Abstract
Background
Although patients with resectable colorectal liver metastasis (CLM), a population with good prognosis, have been treated with upfront surgery, some patients have had a poor prognosis. This study aimed to investigate biologic prognostic factors in patients with resectable CLMs.
Methods
This single-center retrospective study enrolled consecutive patients who underwent liver resection for initial CLMs at the Cancer Institute Hospital between 2010 and 2020. The study defined CLMs as resectable (tumor size < 5 cm; < 4 tumors; no extrahepatic metastasis) or borderline resectable (BR). Preoperative chemotherapy was administered to patients with BR CLMs.
Results
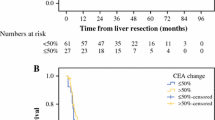
During the study period, 309 CLMs were classified as resectable without preoperative chemotherapy and 345 as BR with preoperative chemotherapy. For the 309 patients with resectable CLMs, the independent poor prognostic factors associated with overall survival in the multivariable analysis were high tumor marker levels (CEA ≥ 25 ng/mL and/or CA19-9 ≥ 50 U/mL; (hazard ratio [HR], 2.45; p = 0.0007), no adjuvant chemotherapy (HR, 1.69; p = 0.043), and age of 75 years or older (HR, 2.09; p = 0.012). The 5-year survival rates for the patients with high tumor marker (TM) levels (CEA ≥25 ng/mL and/or CA19-9 ≥50 U/mL) were significantly worse than for those with low TM levels (CEA < 25 ng/mL and CA19-9 < 50 U/mL) (55.3% vs. 81.1%; p <0.0001) and similar to the rate for those with BR CLMs (52.1%; p = 0.864). Postoperative adjuvant chemotherapy had an impact on prognosis only in the high-TM group (HR, 2.65; p = 0.007).
Conclusions
High TM levels have a prognostic impact on patients with resectable CLMs stratified by tumor number and size. Perioperative chemotherapy improves long-term outcomes for patients with CLM and high TM levels.



Similar content being viewed by others
References
Adam R, De Gramont A, Figueras J, Guthrie A, Kokudo N, Kunstlinger F, et al. The oncosurgery approach to managing liver metastases from colorectal cancer: a multidisciplinary international consensus. Oncologist. 2012;17:1225–39.
Kopetz S, Chang GJ, Overman MJ, Eng C, Sargent DJ, Larson DW, et al. Improved survival in metastatic colorectal cancer is associated with adoption of hepatic resection and improved chemotherapy. J Clin Oncol. 2009;27:3677–83.
Adam R, Delvart V, Pascal G, Valeanu A, Castaing D, Azoulay D, et al. Rescue surgery for unresectable colorectal liver metastases downstaged by chemotherapy: a model to predict long-term survival. Ann Surg. 2004;4(6):44–57.https://doi.org/10.1097/01.sla.0000141198.92114.f6
Adam R, Wicherts DA, de Haas RJ, Ciacio O, Levi F, Paule B, et al. Patients with initially unresectable colorectal liver metastases: is there a possibility of cure? J Clin Oncol. 2009;11:1829–35.
Kobayashi K, Inoue Y, Kitano Y, Sato S, Oba A, Ono Y, et al. Optimizing the selection of technically unresectable colorectal liver metastases. Surgery. 2023;73(2):442–9.
Imai K, Benitez CC, Allard MA, Vibert E, Cunha AS, Cherqui D, et al. Impact of surgical treatment for recurrence after 2-stage hepatectomy for colorectal liver metastases, on patient outcome. Ann Surg. 2019;269:322–30.https://doi.org/10.1097/SLA.0000000000002472
Nordlinger B, Sorbye H, Glimelius B, Poston GJ, Schlag PM, Rougier P, et al. Perioperative chemotherapy with FOLFOX4 and surgery versus surgery alone for resectable liver metastases from colorectal cancer (EORTC Intergroup trial 40983): a randomised controlled trial. Lancet. 2008;371:1007–16.
Primrose J, Falk S, Finch-Jones M, Valle J, O’Reilly D, Siriwardena A, Hornbuckle J, Peterson M, Rees M, Iveson T, Hickish T. Systemic chemotherapy with or without cetuximab in patients with resectable colorectal liver metastasis: the New EPOC randomised controlled trial. Lancet Oncol. 2014;15(6):601–11.
Vauthey JN, Pawlik TM, Ribero D, Wu TT, Zorzi D, Hoff PM, et al. Chemotherapy regimen predicts steatohepatitis and an increase in 90-day mortality after surgery for hepatic colorectal metastases. J Clin Oncol. 2006;24:2065–72.
Tamandl D, Klinger M, Eipeldauer S, Herberger B, Kaczirek K, Gruenberger B, et al. Sinusoidal obstruction syndrome impairs long-term outcome of colorectal liver metastases treated with resection after neoadjuvant chemotherapy. Ann Surg Oncol. 2011;18:421–30.https://doi.org/10.1245/s10434-010-1317-4
Shindoh J, Tzeng CW, Aloia TA, Curley SA, Zimmitti G, Wei SH, et al. Optimal future liver remnant in patients treated with extensive preoperative chemotherapy for colorectal liver metastases. Ann Surg Oncol. 2013;20:2493–500.https://doi.org/10.1245/s10434-012-2864-7
Vauthey JN. Colorectal liver metastases: treat effectively up front and consider the borderline resectable. J Clin Oncol. 2007;25:4524–5.
