Abstract
Background
Body weight loss (BWL) and skeletal muscle loss (SML) are inevitable after gastrectomy for gastric cancer (GC) and can decrease patients’ quality of life (QOL) and survival.
Objective
The aim of this retrospective study was to evaluate the effect of perioperative and post-discharge patient participation in continuous nutritional counseling (CNC) on post-gastrectomy BWL and SML.
Methods
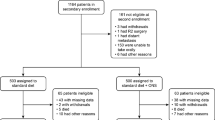
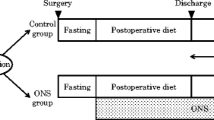
Ninety-three patients with GC who underwent curative gastrectomy between March 2018 and July 2019 were analyzed. Patients received either pre-discharge nutritional counseling alone (control group, n = 49) or patient-participation CNC (CNC group, n = 44) after gastrectomy. Differences between percentage BWL (%BWL), percentage SML (%SML), and nutrition-related blood parameters between the preoperative values and those at 12 months after surgery were compared between the groups.
Results
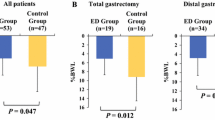
Compared with the control group, %BWL was significantly lower in the CNC group at 1 month (−6.2 ± 2.5% vs. −7.9 ± 3.3%, p = 0.005), 6 months (−7.8 ± 6.6% vs. −12.3 ± 6.4%, p = 0.001) and 12 months (−7.9 ± 7.6% vs. −13.2 ± 8.2%, p = 0.002), and %SML was significantly lower in the CNC group at 12 months (−5.3 ± 10.3% vs. −12.8 ± 12%, p = 0.002). Regarding nutrition-related blood parameters, change in total cholesterol was significantly lower in the CNC group than the control group at 12 months after surgery (p = 0.02). Multivariate analysis identified no CNC as an independent risk factor for severe BWL (p = 0.001) and SML (p = 0.006) at 12 months after surgery.
Conclusions
Following gastrectomy, patient-participation CNC prevented postoperative BWL and SML after surgery. These results support the induction of such a CNC program in these patients.



Similar content being viewed by others
Change history
11 November 2022
Affiliation number 1 was corrected.
References
Wilke H, Muro K, Van Cutsem E, Oh SC, Bodoky G, Shimada Y, et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol. 2014;15:1224–35.
Kang YK, Boku N, Satoh T, Ryu MH, Chao Y, Kato K, et al. Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;390:2461–71.
Shitara K, Van Cutsem E, Bang YJ, Fuchs C, Wyrwicz L, Lee KW, et al. Efficacy and safety of pembrolizumab or pembrolizumab plus chemotherapy vs chemotherapy alone for patients with first-line, advanced gastric cancer: The KEYNOTE-062 phase 3 randomized clinical trial. JAMA Oncol. 2020;6:1571–80.
Kurokawa Y, Sasako M, Sano T, Shibata T, Ito S, Nashimoto A, et al. Functional outcomes after extended surgery for gastric cancer. Br J Surg. 2011;98:239–45.
Takiguchi S, Takata A, Murakami K, Miyazaki Y, Yanagimoto Y, Kurokawa Y, et al. Clinical application of ghrelin administration for gastric cancer patients undergoing gastrectomy. Gastric Cancer. 2014;17:200–5.
Terashima M, Tanabe K, Yoshida M, Kawahira H, Inada T, Okabe H, et al. Postgastrectomy syndrome assessment scale (PGSAS)-45 and changes in body weight are useful tools for evaluation of reconstruction methods following distal gastrectomy. Ann Surg Oncol. 2014;3:S370–8.
Takiguchi N, Takahashi M, Ikeda M, Inagawa S, Ueda S, Nobuoka T, et al. Long-term quality-of-life comparison of total gastrectomy and proximal gastrectomy by postgastrectomy syndrome assessment scale (PGSAS-45): a nationwide multi-institutional study. Gastric Cancer. 2015;18:407–16.
Aoyama T, Yoshikawa T, Shirai J, Hayashi T, Yamada T, Tsuchida K, et al. Body weight loss after surgery is an independent risk factor for continuation of S-1 adjuvant chemotherapy for gastric cancer. Ann Surg Oncol. 2013;20:2000–6.
