR with emphasis placed on the regional and pathological stage and conducted an in-depth investigation of the effect of adjuvant chemotherapy and immunotherapy in resectable MSI-H/dMMR GC.
Interestingly, we found significant geographic disparities in MSI-H/dMMR prevalence. In this respect, the prevalence of MSI-H/dMMR was higher in the West than in Asia, suggesting the presence of ethnic differences. This heterogeneity has been validated in early resectable and late GC, attributed to the influence of factors,63,64,65 and has significant clinical implications. From the genetics perspective, it has been reported that genetic polymorphisms of some metabolic enzymes and genes such as cytochrome p450 2E1 (CYP2E1),63 glutathione S-transferase mu 1 (GSTM1),64 and glutathione S-transferase theta 1 (GSTT1)65 are closely related to the development of gastric cancer. As for genes that influence the expression of microsatellites, the significance of the hMLH1 gene promoter has been established. Hypermethylation of the promoter of the hMLH1 gene reportedly plays an important role in mismatch repair during DNA replication and is significantly associated with microsatellite instability.66 A total of 71.4% of MSI-positive GC tumors showed hypermethylation, whereas only 29.8% of MSI-negative tumors were hypermethylated at the hMLH1 promoter region. In addition, cigarette smoking and alcohol consumption have been associated with the hypermethylation of the hMLH1 gene promoter, which may increase the possibility of microsatellite instability. A population-based case-control study conducted in Italy involving 126 patients with gastric cancer was carried out by Palli et al. to evaluate the role of dietary risk factors in GC according to MSI statuses.67,68 A specific diet pattern was associated with MSI+ gastric cancer, indicating that frequent consumption of fresh fruits and vegetables can greatly reduce the risk of MSI cancer, while high consumption of red meat, meat paste, total protein, and nitrite increased the risk of MSI GCs. A Western-style diet rich in red meat, processed meat (steak, sausage, etc.), and refined compounds (hamburger, bread, etc.) represents a potential cause of the elevated fraction of MSI-H/dMMR among European and American countries. In addition, the high proportion might be attributed to increased alcohol consumption in Western countries. In this respect, a pivotal study showed that the average alcohol consumption of American and Chinese men was 15 L and 10 L, respectively.69 The above studies overlap in their assertion that this heterogeneity is influenced by a combination of genetics, dietary habits, and other factors, emphasizing the need for further studies.
Interestingly, we also found that resectable gastric cancers have a higher frequency of MSI-H/dMMR than advanced-stage GC, especially stage II disease, similar to findings reported in colon cancer. Data from the PETACC-3 trial47 demonstrated that MSI-H/dMMR is more common in stage II colon cancer than in stage III disease (22% vs. 12%; P < 0.0001), and stage IV tumors with MSI-H/dMMR account for the lowest proportion (only 3.5%).48
Given that the proportion of MSI-H/dMMR is generally higher in resectable GC than in advanced-stage GC, it is necessary to evaluate the MSI status of each patient. Nonetheless, it remains unclear whether adjuvant chemotherapy is the optimal therapeutic strategy for patients with resectable GC with MSI-H/dMMR after surgery. Regarding the role of MSI-H/dMMR in predicting chemotherapy efficacy in resectable GC, a growing literature suggests a lack of benefit of 5-FU chemotherapy in patients with MSI-H/dMMR, although inconsistent findings have been reported. The mechanisms involved have not yet been elucidated. One significant hypothesis relates to a high tumor mutation burden (TMB-H), which suggests that the increased presence of T cells in MSI-H/dMMR tumors can be stimulated to fight against cancer, leading to a favorable prognosis. Following chemotherapy, immunosuppressive therapy is commonly administered, which contributes to a reduction in tumor-infiltrating lymphocytes (TILs) and consequently promotes resistance to chemotherapy.70,71 Furthermore, an in vitro study by Tsai and colleagues suggested that the activation of autophagy could induce MSI-H/dMMR gastric cancer resistance against 5-FU.36 These hypotheses for resistance mechanisms to the chemotherapeutic agents have been validated in the effects of adjuvant chemotherapy on MSI-H/dMMR colon cancer. Sargent et al. carried out a retrospective study and illustrated that treatment was associated with reduced overall survival in patients with stage II disease and with MSI-H/dMMR tumors.72 Another retrospective study involving patients with stage II and III with MSI-H/dMMR tumor status indicated that patients with tumors exhibiting microsatellite stable (MSS) or