Abstract
Background
Sentinel lymph node biopsy (SLNB) is often omitted in selected patients with advanced primary melanoma, although the justification/criteria for omission have been debated.
Objective
We sought to determine whether assessment of frailty could serve as an objective marker to guide selection for SLNB in patients with advanced primary melanoma.
Methods
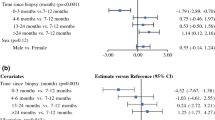
Patients presenting with clinical stage IIC (ulcerated, > 4 mm Breslow thickness) cutaneous melanoma from January 1999 through June 2019 were included. Frailty was assessed using the Memorial Sloan Kettering Frailty Index (MSK FI), a composite score of functional status and medical comorbidities. Five-year melanoma-specific survival (MSS) and overall survival (OS) were estimated using Cox regression, and predictors of OS were identified using competing risk models.
Results
MSS did not differ between patients who did (n = 451) or did not undergo SLNB (n = 179) [63.2% vs. 65.0%, p = 0.14]; however, omission of SLNB was associated with decreased 5-year OS (29% vs. 44%, p < 0.001). In a multivariable competing risk model, selection for SLNB omission was an independent predictor of death from non-melanoma causes (hazard ratio [HR] 1.7, 95% confidence interval [CI] 1.2–2.3, p < 0.001). After incorporation of the MSK FI score into the multivariable model in this subset, MSK FI (HR 2.4, 95% CI 1.5–4.1, p < 0.001), but not SLNB omission, was an independent predictor of poorer OS.
Conclusion
We observed worse OS in patients with thick melanoma selected not to undergo SLNB, which was attributed to death due to non-melanoma causes. Formal assessment of frailty may provide an objective prognostic measure to guide selective use of SLNB in these patients.


Similar content being viewed by others
References
Faries MB, Thompson JF, Cochran AJ, et al. Completion dissection or observation for sentinel-node metastasis in melanoma. N Engl J Med. 2017;376:2211–22.
Leiter U, Stadler R, Mauch C, et al. Complete lymph node dissection versus no dissection in patients with sentinel lymph node biopsy positive melanoma (DeCOG-SLT): a multicentre, randomised, phase 3 trial. Lancet Oncol. 2016;17:757–67.
Morton DL, Thompson JF, Cochran AJ, et al. Final trial report of sentinel-node biopsy versus nodal observation in melanoma. N Engl J Med. 2014;370:599–609.
Morton DL, Thompson JF, Essner R, et al. Validation of the accuracy of intraoperative lymphatic mapping and sentinel lymphadenectomy for early-stage melanoma: a multicenter trial. Multicenter Selective Lymphadenectomy Trial Group. Ann Surg. 1999;230:453–65.
Gershenwald JE, Scolyer RA. Melanoma staging: american joint committee on cancer (AJCC) 8th edition and beyond. Ann Surg Oncol. 2018;25:2105–10.
Bello DM, Han G, Jackson L, et al. The prognostic significance of sentinel lymph node status for patients with thick melanoma. Ann Surg Oncol. 2016;23:938–45.
Boada A, Tejera-Vaquerizo A, Ribero S, et al. Sentinel lymph node biopsy versus observation in thick melanoma: a multicenter propensity score matching study. Int J Cancer. 2018;142:641–8.
Gajdos C, Griffith KA, Wong SL, et al. Is there a benefit to sentinel lymph node biopsy in patients with T4 melanoma? Cancer. 2009;115:5752–60.
Gershenwald JE, Mansfield PF, Lee JE, et al. Role for lymphatic mapping and sentinel lymph node biopsy in patients with thick (> or = 4 mm) primary melanoma. Ann Surg Oncol. 2000;7:160–5.
Ribero S, Osella-Abate S, Sanlorenzo M, et al. Sentinel lymph node biopsy in thick-melanoma patients (N = 350): what is its prognostic role? Ann Surg Oncol. 2015;22:1967–73.
Scoggins CR, Bowen AL, Martin RC 2nd, et al. Prognostic information from sentinel lymph node biopsy in patients with thick melanoma. Arch Surg. 2010;145:622–7.
Gyorki DE, Sanelli A, Herschtal A, et al. Sentinel lymph node biopsy in T4 melanoma: an important risk-stratification tool. Ann Surg Oncol. 2016;23:579–84.
Song Y, Azari FS, Metzger DA, et al. Practice patterns and prognostic value of sentinel lymph node biopsy for thick melanoma: a national cancer database study. Ann Surg Oncol. 2019;26:4651–62.
Wong SL, Faries MB, Kennedy EB, et al. Sentinel lymph node biopsy and management of regional lymph nodes in melanoma: American society of clinical oncology and society of surgical oncology clinical practice guideline update. J Clin Oncol. 2017;36:399–413.
Murtha TD, Han G, Han D. Predictors for use of sentinel node biopsy and the association with improved survival in melanoma patients who have nodal staging. Ann Surg Oncol. 2018;25:903–11.
Shahrokni A, Tin A, Alexander K, et al. Development and evaluation of a new frailty index for older surgical patients with cancer. JAMA Netw Open. 2019;2:e193545.
Yervoy prescribing information. Revised April, 2018. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/125377s094lbl.pdf. Accessed 6 Jan 2021.
Wong SL, Faries MB, Kennedy EB, et al. Sentinel lymph node biopsy and management of regional lymph nodes in melanoma: American society of clinical oncology and society of surgical oncology clinical practice guideline update. Ann Surg Oncol. 2018;25:356–77.
Bartlett EK. Current management of regional lymph nodes in patients with melanoma. J Surg Oncol. 2019;119:200–7.
Hu Y, Shah P, Stukenborg GJ, et al. Utility of sentinel lymph node biopsy for solitary dermal melanomas. J Surg Oncol. 2015;111:800–7.
Kachare SD, Singla P, Vohra NA, et al. Sentinel lymph node biopsy is prognostic but not therapeutic for thick melanoma. Surgery. 2015;158:662–8.
Sperry SM, Charlton ME, Pagedar NA. Association of sentinel lymph node biopsy with survival for head and neck melanoma: survival analysis using the SEER database. JAMA Otolaryngol Head Neck Surg. 2014;140:1101–9.
Kaur A, Ecker BL, Douglass SM, et al. Remodeling of the collagen matrix in aging skin promotes melanoma metastasis and affects immune cell motility. Cancer Discov. 2019;9:64–81.
Balch CM, Thompson JF, Gershenwald JE, et al. Age as a predictor of sentinel node metastasis among patients with localized melanoma: an inverse correlation of melanoma mortality and incidence of sentinel node metastasis among young and old patients. Ann Surg Oncol. 2014;21:1075–81.
Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol. 1992;45:613–9.
Acknowledgment
The authors would like to acknowledge Jessica Moore for her editorial support in the production of this paper.
Funding
This research was supported in part by NIH/NCI Cancer Center Support Grant P30 CA008748 to the institution.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
DISCLOSURE
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, A.S., Flynn, J.R., Panageas, K.S. et al. Assessment of Frailty Can Guide Decision Making for Utilization of Sentinel Lymph Node Biopsy in Patients with Thick Melanoma. Ann Surg Oncol 28, 9031–9038 (2021). https://doi.org/10.1245/s10434-021-10212-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-021-10212-w