Abstract
Background
The aim of this retrospective study was to investigate the incidence of mesenteric lymph node (MLN) involvement, and its prognostic role in advanced ovarian cancer (OC).
Methods
OC patients undergoing rectosigmoid resection during primary debulking surgery or interval debulking surgery were recorded. Progression-free survival (PFS) and overall survival were calculated from the date of diagnosis to the date of relapse/progression, death of disease, or the date of last follow-up.
Results
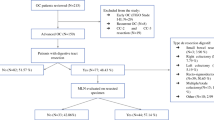
MLNs were detected in 102/148 cases (68.9 %); the rate of MLN involvement was 47.0 %. The percentage of metastatic MLNs was higher in cases with >5 MLNs removed compared with cases with ≤5 MLNs removed (62.7 % vs. 31.3 %; p = 0.0027). A progressive increase in the rate of metastatic MLNs was documented in association with depth of bowel infiltration (p = 0.026). Cases with metastatic MLNs experienced isolated celiac trunk or aortic lymph node recurrences more frequently than patients without MLN involvement (44.8 % vs. 10.7 %; p = 0.0008). PFS did not differ between cases with positive versus negative MLN involvement (2-year PFS = 31 % vs. 43 %; p = 0.58).
Conclusion
OC patients undergoing rectosigmoid resection showed metastatic MLN involvement in 47.0 % of cases. Metastatic MLN status is associated with a high rate of isolated aortic and celiac trunk lymph node recurrences.

Similar content being viewed by others
References
Griffiths CT. Surgical resection of tumor bulk in the primary treatment of ovarian carcinoma. Natl Cancer Inst Monogr. 1975;42:101–4.
Ozols RF, Bundy BN, Greer BE et al; Gynecologic Oncology Group. Phase III trial of carboplatin and paclitaxel compared with cisplatin and paclitaxel in patients with optimally resected stage III ovarian cancer: a Gynecologic Oncology Group study J Clin Oncol. 2003;21(17):3194–3200.
Fader AN, Rose PG. Role of surgery in ovarian carcinoma. J Clin Oncol. 2007;25:2873–2883.
Eisenkop SM, Spirtos NM. Procedures required to accomplish complete cytoreduction of ovarian cancer: is there a correlation with ‘Biological Aggressiveness’ and survival? Gynecol Oncol. 2001;82:435–441.
Bristow RE, Berek JS. Surgery for ovarian cancer: how to improve survival. Lancet. 2006;367(9522):1558–60.
Bristow RE, Tomacruz RS, Armstrong DK, et al. Survival effect of maximal cytoreductive surgery for advanced ovarian carcinoma during the platinum era: a meta-analysis. J Clin Oncol. 2002;20:1248–1259.
Hudson CN, Chir M. Surgical treatment of ovarian cancer. Gynecol Oncol. 1973;1:370–378.
Scarabelli C, Gallo A, Franceschi S, et al. Primary cytoreductive surgery with rectosigmoid colon resection for patients with advanced epithelial ovarian carcinoma. Cancer. 2000;88:389–397.
Obermair A, Hagenauer S, Tamandl D, et al. Safety and efficacy of low anterior en bloc resection as part of cytoreductive surgery for patients with ovarian cancer. Gynecol Oncol. 2001;83:115–120.
Hertel H, Diebolder H, Herrmann J, et al. Is the decision for colorectal resection justified by histopathologic findings: a prospective study of 100 patients with advanced ovarian cancer. Gynecol Oncol. 2001;83:481–484.
Bristow RE, Garmen MG, Kaufman HS, et al. Radical oophorectomy with primary stapled colorectal anastomosis for resection of locally advanced epithelial ovarian cancer. J Am Coll Surg. 2003;197:565–574.
Mourton SM, Temple LK, Abu-Rustum NR, et al. Morbidity of rectosigmoid resection and primary anastomosis in patients undergoing primary cytoreductive surgery for advanced epithelial ovarian cancer. Gynecol Oncol. 2005;99:608–614.
Park JY, Seo S-S, Karg S, et al. The benefits of low anterior en bloc resection as part of cytoreductive surgery for advanced primary and recurrent epithelial ovarian cancer patients outweigh morbidity concerns. Gynecol Oncol. 2006;103:977–84.
Juretzka MM, Barakat RR. Pelvic cytoreduction with rectosigmoid resection. Gynecol Oncol. 2007;104(2 Suppl. 1):40–4.
Gallotta V, Fanfani F, Vizzielli G, Panico G, Rossitto C, Gagliardi ML, et al. Douglas peritonectomy compared to recto-sigmoid resection in optimally cytoreduced advanced ovarian cancer patients: analysis of morbidity and oncological outcome. Eur J Surg Oncol. 2011;37(12):1085–92.