MSI-L tended to benefit from fluorouracil-based adjuvant chemotherapy [HR 0.72 (0.53–0.99)], while worse outcomes were observed among patients with MSI-H [HR 2.14 (0.83–5.49)].73 Taken together, the above findings suggest that chemotherapy does not represent the optimal approach to adjuvant therapy. Furthermore, tumors with MSI-H/dMMR are susceptible to immunotherapy because of infiltrative immune cells and high tumor burdens. The clinical guidelines published by the Chinese Society of Clinical Oncology17 for diagnosing and treating gastric cancer recommend that adjuvant chemotherapy should not be encouraged after radical surgery in MSI-H patients. Instead, these patients should be observed or undergo immunotherapy in clinical trials. Prospective clinical trials such as KEYNOTE059,74 KEYNOTE061,75 and KEYNOTE06276 demonstrated an effective clinical response in patients with advanced MSI-H/dMMR gastric cancer. Accordingly, the FDA approved the application of pembrolizumab and dotalizumab. On the basis of these results, the addition of PD-1 inhibitors to adjuvant therapy may be superior to chemotherapy alone for patients with resectable MSI-H/dMMR GC after radical surgery.
In addition, for patients with stage II GC with the highest prevalence of MSI-H/dMMR, whether neoadjuvant is needed depends on the experience of clinicians, with no theoretical basis currently available. More individualized treatment strategies could be provided to clinicians by exploring the predictive effect of MSI-H/dMMR on the efficacy of neoadjuvant therapy on resectable gastric cancer. In the era of conventional chemotherapy, a relatively large cohort study from New York was performed in 202246 to examine the association between MSI-H/dMMR and survival in patients with resectable gastric cancer receiving chemotherapy, including neoadjuvant, adjuvant, and in combination with radiotherapy and compared with patients treated with surgery alone. The 3 year OS and DFS rates were higher among patients treated with surgery alone, though a survival difference was not observed between patients with neoadjuvant or perioperative chemotherapy and those without (88% vs. 79%, P = 0.48 and 78% vs. 73%, P = 0.66). It was found that MSI-H/dMMR is associated with a positive prognosis in patients treated with surgery alone and a negative prognosis in patients treated with chemotherapy. These findings suggested that chemotherapy alone exhibited limited efficacy in enhancing surgery’s overall efficacy and improving survival outcomes. With significant inroads achieved in immunotherapy, the development of immune preparations has been accompanied by constant attempts to apply them in neoadjuvant regimes, followed by documentation of cases of gastrointestinal cancer to demonstrate their feasibility. Moreover, preliminary evidence of the predictive value of MSI-H/dMMR on neoadjuvant immunotherapy has been obtained from clinical trials, corroborating that neoadjuvant chemotherapy involving PD-1 inhibitors yields better advantages. With the advent of neoadjuvant immunotherapy, it becomes possible to broaden the scope of applicable neoadjuvant treatments, thereby offering a survival benefit to a greater number of patients.
Conclusions
Although the past decade has witnessed unprecedented medical progress, gastric cancer remains an important public health issue. The significant increase in studies published on molecular typing has paved the way to address specific therapeutic strategies. The systematic classifications of GC, such as TCGA and ACRG, have substantiated the important value of the MSI genotype. Significant heterogeneity in the frequency of MSI-H/dMMR among resectable GCs has been found in our literature review, mainly attributed to the heterogeneity in geographic distribution and pathological stages. In the meantime, although the limited studies in the adjuvant treatment of MSI-H/dMMR gastric cancers have been deemed inadequate to determine an explicit treatment regimen after surgery, most studies substantiate the low chemosensitivity of MSI-H/dMMR patients. Additionally, as the treatment schemes of immunotherapy in advanced GCs have been guideline endorsed, their value in adjuvant and neoadjuvant settings has been emphasized. Despite the small number of patients with MSI GC enrolled in the available RCTs and the lacking of prospective studies, our review aimed to point out that better responses might occur if immunotherapy is offered earlier for patients with MSI-H/dMMR gastric cancer. This promising area requires further study, which will hopefully shed more light on the optimal clinical regimen to improve the outcomes of this patient population.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
National comprehensive cancer network: NCCN guidelines: gastric cancer, version 2.2022. https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf.
