Abstract
Background
Primary cutaneous head and neck melanomas (HNM) are reported to be associated with a higher incidence of brain metastasis than trunk and limb melanomas (TLM). In this study, the incidence of brain metastases in patients with HNM and risk factors for the development of brain metastases were analyzed.
Methods
From a large, prospectively-collected database, 1,687 HNM patients and 8,793 TLM patients who presented with American Joint Committee on Cancer (AJCC) stage I and II disease were identified. Survival was assessed using the Kaplan–Meier method and multivariate Cox regression analysis. Independent risk factors were determined by binary logistic regression analysis.
Results
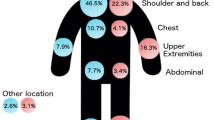
The incidence of brain metastases 5 years after diagnosis of HNM was 6.7 % compared with 4.7 % for brain metastases from TLM (p = 0.003). Patients with scalp melanomas were most likely to develop brain metastases (12.7 %). Independent risk factors for brain metastasis in patients with HNM were Breslow thickness, ulceration, and scalp location.
Conclusion
Patients with primary scalp melanomas had a much higher incidence of brain metastasis than patients with melanomas on other head and neck sites, who in turn had a higher incidence than patients with melanomas on sites elsewhere on the body. More intensive monitoring of patients with scalp melanomas, who are at particularly high risk of brain metastasis, might lead to earlier discovery of metastatic disease in the brain, offering the prospect of earlier intervention and better outcomes.

Similar content being viewed by others
References
Lachiewicz AM, Berwick M, Wiggins CL, Thomas NE. Survival differences between patients with scalp or neck melanoma and those with melanoma of other sites in the surveillance, epidemiology, and end results (SEER) program. Arch Dermatol. 2008;144:515–21.
Law MM, Wong JH. Evaluation of the prognostic significance of the site of origin of cutaneous melanoma. Am Surg. 1994;60:362–6.
Urist MM, Balch CM, Soong SJ, et al. Head and neck melanoma in 534 clinical Stage I patients: a prognostic factors analysis and results of surgical treatment. Ann Surg. 1984;200:769–75.
Zakrzewski J, Geraghty LN, Rose AE, Christos PJ, Mazumdar M, Polsky D, et al. Clinical variables and primary tumor characteristics predictive of the development of melanoma brain metastases and post-brain metastases survival. Cancer. 2011;117:1711–20.
Daryanani D, Plukker JT, de Jong MA, Haaxma-Reiche H, Nap R, Kuiper H, et al. Increased incidence of brain metastases in cutaneous head and neck melanoma. Melanoma Res. 2005;15:119–24.
Sampson JH, Carter JH Jr, Friedman AH, Seigler HF. Demographics, prognosis, and therapy in 702 patients with brain metastases from malignant melanoma. J Neurosurg. 1998;88:11–20.
Fife KM, Colman MH, Stevens GN, Firth IC, Moon D, Shannon KF, et al. Determinants of outcome in melanoma patients with cerebral metastases. J Clin Oncol. 2004;22:1293–300.
Barth A, Wanek LA, Morton DL. Prognostic factors in 1,521 melanoma patients with distant metastases. J Am Coll Surg. 1995;181:193–201.
Balch CM, Gershenwald JE, Soong SJ, Thompson JF, Atkins MB, Byrd DR, et al. Final version of 2009 AJCC melanoma staging and classification. J Clin Oncol. 2009;27:6199–206.
Leong SP, Accortt NA, Essner R, Ross M, Gershenwald JE, Pockaj B, et al. Impact of sentinel node status and other risk factors on the clinical outcome of head and neck melanoma patients. Arch Otolaryngol Head Neck Surg. 2006;132:370–3.
Loree TR, Spiro RH. Cutaneous melanoma of the head and neck. Am J Surg. 1989;158:388–91.
Ozao-Choy J, Hiles JM, Yoon JL, Sim MS, Faries MB, Morton DL. Scalp melanoma: responsible for the poor prognosis of head and neck melanoma? Ann Surg Oncol. 2012;19:S23–4.
Hoersch B, Leiter U, Garbe C. Is head and neck melanoma a distinct entity? A clinical registry-based comparative study in 5702 patients with melanoma. Br J Dermatol. 2006;155:771–7.
Balch CM, Soong SJ, Gershenwald JE, Thompson JF, Reintgen DS, Cascinelli N, et al. Prognostic factors analysis of 17,600 melanoma patients: validation of the American Joint Committee on Cancer melanoma staging system. J Clin Oncol. 2001;19:3622–34.
Richard MA, Grob JJ, Avril MF, Thirion X, Wolkenstein P, Souteyrand P, et al. Melanoma and tumor thickness: challenges of early diagnosis. Arch Dermatol. 1999;135:269–74.
Garbe C, Buttner P, Bertz J, Burg G, d’Hoedt B, Drepper H, et al. Primary cutaneous melanoma: prognostic classification of anatomic location. Cancer. 1995;75:2492–8.
Gillgren P, Brattstrom G, Frisell J, Persson JO, Ringborg U, Hansson J. Effect of primary site on prognosis in patients with cutaneous malignant melanoma: a study using a new model to analyse anatomical locations. Melanoma Res. 2005;15:125–32.
Fogarty G, Morton RL, Vardy J, Nowak AK, Mandel C, Peta M Forder, et al. Whole brain radiotherapy after local treatment of brain metastases in melanoma patients: a randomised phase III trial. BMC Cancer. 2011;11:142.
Wronski M, Arbit E. Surgical treatment of brain metastases from melanoma: a retrospective study of 91 patients. J Neurosurg. 2000;93:9–18.
Lebbe C, McDermott DF, Robert C. Ipilimumab improves survival in previously treated, advanced melanoma patients with poor prognostic factors: subgroup analysis from a phase III trial. 35th ESMO Congress; 8–12 Oct 2010; Milan.
Thompson JF, Hong A, Fogarty G. Publication and interpretation of clinical trial results: the need for caution. Ann Surg Oncol. 2012;19:1745–7.
Long GV, Kefford RF, Carr PJA. Phase 1/2 study of GSK2118436, a selective inhibitor of V600 mutation (mut) BRAF kinase: evidence of activity in melanoma brain metastases (mets). 35th ESMO Congress; 8–12 Oct 2010; Milan.
Lyle M, Long GV. The role of systemic therapies in the management of melanoma brain metastases. Curr Opin Oncol. 2014;26:222–9.
Acknowledgment
Anna M. Huismans received funding from Haak Bastiaanse-Kuneman Stichting, Jan Kornelis de Cock Stichting, Jo Kolk Studiefonds, het Marco Polo fonds, Stichting Sacha Swarttouw-Heijmans, Stichting Groninger Universiteitsfonds, KWF Kankerbestrijding and het VSB fonds.
Conflict of interest
Anna M. Huismans, Lauren E. Haydu, Kerwin F. Shannon, Michael J. Quinn, Robyn P.M. Saw, Andrew J. Spillane, Jonathan R. Stretch, and John F. Thompson have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huismans, A.M., Haydu, L.E., Shannon, K.F. et al. Primary Melanoma Location on the Scalp is an Important Risk Factor for Brain Metastasis: A Study of 1,687 Patients with Cutaneous Head and Neck Melanomas. Ann Surg Oncol 21, 3985–3991 (2014). https://doi.org/10.1245/s10434-014-3829-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-3829-9