Lee HH, Park JM, Song KY, Choi MG, Park CH. Survival impact of postoperative body mass index in gastric cancer patients undergoing gastrectomy. Eur J Cancer. 2016;52:129–37.
Kudou K, Saeki H, Nakashima Y, Sasaki S, Jogo T, Hirose K, et al. Postoperative development of sarcopenia is a strong predictor of a poor prognosis in patients with adenocarcinoma of the esophagogastric junction and upper gastric cancer. Am J Surg. 2019;217:757–63.
Miyazaki Y, Omori T, Fujitani K, Fujita J, Kawabata R, Imamura H, et al. Oral nutritional supplements versus a regular diet alone for body weight loss after gastrectomy: a phase 3, multicenter, open-label randomized controlled trial. Gastric Cancer. 2021;24:1150–9.
Kimura Y, Nishikawa K, Kishi K, Inoue K, Matsuyama J, Akamaru Y, et al. Long-term effects of an oral elemental nutritional supplement on post-gastrectomy body weight loss in gastric cancer patients (KSES002). Ann Gastroenterol Surg. 2019;3:648–56.
Kong SH, Lee HJ, Na JR, Kim WG, Han DS, Park SH, et al. Effect of perioperative oral nutritional supplementation in malnourished patients who undergo gastrectomy: a prospective randomized trial. Surgery. 2018;164:1263–70.
Hatao F, Chen KY, Wu JM, Wang MY, Aikou S, Onoyama H, et al. Randomized controlled clinical trial assessing the effects of oral nutritional supplements in postoperative gastric cancer patients. Langenbecks Arch Surg. 2017;402:203–11.
Ida S, Hiki N, Cho H, Sakamaki K, Ito S, Fujitani K, et al. Randomized clinical trial comparing standard diet with perioperative oral immunonutrition in total gastrectomy for gastric cancer. Br J Surg. 2017;104:377–83.
Baldwin C, de Schueren MA, Kruizenga HM, Weekes CE. Dietary advice with or without oral nutritional supplements for disease-related malnutrition in adults. Cochrane Database Syst Rev. 2021;121(12):CD002008.
Persson CR, Johansson BB, Sjo¨den PO, Glimelius BLG. A randomized study of nutritional support in patients with colorectal and gastric cancer. Nutr Cancer. 2002;42:48–58.
Taylor K. Paternalism, participation and partnership - The evolution of patient centeredness in the consultation. Patient Educ Couns. 2009;74:150–5.
Sahlsten MJ, Larsson IE, Sjo¨stro¨m B, Lindencrona CSC, Plos KAE. Patient participation in nursing care: towards a concept clarification from a nurse perspective. J Clin Nurs. 2007;16:630–7.
Sahlsten MJM, Larsson IE, Sjo¨stro¨m B, Plos KAE. Nurse strategies for optimising patient participation in nursing care. Scand J Caring Sci. 2009;23:490–7.
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–83.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13.
Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer. 2011;14:101–12.
Huang X, Wu L, Gao L, Yu S, Chen X, Wang C, et al. Chinese obesity and metabolic surgery collaborative. impact of self-monitoring on weight loss after bariatric surgery. Obes Surg. 2021;31:4399–404.
Turner-McGrievy GM, Beets MW, Moore JB, Kaczynski AT, Barr-Anderson DJ, Tate DF. Comparison of traditional versus mobile app self-monitoring of physical activity and dietary intake among overweight adults participating in an mHealth weight loss program. J Am Med Inform Assoc. 2013;20:513–8.
Meng Q, Tan S, Jiang Y, Han J, Xi Q, Zhuang Q, et al. Post-discharge oral nutritional supplements with dietary advice in patients at nutritional risk after surgery for gastric cancer: a randomized clinical trial. Clin Nutr. 2021;40:40–6.
Furukawa H, Kurokawa Y, Takiguchi S, Tanaka K, Miyazaki Y, Makino T, et al. Short-term outcomes and nutritional status after laparoscopic subtotal gastrectomy with a very small remnant stomach for cStage I proximal gastric carcinoma. Gastric Cancer. 2018;21:500–7.
