Abstract
Introduction
Preoperative biopsy (PBx) is often recommended for retroperitoneal sarcoma (RPS), but its utilization rate and impact on perioperative management and outcomes remains undefined.
Methods
Using the National Cancer Database, patients who underwent resection of non-metastatic RPS were identified (2006–2014). Patients who did and did not undergo PBx of the primary tumor were compared using propensity matching, and factors associated with survival were assessed by multivariable analysis.
Results
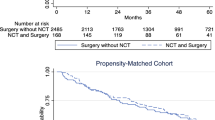
Of 2620 patients, 1110 (42.4%) underwent PBx. Factors significantly associated with performance of PBx included male sex [odds ratio (OR) 1.2, P = 0.035], tumor size ≤ 5 cm (OR 1.5, P = 0.012), tumor size > 5 to ≤ 10 cm (OR 1.3, P = 0.009), non-well-differentiated liposarcoma histology (OR 2.0, P ≤ 0.001), and treatment at a high-volume center (OR 1.3, P = 0.021). Receipt of PBx was significantly associated with administration of neoadjuvant radiation (OR 8.8, P < 0.001) or systemic therapy (OR 3.3, P < 0.001), radical surgical resection (OR 1.6, P < 0.001), and complete tumor resection (OR 1.5, P < 0.003). Neoadjuvant radiation [hazard ratio (HR) 0.7, P = 0.003] and complete tumor resection (HR 0.6, P < 0.001) were significantly associated with improved overall survival (OS). Performance of PBx was not associated with OS (HR 1.1, P = 0.070), and following propensity matching, 5-year OS did not differ between the two groups (56.5% PBx vs 58.4% no PBx, P = 0.247).
Conclusions
A minority of patients with non-metastatic RPS undergo PBx. PBx does not negatively impact survival, but may indirectly improve outcomes in select patients by virtue of receipt of neoadjuvant therapy and attainment of complete tumor resection.


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70(1):7–30. https://doi.org/10.3322/caac.21590.
Fletcher C, Bridge JA, Hogendoorn PCW, Mertens F. World Health Organization lassification of Tumours of Soft Tissue and Bone. 4th edn. Lyon: IARC Press; 2013.
Raut CP, Pisters PW. Retroperitoneal sarcomas: combined-modality treatment approaches. J Surg Oncol. 2006;94(1):81–7. https://doi.org/10.1002/jso.20543.
Fairweather M, Gonzalez RJ, Strauss D, Raut CP. Current principles of surgery for retroperitoneal sarcomas. J Surg Oncol. 2018;117(1):33–41. https://doi.org/10.1002/jso.24919.
van Dalen T, Plooij JM, van Coevorden F, et al. Long-term prognosis of primary retroperitoneal soft tissue sarcoma. Eur J Surg Oncol. 2007;33(2):234–8. https://doi.org/10.1016/j.ejso.2006.09.020.
Singer S, Antonescu CR, Riedel E, Brennan MF. Histologic subtype and margin of resection predict pattern of recurrence and survival for retroperitoneal liposarcoma. Ann Surg. 2003;238(3):358–70. https://doi.org/10.1097/01.sla.0000086542.11899.38 (discussion 370-1).
Canter RJ, Qin LX, Ferrone CR, Maki RG, Singer S, Brennan MF. Why do patients with low-grade soft tissue sarcoma die? Ann Surg Oncol. 2008;15(12):3550–60. https://doi.org/10.1245/s10434-008-0163-0.
Trans-Atlantic RPS Working Group. Management of primary retroperitoneal sarcoma (RPS) in the adult: a consensus approach from the Trans-Atlantic RPS Working Group. Ann Surg Oncol. 2015;22(1):256–63. https://doi.org/10.1245/s10434-014-3965-2.
ESMO/European Sarcoma Network Working Group. Soft tissue and visceral sarcomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2014;25Suppl 3:iii102-12n. https://doi.org/10.1093/annonc/mdu254.
National Comprehensive Cancer Network. Soft Tissue Sarcoma, Version 2.2020, NCCN Clinical Practice Guidelines in Oncology. Accessed October 23, 2020, https://www.nccn.org/professionals/physician_gls/PDF/sarcoma.pdf
Berger-Richardson D, Burtenshaw SM, Ibrahim AM, et al. Early and late complications of percutaneous core needle biopsy of Retroperitoneal Tumors at Two Tertiary Sarcoma Centers. Ann Surg Oncol. Dec ;26(13):4692–8. https://doi.org/10.1245/s10434-019-07656-6.
