Abstract
Background
Although neoadjuvant therapy followed by surgery (NT) is the standard of care for esophageal cancer in Western countries, upfront surgery (US) followed by adjuvant therapy (when indicated) still is commonly used in Asia to minimize overtreatment. This study investigated the cost-effectiveness of NT versus US for patients with esophageal squamous cell carcinoma (ESCC).
Methods
Patients with a diagnosis of ESCC between 2010 and 2015 were divided into NT or US according to the intention to treat. Two propensity score-matched groups of patients with clinical stage 2 (135 pairs) or stage 3 (194 pairs) disease were identified and compared in terms of overall survival (OS) and direct costs incurred within 3 years after diagnosis.
Results
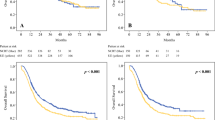
The esophagectomy rates after NT were 82% for stage 2 and 88% for stage 3 disease. Compared with US, surgery after NT was associated with higher R0 resection rates, a lower number of dissected lymph nodes, and similar postoperative mortality. On an intention-to-treat analysis, stage 3 patients who received NT had a significantly better 3-year OS rate (45%) than those treated with US (37%) (p = 0.029) without significant cost increases (p = 0.89). However, NT for clinical stage 2 disease neither increased costs nor improved 3-year OS rates (47% vs 47%; p = 0.88). At a willingness-to-pay level of US$50,000 per life-year, the probability of NT being cost-effective was 92% for stage 3 versus 59% for stage 2 ESCC.
Conclusion
Because of its higher cost-effectiveness, NT is preferable to US for patients with clinical stage 3 ESCC, but US remains a viable option for stage 2 disease.




Similar content being viewed by others
References
Chan KKW, Saluja R, Delos Santos K, Lien K, Shah K, Cramarossa G, et al. Neoadjuvant treatments for locally advanced, resectable esophageal cancer: a network meta-analysis. Int J Cancer. 2018;143:430–7.
Kumar T, Pai E, Singh R, Francis NJ, Pandey M. Neoadjuvant strategies in resectable carcinoma esophagus: a meta-analysis of randomized trials. World J Surg Oncol. 2020;18:1–10.
Shapiro J, van Lanschot JJB, Hulshof M, van Hagen P, van Berge Henegouwen MI, Wijnhoven BPL, et al. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): long-term results of a randomised controlled trial. Lancet Oncol. 2015;16:1090–8.
Yang H, Liu H, Chen Y, Zhu C, Fang W, Yu Z, et al. Neoadjuvant chemoradiotherapy followed by surgery versus surgery alone for locally advanced squamous cell carcinoma of the esophagus (NEOCRTEC5010): a phase III multicenter, randomized, open-label clinical trial. J Clin Oncol. 2018;36:2796–803.
Markar SR, Gronnier C, Pasquer A, Duhamel A, Beal H, Thereaux J, et al. Role of neoadjuvant treatment in clinical T2N0M0 oesophageal cancer: results from a retrospective multi-center European study. Eur J Cancer. 2016;56:59–68.
Hsu P-K, Chen H-S, Liu C-C, Wu S-C. Neoadjuvant chemoradiation versus upfront esophagectomy in clinical stage II and III esophageal squamous cell carcinoma. Ann Surg Oncol. 2019;26:506–13.
Zahoor H, Luketich JD, Levy RM, Awais O, Winger DG, Gibson MK, et al. A propensity-matched analysis comparing survival after primary minimally invasive esophagectomy followed by adjuvant therapy to neoadjuvant therapy for esophagogastric adenocarcinoma. J Thorac Cardiovasc Surg. 2015;149:538–47.
Xiao X, Hong HG, Zeng X, Yang Y-S, Luan S-Y, Li Y, et al. The efficacy of neoadjuvant versus adjuvant therapy for resectable esophageal cancer patients: a systematic review and meta-analysis. World J Surg. 2020;44:4161–74.