Nakauchi M, Suda K, Nakamura K, Shibasaki S, Kikuchi K, Nakamura T, et al. Laparoscopic subtotal gastrectomy for advanced gastric cancer: technical aspects and surgical, nutritional and oncological outcomes. Surg Endosc. 2017;31:4631–40.
Sun Z, Shenoi MM, Nussbaum DP, Keenan JE, Gulack BC, Tyler DS, et al. Feeding jejunostomy tube placement during resection of gastric cancers. J Surg Res. 2016;200:189–94.
Dann GC, Squires MH 3rd, Postlewait LM, Kooby DA, Poultsides GA, Weber SM, et al. An assessment of feeding jejunostomy tube placement at the time of resection for gastric adenocarcinoma: a seven-institution analysis of 837 patients from the U.S. gastric cancer collaborative. J Surg Oncol. 2015;112:195–202.
Muscaritoli M, Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, et al. ESPEN practical guideline: clinical NUTRITION in cancer. Clin Nutr. 2021;40:2898–913.
Poulsen GM, Pedersen LL, Østerlind K, Bæksgaard L, Andersen JR. Randomized trial of the effects of individual nutritional counseling in cancer patients. Clin Nutr. 2014;33:749–53.
Isenring EA, Bauer JD, Capra S. Nutrition support using the American dietetic association medical nutrition therapy protocol for radiation oncology patients improves dietary intake compared with standard practice. J Am Diet Assoc. 2007;107:404–12.
Ravasco P, Monteiro-Grillo I, Vidal PM, Camilo ME. Dietary counseling improves patient outcomes: a prospective, randomized, controlled trial in colorectal cancer patients undergoing radiotherapy. J Clin Oncol. 2005;23:1431–8.
Rosenberg IH. Sarcopenia: origins and clinical relevance. J Nutr. 1997;127(5 Suppl):990S-991S. https://doi.org/10.1093/jn/127.5.990S.
Kuwada K, Kuroda S, Kikuchi S, Yoshida R, Nishizaki M, Kagawa S, et al. Sarcopenia and comorbidity in gastric cancer surgery as a useful combined factor to predict eventual death from other causes. Ann Surg Oncol. 2018;25:1160–6.
Harimoto N, Shirabe K, Yamashita YI, Ikegami T, Yoshizumi T, Soejima Y, et al. Sarcopenia as a predictor of prognosis in patients following hepatectomy for hepatocellular carcinoma. Br J Surg. 2013;100:1523–30.
Okumura S, Kaido T, Hamaguchi Y, Kobayashi A, Shirai H, Fujimoto Y, et al. Impact of skeletal muscle mass, muscle quality, and visceral adiposity on outcomes following resection of intrahepatic cholangiocarcinoma. Ann Surg Oncol. 2017;24:1037–45.
Miyake M, Morizawa Y, Hori S, Marugami N, Shimada K, Gotoh D, et al. Clinical impact of postoperative loss in psoas major muscle and nutrition index after radical cystectomy for patients with urothelial carcinoma of the bladder. BMC Canc. 2017;17:237.
Acknowledgment
The authors wish to thank all patients who contributed their data for analysis in the present study.
Funding
No sources of funding were used to assist in the preparation of this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Nobuo Takata, Satoru Kikuchi, Shinji Kuroda, Shunsuke Tanabe, Naoaki Maeda, Kazuhiro Noma, Ayako Takahashi, Yuzo Umeda, Kenichi Shikata, Kazuhide Ozaki and Toshiyoshi Fujiwara declare they have no conflicts of interest in relation to this work.
Ethical Approval
All procedures followed were in accordance with the standards of the Ethics Committee of Okayama University Hospital and with the Helsinki Declaration of 1964 and later versions.
Informed consent
Information about the aim of this retrospective study was posted on the website of the Department of Gastroenterological Surgery, Okayama University Hospital, and potential participants could decline to participate or opt out at any time.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Takata, N., Kikuchi, S., Kuroda, S. et al. Effect of Patient-Participation Continuous Nutritional Counseling in Gastric Cancer Patients who Underwent Gastrectomy. Ann Surg Oncol 30, 1110–1118 (2023). https://doi.org/10.1245/s10434-022-12572-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-022-12572-3