Wilke H, Muro K, Van Cutsem E, Oh SC, Bodoky G, Shimada Y, et al. Ramucirumab plus paclitaxel vs. placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol. 2014;15(11):1224–35.
Fuchs CS, Tomasek J, Yong CJ, Dumitru F, Passalacqua R, Goswami C, et al. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2014;383(9911):31–9.
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513(7517):202–9.
Cristescu R, Lee J, Nebozhyn M, Kim KM, Ting JC, Wong SS, et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat Med. 2015;21(5):449–56.
Lei Z, Tan IB, Das K, Deng N, Zouridis H, Pattison S, et al. Identification of molecular subtypes of gastric cancer with different responses to PI3-kinase inhibitors and 5-fluorouracil. Gastroenterology. 2013;145(3):554–65.
Baretti M, Le DT. DNA mismatch repair in cancer. Pharmacol Ther. 2018;189:45–62.
Falchetti M, Saieva C, Lupi R, Masala G, Rizzolo P, Zanna I, et al. Gastric cancer with high-level microsatellite instability: target gene mutations, clinicopathologic features, and long-term survival. Hum Pathol. 2008;39(6):925–32.
Halling KC, Harper J, Moskaluk CA, Thibodeau SN, Petroni GR, Yustein AS, et al. Origin of microsatellite instability in gastric cancer. Am J Pathol. 1999;155(1):205–11.
Yuza K, Nagahashi M, Watanabe S, Takabe K, Wakai T. Hypermutation and microsatellite instability in gastrointestinal cancers. Oncotarget. 2017;8(67):112103–15.
Kim HS, Lee BL, Woo DK, Bae SI, Kim WH. Assessment of markers for the identification of microsatellite instability phenotype in gastric neoplasms. Cancer Lett. 2001;164(1):61–8.
Smyth EC, Wotherspoon A, Peckitt C, Gonzalez D, Hulkki-Wilson S, Eltahir Z, et al. Mismatch repair deficiency, microsatellite instability, and survival: an exploratory analysis of the medical research council adjuvant gastric infusional chemotherapy (MAGIC) trial. JAMA Oncol. 2017;3(9):1197–203.
Marrelli D, Polom K, Pascale V, Vindigni C, Piagnerelli R, De Franco L, et al. Strong prognostic value of microsatellite instability in intestinal type non-cardia gastric cancer. Ann Surg Oncol. 2016;23(3):943–50.
An JY, Kim H, Cheong JH, Hyung WJ, Kim H, Noh SH. Microsatellite instability in sporadic gastric cancer: its prognostic role and guidance for 5-FU based chemotherapy after R0 resection. Int J Cancer. 2012;131(2):505–11.
National Comprehensive Cancer Network: NCCN Guidelines: Colon Cancer, Version 1.2022.https://www.nccn.org/professionals/physician_gls/pdf/colon.pdf.
Wang FH, Zhang XT, Li YF, Tang L, Qu XJ, Ying JE, et al. The Chinese society of clinical oncology (CSCO): clinical guidelines for the diagnosis and treatment of gastric cancer, 2021. Cancer Commun (Lond). 2021;41(8):747–95.
Vincenzi B, Imperatori M, Silletta M, Marrucci E, Santini D, Tonini G. Emerging kinase inhibitors of the treatment of gastric cancer. Expert Opin Emerg Drugs. 2015;20(3):479–93.
Oki E, Kakeji Y, Zhao Y, Yoshida R, Ando K, Masuda T, et al. Chemosensitivity and survival in gastric cancer patients with microsatellite instability. Ann Surg Oncol. 2009;16(9):2510–5.
