Abstract
Background
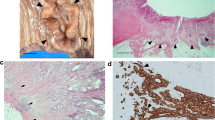
Pancreatic cancer tissue contains abundant stromal components, including extracellular matrix proteins such as tenascin C (TNC), which exists as large (TNC-L) and non-large splice variants. Here, we examined human pancreatic cancer specimens for the expression of total TNC (TNC-ALL) and TNC-L in the stroma and annexin A2 (ANXA2), a cell surface receptor for TNC, and evaluated their significance as prognostic markers for pancreatic cancer.
Methods
Expression of ANXA2, TNC-ALL, and TNC-L was examined in 106 pancreatic cancer tissues from patients who underwent curative resection and who had not received prior therapy or surgery. Protein expression was measured by immunohistochemistry and scored on a semi-quantitative scale. The relationships between protein expression, clinicopathological factors, and prognosis were evaluated by Cox proportional hazards analysis.
Results
TNC-ALL and TNC-L were detected mainly in the stroma, whereas ANXA2 was predominantly expressed in cancer cell membranes. TNC-ALL was also expressed in non-tumor pancreatic tissue. High levels of stromal TNC-L and membranous ANXA2, but not stromal TNC-ALL, were independently associated with cancer progression and poor prognosis. Moreover, high co-expression of stromal TNC-L and membranous ANXA2 was a superior indicator of poor prognosis compared with detection of TNC-ALL, TNC-L, or ANXA2 alone.
Conclusions
Our data suggest that co-expression of stromal TNC-L and membranous ANXA2 is a poor prognostic marker compared with detection of TNC-L or ANXA2 alone for pancreatic cancer patients. Additionally, targeting of crosstalk between stromal TNC and cancer cell ANXA2 could be a promising therapeutic strategy to overcome refractory pancreatic cancer.


Similar content being viewed by others
References
Hidalgo M. Pancreatic cancer. N Engl J Med. 2010;362:1605–17.
Ilic M, Ilic I. Epidemiology of pancreatic cancer. World J Gastroenterol. 2016;22:9694–705.
Qu C, Wang Q, Meng Z, Wang P. Cancer-associated fibroblasts in pancreatic cancer. Integr Cancer Ther. 2018;17:1016–9.
Chen J, Chen Z, Chen M, et al. Role of fibrillary tenascin-C in metastatic pancreatic cancer. Int J Oncol. 2009;34:1029–36.
von Ahrens D, Bhagat TD, Nagrath D, et al. The role of stromal cancer-associated fibroblasts in pancreatic cancer. J Hematol Oncol. 2017;28(10):76.
Sofat N, Robertson SD, Hermansson M, et al. Tenascin-C fragments are endogenous inducers of cartilage matrix degradation. Rheumatol Int. 2012;32:2809–17.
Pas J, Wyszko E, Rolle K, et al. Analysis of structure and function of tenascin-C. Int J Biochem Cell Biol. 2006;38:1594–602.
Tsunoda T, Inada H, Kalembeyi I, et al. Involvement of large tenascin-C splice variants in breast cancer progression. Am J Pathol. 2003;162:1857–67.
Dang C, Gottschling M, Roewert J, et al. Tenascin-C patterns and splice variants in actinic keratosis and cutaneous squamous cell carcinoma. Br J Dermatol. 2006;155:763–70.
Ni WD, Yang ZT, Cui CA, et al. Tenascin-C is a potential cancer-associated fibroblasts marker and predicts poor prognosis in prostate cancer. Biochem Biophys Res Commun. 2017;486:607–12.
Yang ZT, Yeo SY, Yin YX, et al. Tenascin-C, a prognostic determinant of esophageal squamous cell carcinoma. PLoS ONE. 2016;11:e0145807.
Yang Z, Ni W, Cui C, Fang L, Xuan Y. Tenascin C is a prognostic determinant and potential cancer-associated fibroblasts marker for breast ductal carcinoma. Exp Mol Pathol. 2017;102:262–7.
Godier A, Hunt BJ. Plasminogen receptors and their role in the pathogenesis of inflammatory, autoimmune and malignant disease. J Thromb Haemost. 2013;11:26–34.
Ohno Y, Izumi M, Kawamura T, Nishimura T, Mukai K, Tachibana M. Annexin II represents metastatic potential in clear-cell renal cell carcinoma. Br J Cancer. 2009;101:287–94.
Shiozawa Y, Havens AM, Jung Y, et al. Annexin II/annexin II receptor axis regulates adhesion, migration, homing, and growth of prostate cancer. J Cell Biochem. 2008;105:370–80.
Yoneura N, Takano S, Yoshitomi H, et al. Expression of annexin II and stromal tenascin C promotes epithelial to mesenchymal transition and correlates with distant metastasis in pancreatic cancer. Int J Mol Med. 2018;42:821–30.
Chung CY, Erickson HP. Cell surface annexin II is a high affinity receptor for the alternatively spliced segment of tenascin-C. J Cell Biol. 1994;126:539–48.
Esposito I, Penzel R, Chaib-Harrireche M, et al. Tenascin C and annexin II expression in the process of pancreatic carcinogenesis. J Pathol. 2006;208:673–85.