Van Houdt WJ, Schrijver AM, Cohen-Hallaleh RB, et al. Needle tract seeding following core biopsies in retroperitoneal sarcoma. Eur J Surg Oncol. Sep ;43(9):1740–5. https://doi.org/10.1016/j.ejso.2017.06.009.
Fairweather M, Raut CP. To Biopsy, or not to biopsy: Is there really a question? Ann Surg Oncol. 2019;26(13):4182–4. https://doi.org/10.1245/s10434-019-07723-y.
Bilimoria KY, Stewart AK, Winchester DP, Ko CY. The National cancer data base: a powerful initiative to improve cancer care in the United States. Ann Surg Oncol. 2008;15(3):683–90. https://doi.org/10.1245/s10434-007-9747-3.
American College of Surgeons. National Cancer Data Base Participant User File Dictionary. Accessed December 21, 2020, http://facs.org/-/media/files/quality-programs/cancer/ncdb/puf_data_dictionary.ashx
Schemper M, Smith TL. A note on quantifying follow-up in studies of failure time. Control Clin Trials. 1996;17(4):343–6. https://doi.org/10.1016/0197-2456(96)00075-x.
Ho D, Imai K, King G, Stuart E. Matching as nonparametric preprocessing for reducing model dependence in parametric causal inference. Political Anal. 2007. https://doi.org/10.1093/pan/mpl013.
StataCorp. Stata Statistical Software: Release 16. College Station, TX: StataCorp LLC; 2019.
Berger-Richardson D, Swallow CJ. Needle tract seeding after percutaneous biopsy of sarcoma: risk/benefit considerations. Cancer. 2017;123(4):560–7. https://doi.org/10.1002/cncr.30370.
Wilkinson MJ, Martin JL, Khan AA, Hayes AJ, Thomas JM, Strauss DC. Percutaneous core needle biopsy in retroperitoneal sarcomas does not influence local recurrence or overall survival. Ann Surg Oncol. 2015;22(3):853–8. https://doi.org/10.1245/s10434-014-4059-x.
Strauss DC, Qureshi YA, Hayes AJ, Thway K, Fisher C, Thomas JM. The role of core needle biopsy in the diagnosis of suspected soft tissue tumours. J Surg Oncol. 2010;102(5):523–9. https://doi.org/10.1002/jso.21600.
Madhavan VP, Smile SR, Chandra SS, Ratnakar C. Value of core needle biopsy in the diagnosis of soft tissue tumours. Indian J Pathol Microbiol. 2002;45(2):165–8.
Welker JA, Henshaw RM, Jelinek J, Shmookler BM, Malawer MM. The percutaneous needle biopsy is safe and recommended in the diagnosis of musculoskeletal masses. Cancer. 2000;89(12):2677–86. https://doi.org/10.1002/1097-0142(20001215)89:12%3c2677::aid-cncr22%3e3.0.co;2-l.
Lahat G, Madewell JE, Anaya DA, et al. Computed tomography scan-driven selection of treatment for retroperitoneal liposarcoma histologic subtypes. Cancer. 2009;115(5):1081–90. https://doi.org/10.1002/cncr.24045.
Abbott AM, Habermann EB, Parsons HM, Tuttle T, Al-Refaie W. Prognosis for primary retroperitoneal sarcoma survivors. Cancer. 2012;118(13):3321–9. https://doi.org/10.1002/cncr.26665.
Sandrucci S, Ponzetti A, Gianotti C, et al. Different quality of treatment in retroperitoneal sarcomas (RPS) according to hospital-case volume and surgeon-case volume: a retrospective regional analysis in Italy. Clin Sarcoma Res. 2018;8:3. https://doi.org/10.1186/s13569-018-0091-0.
Toulmonde M, Bonvalot S, Méeus P, et al. Retroperitoneal sarcomas: patterns of care at diagnosis, prognostic factors and focus on main histological subtypes: a multicenter analysis of the French Sarcoma Group. Ann Oncol. 2014;25(3):735–42. https://doi.org/10.1093/annonc/mdt577.
Bonvalot S, Miceli R, Berselli M, et al. Aggressive surgery in retroperitoneal soft tissue sarcoma carried out at high-volume centers is safe and is associated with improved local control. Ann Surg Oncol. 2010;17(6):1507–14. https://doi.org/10.1245/s10434-010-1057-5.
Maurice MJ, Yih JM, Ammori JB, Abouassaly R. Predictors of surgical quality for retroperitoneal sarcoma: volume matters. J Surg Oncol. 2017;116(6):766–74. https://doi.org/10.1002/jso.24710.