Chang SJ, Bristow RE, Ryu HS. Prognostic significance of systematic lymphadenectomy as part of primary debulking surgery in patients with advanced ovarian cancer. Gynecol Oncol. 2012;126(3):381–6.
Fagotti A, De Iaco P, Fanfani F, Vizzielli G, Perelli F, Pozzati F, et al. Systematic pelvic and aortic lymphadenectomy in advanced ovarian cancer patients at the time of interval debulking surgery: a double-institution case-control study. Ann Surg Oncol. 2012;19(11):3522–7.
Du Bois A, Reuss A, Harter P, et al. Potential role of lymphadenectomy in advanced ovarian cancer: a combined exploratory analysis of three prospective randomized phase III multicenter trials. J Clin Oncol. 2010;28:1733–9.
Kim HS, Ju W, Jee BC, Kim YB, Park NH, Song YS, et al. Systematic lymphadenectomy for survival in epithelial ovarian cancer: a meta-analysis. Int J Gynecol Cancer. 2010;20:520–8.
O’Hanlan KA, Kargas S, Schreiber M, et al. Ovarian carcinoma metastases to gastrointestinal tract appear to spread like colon carcinoma: implications for surgical resection. Gynecol Oncol. 1995;59:200–206.
McFarlane JK, Ryall RD, Heald RS. Mesorectal excision for rectal cancer. Lancet. 1993; 341:457–60.
Lax SF, Petru E, Holzer E, et al. Mesenteric and mesocolic lymph node metastases from ovarian carcinoma: a clinicopathological analysis. Int J Gynecol Cancer. 1998;8:119–823.
Salani R, Diaz-Montes T, Giuntoli RL, Bristow RE. Surgical management of mesenteric lymph node metastasis in patients undergoing rectosigmoid colectomy for locally advanced ovarian carcinoma. Ann Surg Oncol. 2007;14(12):3552–7.
Baiocchi G, Cestari LA, Macedo MP, Oliveira RA, Fukazawa EM, Faloppa CC, et al. Surgical implications of mesenteric lymph node metastasis from advanced ovarian cancer after bowel resection. J Surg Oncol. 2011;104(3):250–4.
Gouy S, Goetgheluck J, Uzan C, Duclos J, Duvillard P, Morice P. Prognostic factors for and prognostic value of mesenteric lymph node involvement in advanced-stage ovarian cancer. Eur J Surg Oncol. 2012;38(2):170–5.
Zivanovic O, Sima CS, Iasonos A, Hoskins WJ, Pingle PR, et al. The effect of primary cytoreduction on outcomes of patients with FIGO stage IIIC ovarian cancer stratified by the initial tumor burden in the upper abdomen cephalad to the greater omentum. Gynecol Oncol. 2010;116(3):351–7.
Song YJ, Lim MC, Kang S, Seo SS, Kim SH, Han SS, et al. Extended cytoreduction of tumor at the porta hepatis by an interdisciplinary team approach in patients with epithelial ovarian cancer. Gynecol Oncol. 2011;121(2):253–7.
Kaplan E, Meyer P. Non parametric estimation from incomplete observations. J Am Statist Assoc. 1958;53:457–81.
Mantel N. Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep. 1966;50:163–70.
Dvoretsky PM, Richards KA, Angel C, Rabinowitz L, Stoler MH, Beecham JB, et al. Distribution of disease at autopsy in 100 women with ovarian cancer. Hum Pathol. 1988;19(1):57–63.
McDaniel KP, Charnsangavej C, DuBrow RA, Varma DG, Granfield CA, Curley SA. Pathways of nodal metastasis in carcinomas of the cecum, ascending colon, and transverse colon: CT demonstration. AJR Am J Roentgenol. 1993;161(1):61–4.
Lowden S, Heath T. Lymphatic drainage from the distal small intestine in sheep. J Anat. 1993;183 (Pt 1):13–20.
Martinez A, Pomel C, Mery E, Querleu D, Gladieff L, Ferron G. Celiac lymph node resection and porta hepatis disease resection in advanced or recurrent epithelial ovarian, fallopian tube, and primary peritoneal cancer. Gynecol Oncol. 2011;121(2):258-63.
Conflict of interest
The Authors declare they have no conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gallotta, V., Fanfani, F., Fagotti, A. et al. Mesenteric Lymph Node Involvement in Advanced Ovarian Cancer Patients Undergoing Rectosigmoid Resection: Prognostic Role and Clinical Considerations. Ann Surg Oncol 21, 2369–2375 (2014). https://doi.org/10.1245/s10434-014-3558-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-3558-0