Abstract
Background
Combining liver resection (LR) with radiofrequency ablation (RFA) is nowadays an accepted option for treating colorectal liver metastases (CRLMs), but the number of lesions ablated is regularly described as a recurrence risk factor. In this study, we report our experience and determine the impact of RFA on long-term outcomes.
Method
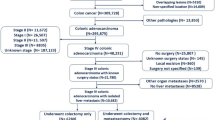
This is a retrospective study including patients undergoing LR with or without RFA for CRLM. All variables influencing disease-free survival (DFS) and disease-specific survival (DSS) were examined through a Cox regression analysis before and after propensity-score matching (PSM).
Results
Among the 128 patients included, 71 (55.5%) underwent LR alone and 57 (44.5%) underwent LR+RFA. With univariate analysis, LR+RFA showed a significantly worse DFS than LR alone (p = 0.028), which was not confirmed after PSM (p = 0.064). Thermal ablation did not influence DSS before or after matching (p = 0.282 and p = 0.189). When analyzing the subgroups of patients according to number of RFAs performed, no difference in long-term outcomes was observed (after PSM: p = 0.192 for DFS and p = 0.624 for DSS). Analysis of site of recurrence revealed that neither performing an RFA (p = 0.893) nor the number of lesions ablated (p = 0.093, p = 0.550, and p = 0.087 for 1, 2, and ≥ 2 RFAs) were associated with an increased risk of liver-only relapse.
Discussion
In the setting of a parenchymal sparing strategy, combining RFA with LR is safe in terms of oncological outcomes. Tumor burden, rather than RFA performed, independently influences risk of recurrence and patient survival.

Similar content being viewed by others
References
Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49. https://doi.org/10.3322/caac.21660.
Surveillance, epidemiology, and end results (SEER) program (www.seer.cancer.gov) SEER*stat database: colon and rectum. SEER 5-Year Relative Survival Rates, 2012-2018. Accessed 20 December 2022.
Manfredi S, Lepage C, Hatem C, Coatmeur O, Faivre J, Bouvier AM. Epidemiology and management of liver metastases from colorectal cancer. Ann Surg. 2006;244(2):254–9. https://doi.org/10.1097/01.sla.0000217629.94941.cf.
Hackl C, Neumann P, Gerken M, Loss M, Klinkhammer-Schalke M, Schlitt HJ. Treatment of colorectal liver metastases in Germany: a ten-year population-based analysis of 5772 cases of primary colorectal adenocarcinoma. BMC Cancer. 2014;14:810. https://doi.org/10.1186/1471-2407-14-810.
Valls C, Ramos E, Leiva D, Ruiz S, Martinez L, Rafecas A. Safety and efficacy of ultrasound-guided radiofrequency ablation of recurrent colorectal cancer liver metastases after hepatectomy. Scand J Surg SJS Off Organ Finn Surg Soc Scand Surg Soc. 2015;104(3):169–75. https://doi.org/10.1177/1457496914553147.
Mimmo A, Pegoraro F, Rhaiem R, et al. Microwave ablation for colorectal liver metastases: a systematic review and pooled oncological analyses. Cancers. 2022;14(5):1305. https://doi.org/10.3390/cancers14051305.
van de Geest TW, van Amerongen MJ, Nierop PMH, et al. Propensity score matching demonstrates similar results for radiofrequency ablation compared to surgical resection in colorectal liver metastases. Eur J Surg Oncol. 2022;48(6):1368–74. https://doi.org/10.1016/j.ejso.2022.01.008.
Masuda T, Margonis GA, Andreatos N, et al. Combined hepatic resection and radio-frequency ablation for patients with colorectal cancer liver metastasis: a viable option for patients with a large number of tumors. Anticancer Res. 2018;38(11):6353–60. https://doi.org/10.21873/anticanres.12993.
Solbiati M, Ierace T, Muglia R, et al. Thermal ablation of liver tumors guided by augmented reality: an initial clinical experience. Cancers. 2022;14(5):1312. https://doi.org/10.3390/cancers14051312.
von Elm E, Altman DG, Egger M, et al. The strengthening of the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Int J Surg Lond Engl. 2014;12(12):1495–9. https://doi.org/10.1016/j.ijsu.2014.07.013.
Amin M, Edge S, Greene F, et al. AJCC Cancer Staging Manual (8th Edition). American Joint Commission on Cancer: Springer International Publishing; 2017.
Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250(2):187–96. https://doi.org/10.1097/SLA.0b013e3181b13ca2.
Ivey GD, Johnston FM, Azad NS, Christenson ES, Lafaro KJ, Shubert CR. Current surgical management strategies for colorectal cancer liver metastases. Cancers. 2022;14(4):1063. https://doi.org/10.3390/cancers14041063.