Feng X, Liu H, Zhang Z, Gu Y, Qiu H, He Z. Annexin A2 contributes to cisplatin resistance by activation of JNK-p53 pathway in non-small cell lung cancer cells. J Exp Clin Cancer Res. 2017;36:123.
Gong XG, Lv YF, Li XQ, Xu FG, Ma QY. Gemcitabine resistance induced by interaction between alternatively spliced segment of tenascin-C and annexin A2 in pancreatic cancer cells. Biol Pharm Bull. 2010;33:1261–7.
Foley K, Muth S, Jaffee E, Zheng L. Hedgehog signaling stimulates Tenascin C to promote invasion of pancreatic ductal adenocarcinoma cells through Annexin A2. Cell Adhes Migr. 2017;11:514–23.
Akaike H. Information theory and an extension of the maximum likelihood principle. In: Proceedings of the 2nd international symposium on information theory. 1973. p. 267–81.
Chung CY, Murphy-Ullrich JE, Erickson HP. Mitogenesis, cell migration, and loss of focal adhesions induced by tenascin-C interacting with its cell surface receptor, annexin II. Mol Biol Cell. 1996;7:883–92.
Chiovaro F, Chiquet-Ehrismann R, Chiquet M. Transcriptional regulation of tenascin genes. Cell Adhes Migr. 2015;9:34–47.
Keller KE, Kelley MJ, Acott TS. Extracellular matrix gene alternative splicing by trabecular meshwork cells in response to mechanical stretching. Invest Ophthalmol Vis Sci. 2007;48:1164–72.
Acknowledgment
The authors thank Anne M. O’Rourke, PhD, and H. Nikki March, PhD, from Edanz Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
The authors have no conflicts of interest directly relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
Immunohistochemical staining of stromal TNC-ALL and TNC-L in pancreatic cancer tissue. Left: Positive staining of TNC-ALL. Right: Absence of TNC-L staining. Original magnification ×200 (TIFF 425 kb)
Supplementary Fig. 2
Disease-free survival curves of pancreatic cancer patients stratified by expression of stromal TNC-ALL and TNC-L and tumor cell ANXA2. (a–f) Kaplan–Meier curves showing disease-free survival of patients stratified by expression or co-expression of proteins. a High vs. low stromal TNC-ALL (P = 0.0192). b High vs. low stromal TNC-L (P = 0.001). c Stromal TNC-NL vs. TNC-L expression (P = 0.0030) and TNC-NL vs. TNC-negative (P = 0.4540). d High vs. low membranous expression of ANXA2 (P = 0.0020). e High stromal TNC-ALL and high membranous ANXA2 co-expression vs. low stromal TNC-ALL and low membranous ANXA2 co-expression (P < 0.001). f High stromal TNC-L and high membranous ANXA2 co-expression vs. low stromal TNC-L and low membranous ANXA2 co-expression (P < 0.001) (TIFF 172 kb)
Supplementary Fig. 3
Overall survival and disease-free survival curves of pancreatic cancer patients at each clinical stage (stage I–II and III–IV) stratified by expression of stromal TNC-ALL and TNC-L and cancer cell ANXA2. a Overall survival (stage IA–IIB): high vs. low stromal TNC-ALL (P = 0.017). b Overall survival (stage IA–IIB): high vs. low stromal TNC-L (P ≤ 0.001). c Overall survival (stage IA–IIB): high vs. low membranous expression of ANXA2 (P = 0.0020). d Overall survival (stage III–IV): high vs. low stromal TNC-ALL (P = 0.609). e Overall survival (stage II–IV): high vs. low stromal TNC-L (P = 0.798). f Overall survival (stage III–IV): high vs. low membranous expression of ANXA2 (P = 0.039) (TIFF 170 kb)
Supplementary Fig. 4
Overall survival and disease-free survival curves of pancreatic cancer patients stratified by cytoplasmic expression of TNC-ALL and TNC-L. a Overall survival: high vs. low cytoplasm TNC-ALL (P = 0.669). b Overall survival: high vs. low cytoplasmic TNC-L (P = 0.586). c Disease-free survival: high vs. low cytoplasmic TNC-ALL (P = 0.787). d Disease-free survival: high vs. low cytoplasmic TNC-ALL (P = 0.586) (TIFF 134 kb)
Supplementary Fig. 5
Overall survival curves of pancreatic cancer patients with high or low ANXA2 expression stratified by expression of TNC-ALL and TNC-L in the stroma. a Overall survival (ANXA2 high): high vs. low stromal TNC-ALL (P = 0.060). b Overall survival (ANXA2 high): high vs. low stromal TNC-L (P < 0.001). c Overall survival (ANXA2 low): high vs. low stromal TNC-ALL (P = 0.351). d Overall survival (ANXA2 low): high vs. low stromal TNC-L (P = 0.144) (TIFF 92 kb)
Rights and permissions
About this article
Cite this article
Hagiwara, K., Harimoto, N., Yokobori, T. et al. High Co-expression of Large Tenascin C Splice Variants in Stromal Tissue and Annexin A2 in Cancer Cell Membranes is Associated with Poor Prognosis in Pancreatic Cancer. Ann Surg Oncol 27, 924–930 (2020). https://doi.org/10.1245/s10434-019-07708-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-019-07708-x