Bacani J, Zwingerman R, Di Nicola N, Spencer S, Wegrynowski T, Mitchell K, et al. Tumor microsatellite instability in early onset gastric cancer. J Mol Diagn. 2005;7(4):465–77.
Kim JY, Shin NR, Kim A, Lee HJ, Park WY, Kim JY, et al. Microsatellite instability status in gastric cancer: a reappraisal of its clinical significance and relationship with mucin phenotypes. Korean J Pathol. 2013;47(1):28–35.
Kim H, An JY, Noh SH, Shin SK, Lee YC, Kim H. High microsatellite instability predicts good prognosis in intestinal-type gastric cancers. J Gastroenterol Hepatol. 2011;26(3):585–92.
Seo HM, Chang YS, Joo SH, Kim YW, Park YK, Hong SW, et al. Clinicopathologic characteristics and outcomes of gastric cancers with the MSI-H phenotype. J Surg Oncol. 2009;99(3):143–7.
Choi YY, Kim H, Shin SJ, Kim HY, Lee J, Yang HK, et al. Microsatellite instability and programmed cell death-ligand 1 expression in stage II/III gastric cancer: post hoc analysis of the CLASSIC randomized controlled study. Ann Surg. 2019;270(2):309–16.
Kim JW, Cho SY, Chae J, Kim JW, Kim TY, Lee KW, et al. Adjuvant chemotherapy in microsatellite instability-high gastric cancer. Cancer Res Treat. 2020;52(4):1178–87.
Choi HS, Lee SY, Kim JH, Sung IK, Park HS, Shim CS, et al. Low prevalence of microsatellite instability in interval gastric cancers. Dig Dis Sci. 2014;59(2):322–7.
Choi J, Nam SK, Park DJ, Kim HW, Kim HH, Kim WH, et al. Correlation between microsatellite instability-high phenotype and occult lymph node metastasis in gastric carcinoma. Apmis. 2015;123(3):215–22.
Kim KJ, Lee TH, Cho NY, Yang HK, Kim WH, Kang GH. Differential clinicopathologic features in microsatellite-unstable gastric cancers with and without MLH1 methylation. Hum Pathol. 2013;44(6):1055–64.
Cho J, Kang SY, Kim KM. MMR protein immunohistochemistry and microsatellite instability in gastric cancers. Pathology. 2019;51(1):110–3.
Zhao Y, Zheng ZC, Luo YH, Piao HZ, Zheng GL, Shi JY, et al. Low-frequency microsatellite instability in genomic di-nucleotide sequences correlates with lymphatic invasion and poor prognosis in gastric cancer. Cell Biochem Biophys. 2015;71(1):235–41.
Cai L, Sun Y, Wang K, Guan W, Yue J, Li J, et al. The better survival of MSI subtype is associated with the oxidative stress related pathways in gastric cancer. Front Oncol. 2020;10:1269.
Zhang L, Wang Y, Li Z, Lin D, Liu Y, Zhou L, et al. Clinicopathological features of tumor mutation burden, Epstein-Barr virus infection, microsatellite instability and PD-L1 status in Chinese patients with gastric cancer. Diagn Pathol. 2021;16(1):38.
Wang XY, Hu YJ, Dong K, Zhao C, Huang XZ, Lian SY, et al. Expression level and prognostic value of PD-L1 in microsatellite instability-high gastric cancer. Zhonghua Bing Li Xue Za Zhi. 2020;49(11):1114–9.
Fang WL, Chang SC, Lan YT, Huang KH, Chen JH, Lo SS, et al. Microsatellite instability is associated with a better prognosis for gastric cancer patients after curative surgery. World J Surg. 2012;36(9):2131–8.
Fang WL, Chang SC, Lan YT, Huang KH, Lo SS, Li AF, et al. Molecular and survival differences between familial and sporadic gastric cancers. Biomed Res Int. 2013;2013:396272.
