Abstract
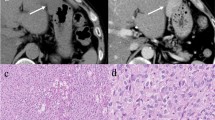
Gastrointestinal stromal tumor (GIST) is the most common human sarcoma and usually results from a sporadic mutation in KIT or, less frequently, platelet-derived growth factor alpha (PDGFRA). Rarely, a germline mutation in the KIT, PDGFRA, succinate dehydrogenase (SDH), or neurofibromatosis 1 (NF1) gene is responsible for GIST. These tumors are found in the stomach (PDGFRA and SDH), small bowel (NF1), or a combination of both (KIT). There is a need to improve care for these patients regarding genetic testing, screening, and surveillance. Since most GISTs due to a germline mutation do not respond to tyrosine kinase inhibitors, the role of surgery is critical, especially when considering germline gastric GIST. However, in contrast to the established recommendation for prophylactic total gastrectomy in cadherin 1 (CDH1) mutation carriers once they reach adulthood, there are no formal guidelines as to the timing or extent of surgical resection for patients who are either carriers of a germline GIST mutation causing gastric GIST or have already developed gastric GIST(s). Surgeons must balance treating what is often multicentric, yet initially indolent disease with the chance of cure and the complications associated with total gastrectomy. Here, we consider the major issues in performing surgery in patients with germline GIST and illustrate the principles with a previously unreported patient harboring a germline KIT 579 deletion.
Graphical abstract





Similar content being viewed by others
References
Joensuu H, DeMatteo RP. The management of gastrointestinal stromal tumors: a model for targeted and multidisciplinary therapy of malignancy. Annu Rev Med. 2012;63:247–58. https://doi.org/10.1146/annurev-med-043010-091813.
Ricci R. Syndromic gastrointestinal stromal tumors. Hered Cancer Clin Pract. 2016;14:15. https://doi.org/10.1186/s13053-016-0055-4.
Fornasarig M, Gasparotto D, Foltran L, et al. A novel kindred with familial gastrointestinal stromal tumors caused by a rare KIT germline mutation (N655K): clinico-pathological presentation and TKI sensitivity. J Pers Med. 2020. https://doi.org/10.3390/jpm10040234.
Brodey A, Kounnis V, Hawkes L, Jones RL, McVeigh TP, Cojocaru E. KIT-associated familial GIST syndrome: response to tyrosine kinase inhibitors and implications for risk management. Oncologist. 2022;27(8):615–20. https://doi.org/10.1093/oncolo/oyac120.
Maeyama H, Hidaka E, Ota H, et al. Familial gastrointestinal stromal tumor with hyperpigmentation: association with a germline mutation of the c-kit gene. Gastroenterology. 2001;120(1):210–5. https://doi.org/10.1053/gast.2001.20880.
Wali GN, Halliday D, Dua J, Ieremia E, McPherson T, Matin RN. Cutaneous hyperpigmentation and familial gastrointestinal stromal tumour associated with KIT mutation. Clin Exp Dermatol. 2019;44(4):418–21. https://doi.org/10.1111/ced.13757.
Bachet JB, Landi B, Laurent-Puig P, et al. Diagnosis, prognosis and treatment of patients with gastrointestinal stromal tumour (GIST) and germline mutation of KIT exon 13. Eur J Cancer. 2013;49(11):2531–41. https://doi.org/10.1016/j.ejca.2013.04.005.
Belinsky MG, Rink L, von Mehren M. Succinate dehydrogenase deficiency in pediatric and adult gastrointestinal stromal tumors. Front Oncol. 2013;3:117. https://doi.org/10.3389/fonc.2013.00117.
Boikos SA, Pappo AS, Killian JK, et al. Molecular subtypes of KIT/PDGFRA wild-type gastrointestinal stromal tumors: a report from the national institutes of health gastrointestinal stromal tumor clinic. JAMA Oncol. 2016;2(7):922–8. https://doi.org/10.1001/jamaoncol.2016.0256.
MacFarlane J, Seong KC, Bisambar C, et al. A review of the tumour spectrum of germline succinate dehydrogenase gene mutations: beyond phaeochromocytoma and paraganglioma. Clin Endocrinol (Oxf). 2020;93(5):528–38. https://doi.org/10.1111/cen.14289.
Manley PN, Abu-Abed S, Kirsch R, et al. Familial PDGFRA-mutation syndrome: somatic and gastrointestinal phenotype. Hum Pathol. 2018;76:52–7. https://doi.org/10.1016/j.humpath.2018.02.014.
Gopie P, Mei L, Faber AC, Grossman SR, Smith SC, Boikos SA. Classification of gastrointestinal stromal tumor syndromes. Endocr Relat Cancer. 2018;25(2):R49-r58. https://doi.org/10.1530/erc-17-0329.
Neppala P, Banerjee S, Fanta PT, et al. Current management of succinate dehydrogenase-deficient gastrointestinal stromal tumors. Cancer Metastasis Rev. 2019;38(3):525–35. https://doi.org/10.1007/s10555-019-09818-0.
Elston MS, Sehgal S, Dray M, et al. A duodenal SDH-deficient gastrointestinal stromal tumor in a patient with a germline SDHB mutation. J Clin Endocrinol Metab. 2017;102(5):1447–50. https://doi.org/10.1210/jc.2017-00165.
Nannini M, Rizzo A, Indio V, Schipani A, Astolfi A, Pantaleo MA. Targeted therapy in SDH-deficient GIST. Ther Adv Med Oncol. 2021;13:17588359211023278. https://doi.org/10.1177/17588359211023278.