Berger NG, Silva JP, Mogal H, et al. Overall survival after resection of retroperitoneal sarcoma at academic cancer centers versus community cancer centers: an analysis of the National Cancer Data Base. Surgery. 2018;163(2):318–23.
Bagaria SP, Neville M, Gray RJ, et al. The volume-outcome relationship in retroperitoneal soft tissue sarcoma: evidence of improved short- and long-term Outcomes at High-Volume Institutions. Sarcoma. 2018. https://doi.org/10.1155/2018/3056562.
Jones JJ, Catton CN, O’Sullivan B, et al. Initial results of a trial of preoperative external-beam radiation therapy and postoperative brachytherapy for retroperitoneal sarcoma. Ann Surg Oncol. 2002;9(4):346–54. https://doi.org/10.1007/BF02573869.
Roeder F, Ulrich A, Habl G, et al. Clinical phase I/II trial to investigate preoperative dose-escalated intensity-modulated radiation therapy (IMRT) and intraoperative radiation therapy (IORT) in patients with retroperitoneal soft tissue sarcoma: interim analysis. BMC Cancer. 2014;14:617. https://doi.org/10.1186/1471-2407-14-617.
Issels RD, Lindner LH, Verweij J, et al. Neo-adjuvant chemotherapy alone or with regional hyperthermia for localised high-risk soft-tissue sarcoma: a randomised phase 3 multicentre study. Lancet Oncol. 2010;11(6):561–70. https://doi.org/10.1016/S1470-2045(10)70071-1.
Bonvalot S, Gronchi A, Le Péchoux C, et al. Preoperative radiotherapy plus surgery versus surgery alone for patients with primary retroperitoneal sarcoma (EORTC-62092: STRASS): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2020;21(10):1366–77. https://doi.org/10.1016/S1470-2045(20)30446-0.
Almond LM, Tirotta F, Tattersall H, et al. Diagnostic accuracy of percutaneous biopsy in retroperitoneal sarcoma. Br J Surg. 2019;106(4):395–403. https://doi.org/10.1002/bjs.11064.
Tirotta F, Morosi C, Hodson J, et al. Improved biopsy accuracy in retroperitoneal dedifferentiated liposarcoma. Ann Surg Oncol. 2020;27(11):4574–81. https://doi.org/10.1245/s10434-020-08519-1.
Mullinax JE, Zager JS, Gonzalez RJ. Current diagnosis and management of retroperitoneal sarcoma. Cancer Control. 2011;18(3):177–87. https://doi.org/10.1177/107327481101800305.
Diamantis A, Baloyiannis I, Magouliotis DE, et al. Perioperative radiotherapy versus surgery alone for retroperitoneal sarcomas: a systematic review and meta-analysis. Radiol Oncol. 2020;54(1):14–21. https://doi.org/10.2478/raon-2020-0012.
Turner BT, Hampton L, Schiller D, et al. Neoadjuvant radiotherapy followed by surgery compared with surgery alone in the treatment of retroperitoneal sarcoma: a population-based comparison. Curr Oncol. 2019;26(6):e766–72. https://doi.org/10.3747/co.26.5185.
Cardona K. The STRASS trial: an important step in the right direction. Lancet Oncol. 2020;21(10):1257–8. https://doi.org/10.1016/S1470-2045(20)30429-0.
Messiou C, Moskovic E, Vanel D, et al. Primary retroperitoneal soft tissue sarcoma: imaging appearances, pitfalls and diagnostic algorithm. Eur J Surg Oncol. 2017;43(7):1191–8. https://doi.org/10.1016/j.ejso.2016.10.032.
Funding
No external funding was received for this study.
Author information
Authors and Affiliations
Contributions
Conception and design of the study: Straker, Karakousis Acquisition of data: Straker, Song, Karakousis Analysis and interpretation of data: Straker, Song, Shannon, Marcinak, Miura, Fraker, Karakousis Drafting and critically revising the manuscript: Straker, Song, Shannon, Marcinak, Miura, Fraker, Karakousis All authors have reviewed and approved the submitted manuscript.
Corresponding author
Ethics declarations
DISCLOSURE
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Straker, R.J., Song, Y., Shannon, A.B. et al. Preoperative Biopsy in Patients with Retroperitoneal Sarcoma: Usage and Outcomes in a National Cohort. Ann Surg Oncol 28, 6868–6879 (2021). https://doi.org/10.1245/s10434-021-09691-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-021-09691-8