Tsai CY, Lin TA, Huang SC, Hsu JT, Yeh CN, Chen TC, et al. Is adjuvant chemotherapy necessary for patients with deficient mismatch repair gastric cancer?-Autophagy inhibition matches the mismatched. Oncologist. 2020;25(7):e1021–30.
Sakurai M, Zhao Y, Oki E, Kakeji Y, Oda S, Maehara Y. High-resolution fluorescent analysis of microsatellite instability in gastric cancer. Eur J Gastroenterol Hepatol. 2007;19(8):701–9.
Hewitt LC, Inam IZ, Saito Y, Yoshikawa T, Quaas A, Hoelscher A, et al. Epstein-Barr virus and mismatch repair deficiency status differ between oesophageal and gastric cancer: a large multi-centre study. Eur J Cancer. 2018;94:104–14.
Solcia E, Klersy C, Mastracci L, Alberizzi P, Candusso ME, Diegoli M, et al. A combined histologic and molecular approach identifies three groups of gastric cancer with different prognosis. Virchows Arch. 2009;455(3):197–211.
Wirtz HC, Müller W, Noguchi T, Scheven M, Rüschoff J, Hommel G, et al. Prognostic value and clinicopathological profile of microsatellite instability in gastric cancer. Clin Cancer Res. 1998;4(7):1749–54.
Keller G, Rudelius M, Vogelsang H, Grimm V, Wilhelm MG, Mueller J, et al. Microsatellite instability and loss of heterozygosity in gastric carcinoma in comparison to family history. Am J Pathol. 1998;152(5):1281–9.
Kohlruss M, Grosser B, Krenauer M, Slotta-Huspenina J, Jesinghaus M, Blank S, et al. Prognostic implication of molecular subtypes and response to neoadjuvant chemotherapy in 760 gastric carcinomas: role of Epstein-Barr virus infection and high- and low-microsatellite instability. J Pathol Clin Res. 2019;5(4):227–39.
dos Santos NR, Seruca R, Constância M, Seixas M, Sobrinho-Simões M. Microsatellite instability at multiple loci in gastric carcinoma: clinicopathologic implications and prognosis. Gastroenterology. 1996;110(1):38–44.
Schneider BG, Bravo JC, Roa JC, Roa I, Kim MC, Lee KM, et al. Microsatellite instability, prognosis and metastasis in gastric cancers from a low-risk population. Int J Cancer. 2000;89(5):444–52.
Hause RJ, Pritchard CC, Shendure J, Salipante SJ. Classification and characterization of microsatellite instability across 18 cancer types. Nat Med. 2016;22(11):1342–50.
Vos EL, Maron SB, Krell RW, Nakauchi M, Fiasconaro M, Capanu M, et al. Survival of locally advanced msi-high gastric cancer patients treated with perioperative chemotherapy: a retrospective cohort study. Ann Surg. 2022;277:798.
Klingbiel D, Saridaki Z, Roth AD, Bosman FT, Delorenzi M, Tejpar S. Prognosis of stage II and III colon cancer treated with adjuvant 5-fluorouracil or FOLFIRI in relation to microsatellite status: results of the PETACC-3 trial. Ann Oncol. 2015;26(1):126–32.
Koopman M, Kortman GA, Mekenkamp L, Ligtenberg MJ, Hoogerbrugge N, Antonini NF, et al. Deficient mismatch repair system in patients with sporadic advanced colorectal cancer. Br J Cancer. 2009;100(2):266–73.
Pietrantonio F, Miceli R, Raimondi A, Kim YW, Kang WK, Langley RE, et al. Individual patient data meta-analysis of the value of microsatellite instability as a biomarker in gastric cancer. J Clin Oncol. 2019;37(35):3392–400.
Wang JB, Li P, Liu XL, Zheng QL, Ma YB, Zhao YJ, et al. An immune checkpoint score system for prognostic evaluation and adjuvant chemotherapy selection in gastric cancer. Nat Commun. 2020;11(1):6352.
Yang N, Wu Y, Jin M, Jia Z, Wang Y, Cao D, et al. Microsatellite instability and Epstein-Barr virus combined with PD-L1 could serve as a potential strategy for predicting the prognosis and efficacy of postoperative chemotherapy in gastric cancer. Peer J. 2021;9:e11481.
