Abstract
Background
Few studies have compared the survival advantage of complete pathologic response (cPR) achieved through neoadjuvant chemotherapy (nCT) versus neoadjuvant chemoradiotherapy (nCRT) in gastric adenocarcinoma. Our study utilizes a large national cancer database to address this question.
Patients and Methods
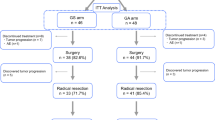
This is a retrospective review of patients with clinical stage I to III gastric adenocarcinoma from 2004 to 2013 who received nCT or nCRT. Patients who achieved cPR were selected. Associations were evaluated using Mann–Whitney U and Fisher’s exact tests. Survival information was summarized using standard Kaplan–Meier methods, where estimates of the median and 5-year survival rates were estimated with 95% confidence intervals.
Results
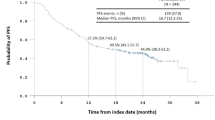
A total of 413 patients who had cPR were identified. Eighty-four patients received nCT and 329 patients received nCRT. Patients in the nCRT group had higher clinical stage (88.4% vs. 75.0%) and more proximal location of tumors (95.4% vs. 45.2%). The nCT group (n = 84) had a 94% 5-year survival rate, while the nCRT group’s (n = 329) rate was 60% (p < 0.001). On Cox regression modeling using a propensity-weighted approach, nCT treatment was an independent predictor of improved overall survival (nCRT vs. nCT; HR 10.44, p < 0.001).
Conclusions
The use of nCT leads to a significant increase in overall survival in patients when compared with nCRT for those who achieved cPR in gastric adenocarcinoma. While this study is limited in identifying the cause for this difference in overall survival, this important finding nonetheless requires further investigation and should be considered in the development of future gastric cancer trials.


Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:359–86.
SEER Cancer Statistics Review, 1975–2016. SEER. https://seer.cancer.gov/csr/1975_2016/index.html. Accessed 6 Aug 2019.
Spiegel D, Palta M, Uronis H. Role of chemotherapy and radiation therapy in the management of gastric adenocarcinoma. Surg Clin North Am. 2017;97:421–35.
Cai Z, Yin Y, Shen C, et al. Comparative effectiveness of preoperative, postoperative and perioperative treatments for resectable gastric cancer: a network meta-analysis of the literature from the past 20 years. Surg Oncol. 2018;27:563–74.
National Comprehensive Cancer Network. Gastric Cancer (Version 3.2019). https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf. Accessed 6 Aug 2019.
Cats A, Jansen EPM, van Grieken N, et al. Chemotherapy versus chemoradiotherapy after surgery and preoperative chemotherapy for resectable gastric cancer (CRITICS): an international, open-label, randomised phase 3 trial. Lancet Oncol. 2018;19:616–28.
Hizal M, Sendur MA, Bilgin B, Bulent Akinci M, Sener Dede D, Yalcin B. Expanding treatment options for resectable gastric cancer: is it a countdown for radiotherapy? J BUON. 2019;24:1367–70.
Lane WO, Nussbaum DP, Sun Z, Blazer DG. Preoperative radiation therapy in the surgical management of gastric and junctional adenocarcinoma: should lymph node retrieval guidelines be altered? J Surg Oncol. 2018;117:1708–15.
Newton AD, Datta J, Loaiza-Bonilla A, Karakousis GC, Roses RE. Neoadjuvant therapy for gastric cancer: current evidence and future directions. J Gastrointest Oncol. 2015;6:534–43.
Ott K, Sendler A, Becker K, et al. Neoadjuvant chemotherapy with cisplatin, 5-FU, and leucovorin (PLF) in locally advanced gastric cancer: a prospective phase II study. Gastric Cancer. 2003;6:159–67.
Valenti V, Hernandez-Lizoaín JL, Beorlegui MC, et al. Morbidity, mortality, and pathological response in patients with gastric cancer preoperatively treated with chemotherapy or chemoradiotherapy. J Surg Oncol. 2011;104:124–9.
Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355:11–20.
Adams S, Chakravarthy AB, Donach M, et al. Preoperative concurrent paclitaxel-radiation in locally advanced breast cancer: pathologic response correlates with five-year overall survival. Breast Cancer Res Treat. 2010;124:723–32.
Chavez-MacGregor M, Litton J, Chen H, et al. Pathological complete response in breast cancer patients receiving anthracycline and taxane-based neoadjuvant chemotherapy: evaluating the effect of race/ethnicity. Cancer. 2010;116:4168–77.
Park JW, Kim JH, Choi EK, et al. Prognosis of esophageal cancer patients with pathologic complete response after preoperative concurrent chemoradiotherapy. Int J Radiat Oncol Biol Phys. 2011;81:691–7.
Alnaji RM, Du W, Gabriel E, et al. Pathologic complete response is an independent predictor of improved survival following neoadjuvant chemoradiation for esophageal adenocarcinoma. J Gastrointest Surg. 2016;20:1541–6.
Maas M, Nelemans PJ, Valentini V, et al. Long-term outcome in patients with a pathological complete response after chemoradiation for rectal cancer: a pooled analysis of individual patient data. Lancet Oncol. 2010;11:835–44.
Li Z, Shan F, Wang Y, et al. Correlation of pathological complete response with survival after neoadjuvant chemotherapy in gastric or gastroesophageal junction cancer treated with radical surgery: A meta-analysis. PLoS ONE. 2018;13:0189294.
Cho H, Nakamura J, Asaumi Y, et al. Long-term survival outcomes of advanced gastric cancer patients who achieved a pathological complete response with neoadjuvant chemotherapy: a systematic review of the literature. Ann Surg Oncol. 2015;22:787–92.