The Health and Welfare Data Science Center (HWDC) database. 2015. Retrieved 2 August 2021, http://www.mohw.gov.tw/cht/DOS/DM1.aspx?f_list_no=812.
Bureau of National Health Insurance, Department of Health, Universal Health Coverage in Taiwan. 2012. Retrieved 2 August 2021, http://www.nhi.gov.tw/Resource/webdata/21717_1_20120808UniversalHealthCoverage.pdf.
Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D, et al. Consolidated Health Economic Evaluation Reporting Standards (CHEERS) statement. Eur J Health Econ. 2013;14:367–72.
Neumann PJ, Cohen JT, Weinstein MC. Updating cost-effectiveness: the curious resilience of the $50,000-per-QALY threshold. N Engl J Med. 2014;371:796–7.
Mokdad AA, Yopp AC, Polanco PM, Mansour JC, Reznik SI, Heitjan DF, et al. Adjuvant chemotherapy vs postoperative observation following preoperative chemoradiotherapy and resection in gastroesophageal cancer. JAMA Oncol. 2018;4:31.
Zhan M, Zheng H, Yang Y, Xu T, Li Q. Cost-effectiveness analysis of neoadjuvant chemoradiotherapy followed by surgery versus surgery alone for locally advanced esophageal squamous cell carcinoma based on the NEOCRTEC5010 trial. Radiother Oncol. 2019;141:27–32.
Chen H-S, Hsu P-K, Liu C-C, Wu S-C. Upfront surgery and pathological stage-based adjuvant chemoradiation strategy in locally advanced esophageal squamous cell carcinoma. Sci Rep. 2018;8:1–9.
Ajani JA, D’Amico TA, Bentrem DJ, Chao J, Corvera C, Das P, et al. Esophageal and esophagogastric junction cancers, version 2.2019, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Cancer Netw. 2019;17:855–83.
Fang H-Y, Chao Y-K, Chang H-K, Tseng C-K, Liu Y-H. Survival outcomes of consolidation chemoradiotherapy in esophageal cancer patients who achieve clinical complete response but refuse surgery after neoadjuvant chemoradiotherapy. Dis Esophagus. 2016;30:1–8.
Abbott DE, Tzeng CW, Merkow RP, Cantor SB, Chang GJ, Katz MH, et al. The cost-effectiveness of neoadjuvant chemoradiation is superior to a surgery-first approach in the treatment of pancreatic head adenocarcinoma. Ann Surg Oncol. 2013;20(Suppl 3):S500–8.
Wright ME, Beaty JS, Thorson AG, Rojas R, Ternent CA. Cost-effectiveness analysis of total neoadjuvant therapy followed by radical resection versus conventional therapy for locally advanced rectal cancer. Dis Colon Rectum. 2019;62:568–78.
Murray CJ, Evans DB, Acharya A, Baltussen RM. Development of WHO guidelines on generalized cost-effectiveness analysis. Health Econ. 2000;9:235–51.
Eichler H-G, Kong SX, Gerth WC, Mavros P, Jönsson B. Use of cost-effectiveness analysis in health-care resource allocation decision-making: how are cost-effectiveness thresholds expected to emerge? Value Health. 2004;7:518–28.
Acknowledgment
This study was financially supported by grants CORPD1J0041 and BMRPC55 from the Chang Gung University and Grant MOST 110-2410-H-182-009 from the Ministry of Science and Technology, Taiwan. The Research Service Center For Health Information of Chang Gung University, Taiwan, provided help with the data analysis. The Nijbakker-Morra foundation (the Netherlands) provided funding during the writing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, X., Wen, YW., van Lanschot, J.J.B. et al. Neoadjuvant Therapy Versus Upfront Surgery for Patients With Clinical Stage 2 or 3 Esophageal Squamous Cell Carcinoma: A Cost-Effectiveness Analysis. Ann Surg Oncol 29, 3644–3653 (2022). https://doi.org/10.1245/s10434-021-11207-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-021-11207-3