Ben-Ami E, Barysauskas CM, von Mehren M, et al. Long-term follow-up results of the multicenter phase II trial of regorafenib in patients with metastatic and/or unresectable GI stromal tumor after failure of standard tyrosine kinase inhibitor therapy. Ann Oncol. 2016;27(9):1794–9. https://doi.org/10.1093/annonc/mdw228.
Casali PG, Abecassis N, Aro HT, et al. Gastrointestinal stromal tumours: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018;29(Suppl 4):iv68–78. https://doi.org/10.1093/annonc/mdy095.
Mandelker D, Marra A, Mehta N, et al. Expanded genetic testing of GIST patients identifies high proportion of non-syndromic patients with germline alterations. NPJ Precis Oncol. 2023;7(1):1. https://doi.org/10.1038/s41698-022-00342-z.
Syngal S, Brand RE, Church JM, Giardiello FM, Hampel HL, Burt RW. ACG clinical guideline: Genetic testing and management of hereditary gastrointestinal cancer syndromes. Am J Gastroenterol. 2015;110(2):223–62. https://doi.org/10.1038/ajg.2014.435.
Gamble LA, Heller T, Davis JL. Hereditary diffuse gastric cancer syndrome and the role of CDH1: a review. JAMA Surg. 2021;156(4):387–92. https://doi.org/10.1001/jamasurg.2020.6155.
Janeway KA, Weldon CB. Pediatric gastrointestinal stromal tumor. Semin Pediatr Surg. 2012;21(1):31–43. https://doi.org/10.1053/j.sempedsurg.2011.10.003.
Pappo AS, Janeway K, Laquaglia M, Kim SY. Special considerations in pediatric gastrointestinal tumors. J Surg Oncol. 2011;104(8):928–32. https://doi.org/10.1002/jso.21868.
Weldon CB, Madenci AL, Boikos SA, et al. Surgical management of wild-type gastrointestinal stromal tumors: a report from the national institutes of health pediatric and wildtype GIST clinic. J Clin Oncol. 2017;35(5):523–8. https://doi.org/10.1200/jco.2016.68.6733.
Vos EL, Salo-Mullen EE, Tang LH, et al. Indications for total gastrectomy in CDH1 mutation carriers and outcomes of risk-reducing minimally invasive and open gastrectomies. JAMA Surg. 2020;155(11):1050–7. https://doi.org/10.1001/jamasurg.2020.3356.
Pandalai PK, Lauwers GY, Chung DC, Patel D, Yoon SS. Prophylactic total gastrectomy for individuals with germline CDH1 mutation. Surgery. 2011;149(3):347–55. https://doi.org/10.1016/j.surg.2010.07.005.
Yoo C, Ryu MH, Kang BW, et al. Cross-sectional study of imatinib plasma trough levels in patients with advanced gastrointestinal stromal tumors: impact of gastrointestinal resection on exposure to imatinib. J Clin Oncol. 2010;28(9):1554–9. https://doi.org/10.1200/JCO.2009.26.5785.
Lubberman FJE, Gelderblom H, Wilmer CM, et al. Does a glass of Coke boost the exposure to imatinib in gastrointestinal stromal tumour patients after gastrectomy? Br J Clin Pharmacol. 2017;83(10):2312–4. https://doi.org/10.1111/bcp.13333.
Forde PM, Cochran RL, Boikos SA, et al. Familial GI stromal tumor with loss of heterozygosity and amplification of mutant KIT. J Clin Oncol. 2016;34(3):e13–6. https://doi.org/10.1200/jco.2013.51.6633.
Jones DH, Caracciolo JT, Hodul PJ, Strosberg JR, Coppola D, Bui MM. Familial gastrointestinal stromal tumor syndrome: report of 2 cases with KIT exon 11 mutation. Cancer Control. 2015;22(1):102–8. https://doi.org/10.1177/107327481502200113.
Kleinbaum EP, Lazar AJ, Tamborini E, et al. Clinical, histopathologic, molecular and therapeutic findings in a large kindred with gastrointestinal stromal tumor. Int J Cancer. 2008;122(3):711–8. https://doi.org/10.1002/ijc.23137.
Lasota J, Miettinen M. A new familial GIST identified. Am J Surg Pathol. 2006;30(10):1342. https://doi.org/10.1097/01.pas.0000213364.56498.3b.
Miettinen M, Makhlouf H, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the jejunum and ileum: a clinicopathologic, immunohistochemical, and molecular genetic study of 906 cases before imatinib with long-term follow-up. Am J Surg Pathol. 2006;30(4):477–89. https://doi.org/10.1097/00000478-200604000-00008.
Tarn C, Merkel E, Canutescu AA, et al. Analysis of KIT mutations in sporadic and familial gastrointestinal stromal tumors: therapeutic implications through protein modeling. Clin Cancer Res. 2005;11(10):3668–77. https://doi.org/10.1158/1078-0432.Ccr-04-2515.
Acknowledgment
The investigators were supported by NIH grants R01 CA102613 and T32 CA251063, the David Foundation, Betsy Levine-Brown and Marc Brown, and the GIST Cancer Research Fund (RPD).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Hyunjee V. Kwak, Katherine J. Tardy, Andrew Allbee, Kristen Stashek, and Ronald P. DeMatteo declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kwak, H.V., Tardy, K.J., Allbee, A. et al. Surgical Management of Germline Gastrointestinal Stromal Tumor. Ann Surg Oncol 30, 4966–4974 (2023). https://doi.org/10.1245/s10434-023-13519-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-023-13519-y