Badgwell B, Blum M, Estrella J, et al. Predictors of survival in patients with resectable gastric cancer treated with preoperative chemoradiation therapy and gastrectomy. J Am Coll Surg. 2015;221:83–90.
Fields RC, Strong VE, Gönen M, et al. Recurrence and survival after pathologic complete response to preoperative therapy followed by surgery for gastric or gastrooesophageal adenocarcinoma. Br J Cancer. 2011;104:1840–7.
Ajani JA, Mansfield PF, Crane CH, et al. Paclitaxel-based chemoradiotherapy in localized gastric carcinoma: degree of pathologic response and not clinical parameters dictated patient outcome. J Clin Oncol. 2005;23:1237–44.
Ajani JA, Mansfield PF, Janjan N, et al. Multi-institutional trial of preoperative chemoradiotherapy in patients with potentially resectable gastric carcinoma. J Clin Oncol. 2004;22:2774–80.
Ajani JA, Winter K, Okawara GS, et al. Phase II trial of preoperative chemoradiation in patients with localized gastric adenocarcinoma (RTOG 9904): quality of combined modality therapy and pathologic response. J Clin Oncol. 2006;24:3953–8.
Al-Batran S-E, Hofheinz RD, Pauligk C, et al. Histopathological regression after neoadjuvant docetaxel, oxaliplatin, fluorouracil, and leucovorin versus epirubicin, cisplatin, and fluorouracil or capecitabine in patients with resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4-AIO): results from the phase 2 part of a multicentre, open-label, randomised phase 2/3 trial. Lancet Oncol. 2016;17:1697–708.
Trip AK, Poppema BJ, van Berge Henegouwen MI, et al. Preoperative chemoradiotherapy in locally advanced gastric cancer, a phase I/II feasibility and efficacy study. Radiother Oncol. 2014;112:284–8.
Martin-Romano P, Sola JJ, Diaz-Gonzalez JA, et al. Role of histological regression grade after two neoadjuvant approaches with or without radiotherapy in locally advanced gastric cancer. Br J Cancer. 2016;115:655–63.
Zhang Y, Peng Z, Chen L. Survival analysis of gastric cancer cases with pathological complete response received neoadjuvant chemotherapy. Zhonghua Yi Xue Za Zhi. 2016;96:1582–4.
Stark AP, Ikoma N, Chiang YJ, et al. Characteristics and survival of gastric cancer patients with pathologic complete response to preoperative therapy. Ann Surg Oncol. 2019;26:3602–10.
Spolverato G, Ejaz A, Kim Y, et al. Rates and patterns of recurrence after curative intent resection for gastric cancer: a United States multi-institutional analysis. J Am Coll Surg. 2014;219:664–75.
Nakagawa M, Kojima K, Inokuchi M, et al. Patterns, timing and risk factors of recurrence of gastric cancer after laparoscopic gastrectomy: reliable results following long-term follow-up. Eur J Surg Oncol. 2014;40:1376–82.
Wong RKS, Jang R, Darling G. Postoperative chemoradiotherapy vs. preoperative chemoradiotherapy for locally advanced (operable) gastric cancer: clarifying the role and technique of radiotherapy. J Gastrointest Oncol. 2015;6:89–107.
Fujitani K, Ajani JA, Crane CH, et al. Impact of induction chemotherapy and preoperative chemoradiotherapy on operative morbidity and mortality in patients with locoregional adenocarcinoma of the stomach or gastroesophageal junction. Ann Surg Oncol. 2007;14:2010–7.
Slagter AE, Jansen EPM, van Laarhoven HWM, et al. CRITICS-II: a multicentre randomised phase II trial of neo-adjuvant chemotherapy followed by surgery versus neo-adjuvant chemotherapy and subsequent chemoradiotherapy followed by surgery versus neo-adjuvant chemoradiotherapy followed by surgery in resectable gastric cancer. BMC Cancer. 2018;18:877.
Leong T, Smithers BM, Michael M, et al. TOPGEAR: a randomised phase III trial of perioperative ECF chemotherapy versus preoperative chemoradiation plus perioperative ECF chemotherapy for resectable gastric cancer (an international, intergroup trial of the AGITG/TROG/EORTC/NCIC CTG). BMC Cancer. 2015;15:532.
Leong T, Smithers BM, Haustermans K, et al. TOPGEAR: a randomized, phase III trial of perioperative ECF chemotherapy with or without preoperative chemoradiation for resectable gastric cancer: interim results from an international, intergroup trial of the AGITG, TROG, EORTC and CCTG. Ann Surg Oncol. 2017;24:2252–8.
Amini N, Spolverato G, Kim Y, et al. Clinicopathological features and prognosis of gastric cardia adenocarcinoma: a multi-institutional U.S. study. J Surg Oncol. 2015;111:285–92.
Zhao J, Zhao J, Du F, et al. Cardia and non-cardia gastric cancer have similar stage-for-stage prognoses after R0 resection: a large-scale, multicenter study in China. J Gastrointest Surg. 2016;20:700–7.
Acknowledgment
We acknowledge and thank the American College of Surgeons Committee on Cancer for providing access to the Participant User File from the National Cancer Data Base.
Funding
This work was supported by Roswell Park Cancer Institute and National Cancer Institute (NCI) (Grant No. P30CA016056).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Trumbull, D.A., Lemini, R., Díaz Vico, T. et al. Prognostic Significance of Complete Pathologic Response Obtained with Chemotherapy Versus Chemoradiotherapy in Gastric Cancer. Ann Surg Oncol 28, 766–773 (2021). https://doi.org/10.1245/s10434-020-08921-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-020-08921-9