Memeo R, de Blasi V, Adam R, et al. Parenchymal-sparing hepatectomies (PSH) for bilobar colorectal liver metastases are associated with a lower morbidity and similar oncological results: a propensity score matching analysis. HPB. 2016;18(9):781–90. https://doi.org/10.1016/j.hpb.2016.06.004.
Imai K, Allard MA, Castro Benitez C, et al. Long-term outcomes of radiofrequency ablation combined with hepatectomy compared with hepatectomy alone for colorectal liver metastases. Br J Surg. 2017;104(5):570–9. https://doi.org/10.1002/bjs.10447.
Mima K, Beppu T, Chikamoto A, et al. Hepatic resection combined with radiofrequency ablation for initially unresectable colorectal liver metastases after effective chemotherapy is a safe procedure with a low incidence of local recurrence. Int J Clin Oncol. 2013;18(5):847–55. https://doi.org/10.1007/s10147-012-0471-z.
Wang LJ, Zhang ZY, Yan XL, Yang W, Yan K, Xing BC. Radiofrequency ablation versus resection for technically resectable colorectal liver metastasis: a propensity score analysis. World J Surg Oncol. 2018;16(1):207. https://doi.org/10.1186/s12957-018-1494-3.
Cervantes A, Adam R, Roselló S, et al. Metastatic colorectal cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34(1):10–32. https://doi.org/10.1016/j.annonc.2022.10.003.
Ruers T, Van Coevorden F, Punt CJA, et al. Local Treatment of Unresectable Colorectal Liver Metastases: Results of a Randomized Phase II Trial. JNCI J Natl Cancer Inst. 2017. https://doi.org/10.1093/jnci/djx015.
Puijk RS, Ruarus AH, Vroomen LGPH, et al. Colorectal liver metastases: surgery versus thermal ablation (COLLISION) - a phase III single-blind prospective randomized controlled trial. BMC Cancer. 2018;18(1):821. https://doi.org/10.1186/s12885-018-4716-8.
Di Martino M, Rompianesi G, Mora-Guzmán I, Martín-Pérez E, Montalti R, Troisi RI. Systematic review and meta-analysis of local ablative therapies for resectable colorectal liver metastases. Eur J Surg Oncol 2020;46(5):772–81. https://doi.org/10.1016/j.ejso.2019.12.003.
Abdalla EK, Vauthey JN, Ellis LM, et al. Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg. 2004;239(6):818–27. https://doi.org/10.1097/01.sla.0000128305.90650.71.
Hao W, Binbin J, Wei Y, Kun Y. Can radiofrequency ablation replace liver resection for solitary colorectal liver metastasis? a systemic review and meta-analysis. Front Oncol. 2020;10:561669. https://doi.org/10.3389/fonc.2020.561669.
Hokuto D, Nomi T, Yamato I, et al. The prognosis of liver resection for patients with four or more colorectal liver metastases has not improved in the era of modern chemotherapy. J Surg Oncol. 2016;114(8):959–65. https://doi.org/10.1002/jso.24461.
Margonis GA, Sasaki K, Kim Y, et al. Tumor biology rather than surgical technique dictates prognosis in colorectal cancer liver metastases. J Gastrointest Surg Off J Soc Surg Aliment Tract. 2016;20(11):1821–9. https://doi.org/10.1007/s11605-016-3198-8.
Sadot E, Groot Koerkamp B, Leal JN, et al. Resection margin and survival in 2368 patients undergoing hepatic resection for metastatic colorectal cancer: surgical technique or biologic surrogate? Ann Surg. 2015;262(3):476–85. https://doi.org/10.1097/SLA.0000000000001427.
Chen Y, Xu Y, Xu L, et al. Neoadjuvant chemotherapy followed by radiofrequency ablation prolongs survival for ablatable colorectal liver metastasis: a propensity score matching comparative study. Front Oncol. 2021;11:758552. https://doi.org/10.3389/fonc.2021.758552.
de Jong MC, van Vledder MG, Ribero D, et al. Therapeutic efficacy of combined intraoperative ablation and resection for colorectal liver metastases: an international, multi-institutional analysis. J Gastrointest Surg Off J Soc Surg Aliment Tract. 2011;15(2):336–44. https://doi.org/10.1007/s11605-010-1391-8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
The authors have no conflicts of interest and no funding source to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Giannone, F., Grollemund, A., Felli, E. et al. Combining Radiofrequency Ablation with Hepatic Resection for Liver-Only Colorectal Metastases: A Propensity-Score Based Analysis of Long-Term Outcomes. Ann Surg Oncol 30, 4856–4866 (2023). https://doi.org/10.1245/s10434-023-13530-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-023-13530-3