Abstract
Background
The treatment of patients with metastatic rectal cancer remains controversial. We developed a reverse strategy, the liver-first approach, to optimize the chance of a curative resection. The aim of this study was to assess rectal outcomes after reverse treatment of patients with metastatic rectal cancer.
Methods
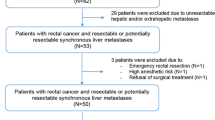
From May 2000 to November 2013, a total of 34 consecutive selected patients with histology-proven adenocarcinoma of the rectum and liver metastases were prospectively entered into a dedicated computerized database. All patients were treated via our reverse strategy. Rectal and overall survival outcomes were analyzed.
Results
Most patients presented with advanced disease (median Fong clinical risk score of 3; range 2–5). One patient failed to complete the whole treatment (3 %). Rectal surgery was performed after a median of 3.9 months (range 0.4–17.8 months). A total of 73.3 % patients received preoperative radiotherapy. Perioperative mortality and morbidity rates were 0 and 27.3 % after rectal surgery. Severe complications were reported in two patients (6.1 %): one anastomotic leak and one systemic inflammatory response syndrome. The median hospital stay was 11 days (range 5–23 days). Complete local pathological response was observed in three patients (9.1 %). The median number of lymph nodes collected was 14. The R0 rate was 93.9 %. There was no positive circumferential margin. After a mean follow-up of 36 months after rectal surgery, 5-year overall survival was 52.5 %. Five patients experienced pelvic recurrence.
Conclusions
In our cohort of selected patients with stage IV rectal cancer, the reverse strategy was not only safe and effective, but also oncologically promising, with a low morbidity rate and high long-term survival.



Similar content being viewed by others
References
Manfredi S, Lepage C, Hatem C, Coatmeur O, Faivre J, Bouvier AM. Epidemiology and management of liver metastases from colorectal cancer. Ann Surg. 2006;244:254–9.
Lemmens VE, de Haan N, Rutten HJ, Martijn H, Loosveld OJ, Roumen RM, et al. Improvements in population-based survival of patients presenting with metastatic rectal cancer in the south of the Netherlands, 1992–2008. Clin Exp Metastasis. 2011;28:283–90.
Simmonds PC, Primrose JN, Colquitt JL, Garden OJ, Poston GJ, Rees M. Surgical resection of hepatic metastases from colorectal cancer: a systematic review of published studies. Br J Cancer. 2006;94:982–99.
Vigano L, Karoui M, Ferrero A, Tayar C, Cherqui D, Capussotti L. Locally advanced mid/low rectal cancer with synchronous liver metastases. World J Surg. 2011;35:2788–95.
Julien LA, Thorson AG. Current neoadjuvant strategies in rectal cancer. J Surg Oncol. 2010;101:321–6.
Slesser AA, Bhangu A, Brown G, Mudan S, Tekkis PP. The management of rectal cancer with synchronous liver metastases: a modern surgical dilemma. Tech Coloproctol. 2013;17:1–12.
Mentha G, Roth AD, Terraz S, Giostra E, Gervaz P, Andres A, et al. “Liver first” approach in the treatment of colorectal cancer with synchronous liver metastases. Dig Surg. 2008;25:430–5.
Gervaz P, Rubbia-Brandt L, Andres A, Majno P, Roth A, Morel P, et al. Neoadjuvant chemotherapy in patients with stage IV colorectal cancer: a comparison of histological response in liver metastases, primary tumors, and regional lymph nodes. Ann Surg Oncol. 2010;17:2714–9.
Mentha G, Majno PE, Andres A, Rubbia-Brandt L, Morel P, Roth AD. Neoadjuvant chemotherapy and resection of advanced synchronous liver metastases before treatment of the colorectal primary. Br J Surg. 2006;93:872–8.
Mentha G, Terraz S, Andres A, Toso C, Rubbia-Brandt L, Majno P. Operative management of colorectal liver metastases. Semin Liver Dis. 2013;33:262–72.
Mentha G, Majno P, Terraz S, Rubbia-Brandt L, Gervaz P, Andres A, et al. Treatment strategies for the management of advanced colorectal liver metastases detected synchronously with the primary tumour. Eur J Surg Oncol. 2007;33(Suppl 2):S76–83.