Nie RC, Chen GM, Yuan SQ, Kim JW, Zhou J, Nie M, et al. Adjuvant chemotherapy for gastric cancer patients with mismatch repair deficiency or microsatellite instability: systematic review and meta-analysis (Nov 2021). Ann Surg Oncol. 2022;29(5):3193. https://doi.org/10.1245/S10434-021-11050-6.
赵福星. 胃癌微卫星不稳定与氟尿嘧啶为基础辅助化疗疗效的关系: 青海大学; 2020.5.
Janjigian YY, Shitara K, Moehler M, Garrido M, Salman P, Shen L, et al. First-line nivolumab plus chemotherapy vs. chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021;398(10294):27–40.
LBA53 - Sintilimab plus chemotherapy (chemo) vs. chemo as first-line treatment for advanced gastric or gastroesophageal junction (G/GEJ) adenocarcinoma (ORIENT-16): first results of a randomized, double-blind, phase III study. https://oncologypro.esmo.org/meeting-resources/esmo-congress-2021/sintilimab-plus-chemotherapy-chemo-vs.-chemo-as-first-line-treatment-for-advanced-gastric-or-gastroesophageal-junction-g-gej-adenocarcinoma.
Felip E, Altorki N, Zhou C, Csőszi T, Vynnychenko I, Goloborodko O, et al. Adjuvant atezolizumab after adjuvant chemotherapy in resected stage IB-IIIA non-small-cell lung cancer (IMpower010): a randomised, multicentre, open-label, phase 3 trial. Lancet. 2021;398(10308):1344–57.
Zhang Z, Cheng S, Gong J, Lu M, Zhou J, Zhang X, et al. Efficacy and safety of neoadjuvant immunotherapy in patients with microsatellite instability-high gastrointestinal malignancies: a case series. Eur J Surg Oncol. 2020;46(10 Pt B):e33–9.
Neoadjuvant nivolumab i, and celecoxib in MMR-proficient and MMR-deficient colon cancers: final clinical analysis of the NICHE study. doi https://doi.org/10.1200/JCO.2022.40.16_suppl.3511.
André T, Tougeron D, Piessen G, de la Fouchardière C, Louvet C, Adenis A, et al. Neoadjuvant nivolumab plus ipilimumab and adjuvant nivolumab in localized deficient mismatch repair/microsatellite instability-high gastric or esophagogastric junction adenocarcinoma: the GERCOR NEONIPIGA phase II study. J Clin Oncol. 2023;41(2):255–65.
Raimondi A, Palermo F, Prisciandaro M, Aglietta M, Antonuzzo L, Aprile G, et al. TremelImumab and durvalumab combination for the non-operative management (NOM) of microsatellite instabiliTY (MSI)-high resectable gastric or gastroesophageal junction cancer: the multicentre, single-arm, multi-cohort, phase II INFINITY study. Cancers (Basel). 2021;13(11):2839.
Pietrantonio F, Raimondi A, Lonardi S, Murgioni S, Cardellino GG, Tamberi S, et al. INFINITY: a multicentre, single-arm, multi-cohort, phase II trial of tremelimumab and durvalumab as neoadjuvant treatment of patients with microsatellite instability-high (MSI) resectable gastric or gastroesophageal junction adenocarcinoma (GAC/GEJAC). J Clin Oncol. 2023;41(4_suppl):358.
Chubenko V, Inusilaev G, Imyanitov E, Moiseyenko V. Clinical case of the neoadjuvant treatment with nivolumab in a patient with microsatellite unstable (MSI-H) locally advanced gastric cancer. BMJ Case Rep. 2020;13(9):e236144.
Nishimoto IN, Hanaoka T, Sugimura H, Nagura K, Ihara M, Li XJ, et al. Cytochrome P450 2E1 polymorphism in gastric cancer in Brazil: case-control studies of Japanese Brazilians and non-Japanese Brazilians. Cancer Epidemiol Biomarkers Prev. 2000;9(7):675–80.