Jones RP, Malik HZ, Fenwick SW, Poston GJ. Perioperative chemotherapy for resectable colorectal liver metastases: where now? Eur J Surg Oncol. 2013;39:807–11.
Saiura A, Yamamoto J, Hasegawa K, Koga R, Sakamoto Y, Hata S, et al. Liver resection for multiple colorectal liver metastases with surgery up-front approach: bi-institutional analysis of 736 consecutive cases. World J Surg. 2012;36:2171–8.
Ichida H, Mise Y, Ito H, Ishizawa T, Inoue Y, Takahashi Y, et al. Optimal indication criteria for neoadjuvant chemotherapy in patients with resectable colorectal liver metastases. World J Surg Oncol. 2019;17:100.
Brudvik KW, Kopetz SE, Li L, Conrad C, Aloia TA, Vauthey JN. Meta-analysis of KRAS mutations and survival after resection of colorectal liver metastases. Br J Surg. 2015;102:1175–83.
Arita J, Ono Y, Takahashi M, Inoue Y, Takahashi Y, Saiura A. Usefulness of contrast-enhanced intraoperative ultrasound in identifying disappearing liver metastases from colorectal carcinoma after chemotherapy. Ann Surg Oncol. 2014;21(3):S390–7.https://doi.org/10.1245/s10434-014-3576-y
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13.https://doi.org/10.1097/01.sla.0000133083.54934.ae
Strasberg SM, Phillips C. Use and dissemination of the brisbane 2000 nomenclature of liver anatomy and resections. Ann Surg. 2013;257:377–82.https://doi.org/10.1097/SLA.0b013e31825a01f6
Bonney GK, Coldham C, Adam R, Kaiser G, Barroso E, Capussotti L, et al. Role of neoadjuvant chemotherapy in resectable synchronous colorectal liver metastasis: an international multi-center data analysis using LiverMetSurvey. J Surg Oncol. 2015;111:716–24.
Ayez N, van der Stok EP, Grünhagen DJ, Rothbarth J, van Meerten E, Eggermont AM, et al. The use of neo-adjuvant chemotherapy in patients with resectable colorectal liver metastases: clinical risk score as possible discriminator. Eur J Surg Oncol. 2015;41:859–67.
Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg. 1999;3(3):09–18.https://doi.org/10.1097/00000658-199909000-00004
Beppu T, Sakamoto Y, Hasegawa K, Honda G, Tanaka K, Kotera Y, et al. A nomogram predicting disease-free survival in patients with colorectal liver metastases treated with hepatic resection: multicenter data collection as a project study for hepatic surgery of the Japanese society of Hepato-biliary-pancreatic surgery. J Hepatobiliary Pancreat Sci. 2012;19:72–84.
Brudvik KW, Jones RP, Giuliante F, Shindoh J, Passot G, Chung MH, et al. RAS mutation clinical risk score to predict survival after resection of colorectal liver metastases. Ann Surg. 2019;69(1):120–6.https://doi.org/10.1097/SLA.0000000000002319
Takeda Y, Mise Y, Takahashi Y, Ito H, Inoue Y, Yoshioka R, et al. Limited prognostic value of KRAS in patients undergoing hepatectomy for colorectal liver metastases. Ann Surg Oncol. 2022;29:2383–91.https://doi.org/10.1245/s10434-021-11015-9
Lu Z, Peng J, Wang Z, Pan Z, Yuan Y, Wan D, et al. High preoperative serum CA19-9 level is predictive of poor prognosis for patients with colorectal liver oligometastases undergoing hepatic resection. Med Oncol. 2016;3(11):121.
Kanemitsu Y, Shimizu Y, Mizusawa J, Inaba Y, Hamaguchi T, Shida D, et al. Hepatectomy followed by mFOLFOX6 versus hepatectomy alone for liver-only metastatic colorectal cancer (JCOG0603): a phase II or III randomized controlled trial. J Clin Oncol. 2021;39:3789–99.
Hasegawa K, Saiura A, Takayama T, Miyagawa S, Yamamoto J, Ijichi M, et al. Adjuvant oral uracil-tegafur with leucovorin for colorectal cancer liver metastases: a randomized controlled trial. PLoS One. 2016;11:e0162400.
Funding
No specific grants from funding agencies in the public, commercial, or not for profit sectors were received to fund this study.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosures
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10434_2023_13781_MOESM3_ESM.jpg
Longterm outcomes of resectable colorectal liver metastases. The 3year and 5 year overall survival, time to surgical failure, and recurrence free survival were 82.9% and 73.9%, 68.5%and 64.8%, and 52.3% and 48.9% respectively. (JPG 186 kb)
10434_2023_13781_MOESM4_ESM.jpg
Overall survival in high and low tumor marker groups of resectable colorectal livermetastases and borderline resectable colorectal liver metastases. The 5year overall survival in patients with resectable colorectal liver metastases with high tumor marker levels(55.3%) was similar to those with borderline resectable colorectal liver metastases (52.1%) (p = 0.864) (JPG 326 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kobayashi, K., Ono, Y., Kitano, Y. et al. Prognostic Impact of Tumor Markers (CEA and CA19-9) on Patients with Resectable Colorectal Liver Metastases Stratified by Tumor Number and Size: Potentially Valuable Biologic Markers for Preoperative Treatment. Ann Surg Oncol 30, 7338–7347 (2023). https://doi.org/10.1245/s10434-023-13781-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-023-13781-0