Verhoef C, van der Pool AE, Nuyttens JJ, Planting AS, Eggermont AM, de Wilt JH. The “liver-first approach” for patients with locally advanced rectal cancer and synchronous liver metastases. Dis Colon Rectum. 2009;52:23–30.
de Rosa A, Gomez D, Hossaini S, Duke K, Fenwick SW, Brooks A, et al. Stage IV colorectal cancer: outcomes following the liver-first approach. J Surg Oncol. 2013;108:444–9.
Jegatheeswaran S, Mason JM, Hancock HC, Siriwardena AK. The liver-first approach to the management of colorectal cancer with synchronous hepatic metastases: a systematic review. JAMA Surg. 2013;148:385–91.
van der Pool AE, de Wilt JH, Lalmahomed ZS, Eggermont AM, Ijzermans JN, Verhoef C. Optimizing the outcome of surgery in patients with rectal cancer and synchronous liver metastases. Br J Surg. 2010;97:383–90.
Tzeng CW, Aloia TA, Vauthey JN, Chang GJ, Ellis LM, Feig BW, et al. Morbidity of staged proctectomy after hepatectomy for colorectal cancer: a matched case–control analysis. Ann Surg Oncol. 2013;20:482–90.
Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg. 1999;230:309–18.
Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13.
Cellini C, Hunt SR, Fleshman JW, Birnbaum EH, Bierhals AJ, Mutch MG. Stage IV rectal cancer with liver metastases: is there a benefit to resection of the primary tumor? World J Surg. 2010;34:1102–8.
Lykoudis PM, O’Reilly D, Nastos K, Fusai G. Systematic review of surgical management of synchronous colorectal liver metastases. Br J Surg. 2014;101:605–12.
Abdalla EK, Bauer TW, Chun YS, D’Angelica M, Kooby DA, Jarnagin WR. Locoregional surgical and interventional therapies for advanced colorectal cancer liver metastases: expert consensus statements. HPB (Oxford). 2013;15:119–30.
Salmenkyla S, Kouri M, Osterlund P, Pukkala E, Luukkonen P, Hyoty M, et al. Does preoperative radiotherapy with postoperative chemotherapy increase acute side-effects and postoperative complications of total mesorectal excision? Report of the randomized Finnish rectal cancer trial. Scand J Surg. 2012;101:275–82.
Buchs NC, Gervaz P, Secic M, Bucher P, Mugnier-Konrad B, Morel P. Incidence, consequences, and risk factors for anastomotic dehiscence after colorectal surgery: a prospective monocentric study. Int J Colorectal Dis. 2008;23:265–70.
Matthiessen P, Hallbook O, Rutegard J, Simert G, Sjodahl R. Defunctioning stoma reduces symptomatic anastomotic leakage after low anterior resection of the rectum for cancer: a randomized multicenter trial. Ann Surg. 2007;246:207–14.
Martin R, Paty P, Fong Y, Grace A, Cohen A, DeMatteo R, et al. Simultaneous liver and colorectal resections are safe for synchronous colorectal liver metastasis. J Am Coll Surg. 2003;197:233–41.
Slesser AA, Simillis C, Goldin R, Brown G, Mudan S, Tekkis PP. A meta-analysis comparing simultaneous versus delayed resections in patients with synchronous colorectal liver metastases. Surg Oncol. 2013;22:36–47.
Bolton JS, Fuhrman GM. Survival after resection of multiple bilobar hepatic metastases from colorectal carcinoma. Ann Surg. 2000;231:743–51.
de Jong MC, van Dam RM, Maas M, Bemelmans MH, Olde Damink SW, Beets GL, et al. The liver-first approach for synchronous colorectal liver metastasis: a 5-year single-centre experience. HPB (Oxford). 2011;13:745–52.
Buchs NC, Gervaz P, Bucher P, Huber O, Mentha G, Morel P. Lessons learned from one thousand consecutive colonic resections in a teaching hospital. Swiss Med Wkly. 2007;137:259–64.
Acknowledgment
We thank Stéphanie Meyer, data manager, for her help in maintaining the database. Christian Toso was supported by a professorship from the Swiss National Science Foundation (PP00P3_139021).
Disclosure
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Buchs, N.C., Ris, F., Majno, P.E. et al. Rectal Outcomes After a Liver-First Treatment of Patients with Stage IV Rectal Cancer. Ann Surg Oncol 22, 931–937 (2015). https://doi.org/10.1245/s10434-014-4069-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-4069-8