Setiawan VW, Zhang ZF, Yu GP, Li YL, Lu ML, Tsai CJ, et al. GSTT1 and GSTM1 null genotypes and the risk of gastric cancer: a case-control study in a Chinese population. Cancer Epidemiol Biomarkers Prev. 2000;9(1):73–80.
Harada S, Misawa S, Nakamura T, Tanaka N, Ueno E, Nozoe M. Detection of GST1 gene deletion by the polymerase chain reaction and its possible correlation with stomach cancer in Japanese. Hum Genet. 1992;90(1–2):62–4.
Nan HM, Song YJ, Yun HY, Park JS, Kim H. Effects of dietary intake and genetic factors on hypermethylation of the hMLH1 gene promoter in gastric cancer. World J Gastroenterol. 2005;11(25):3834–41.
Palli D, Russo A, Saieva C, Salvini S, Amorosi A, Decarli A. Dietary and familial determinants of 10-year survival among patients with gastric carcinoma. Cancer. 2000;89(6):1205–13.
Palli D, Russo A, Ottini L, Masala G, Saieva C, Amorosi A, et al. Red meat, family history, and increased risk of gastric cancer with microsatellite instability. Cancer Res. 2001;61(14):5415–9.
Alcohol use and burden for 195 countries and territories, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 392(10152):1015-35.
Lee HE, Chae SW, Lee YJ, Kim MA, Lee HS, Lee BL, et al. Prognostic implications of type and density of tumour-infiltrating lymphocytes in gastric cancer. Br J Cancer. 2008;99(10):1704–11.
Phillips SM, Banerjea A, Feakins R, Li SR, Bustin SA, Dorudi S. Tumour-infiltrating lymphocytes in colorectal cancer with microsatellite instability are activated and cytotoxic. Br J Surg. 2004;91(4):469–75.
Sargent DJ, Marsoni S, Monges G, Thibodeau SN, Labianca R, Hamilton SR, et al. Defective mismatch repair as a predictive marker for lack of efficacy of fluorouracil-based adjuvant therapy in colon cancer. J Clin Oncol. 2010;28(20):3219–26.
Ribic CM, Sargent DJ, Moore MJ, Thibodeau SN, French AJ, Goldberg RM, et al. Tumor microsatellite-instability status as a predictor of benefit from fluorouracil-based adjuvant chemotherapy for colon cancer. N Engl J Med. 2003;349(3):247–57.
Fuchs CS, Doi T, Jang RW, Muro K, Satoh T, Machado M, et al. Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 clinical KEYNOTE-059 trial. JAMA Oncol. 2018;4(5):e180013.
Shitara K, Özgüroğlu M, Bang YJ, Di Bartolomeo M, Mandalà M, Ryu MH, et al. Pembrolizumab vs. paclitaxel for previously treated, advanced gastric or gastro-oesophageal junction cancer (KEYNOTE-061): a randomised, open-label, controlled, phase 3 trial. Lancet. 2018;392(10142):123–33.
Shitara K, Van Cutsem E, Bang YJ, Fuchs C, Wyrwicz L, Lee KW, et al. Efficacy and safety of pembrolizumab or pembrolizumab plus chemotherapy vs chemotherapy alone for patients with first-line, advanced gastric cancer: the KEYNOTE-062 phase 3 randomized clinical trial. JAMA Oncol. 2020;6(10):1571–80.
Funding
This work was supported by Natural Science Foundation of Zhejiang Province (Grant No.: LQ19H160031).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The ideas for the review were performed by HJ and YD The literature search, data analysis, and the first draft of the manuscript were written by HW and WM, and YD critically revised the work. All authors commented on previous versions of the manuscript and approved the final manuscript.
Corresponding authors
Ethics declarations
DISCLOSURE
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wu, H., Ma, W., Jiang, C. et al. Heterogeneity and Adjuvant Therapeutic Approaches in MSI-H/dMMR Resectable Gastric Cancer: Emerging Trends in Immunotherapy. Ann Surg Oncol 30, 8572–8587 (2023). https://doi.org/10.1245/s10434-023-14103-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-023-14103-0