Abstract
Background
Merkel cell carcinoma (MCC) is a rare cutaneous malignancy. Few single-institution series have been reported.
Methods
Review of MCC patients treated at our institution between 1980 and 2010. Patient, tumor, and treatment variables were analyzed to determine MCC-specific outcomes.
Results
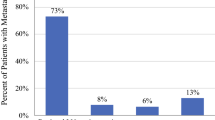
We identified 161 patients with MCC. There was a 2.5-fold increase in cases over the last decade. Median length of follow-up was 36 months. Stage at diagnosis was I in 35 %, II in 21 %, IIIa in 12 %, IIIb in 23 %, and IV in 9 %. The 5-year MCC-specific survival rates were 87, 63, 42, and 0 % for stages I, II, III, and IV, respectively. Death from the disease occurred in 10 % of patients with T1 and in 50 % with larger lesions. One-third of patients presented with nodal disease. Sentinel lymph node biopsy (SLNB) identified micrometastases in 9 out of 27 (33 %) early-stage patients. Recurrence developed in 56 % of SLNB-positive and 39 % of SLNB-negative patients. Half of patients recurred after a median time of 9 months. Proportions of first recurrence location were distant (52 %), nodal (27 %), and local (21 %). Adjuvant treatments did not improve recurrence or survival rates. One-third of patients died of the disease.
Conclusions
SLNB identifies micrometastasis in one-third of early-stage patients. Negative SLNB may predict for improved but not necessarily favorable outcome. Initial tumor size and clinical nodal disease predict for poor outcome. High recurrence rates warrant the development of more effective adjuvant therapies, and better markers of recurrence and treatment response for MCC are needed.





Similar content being viewed by others
References
Albores-Saavedra J, Batich K, Chable-Montero F, Sagy N, Schwartz AM, Henson DE. Merkel cell carcinoma demographics, morphology, and survival based on 3870 cases: a population based study. J Cutan Pathol. 2010;37:20–7.
Agelli M, Clegg LX, Becker JC, Rollison DE. The etiology and epidemiology of Merkel cell carcinoma. Curr Probl Cancer. 2010;34:14–37.
Agelli M, Clegg LX. Epidemiology of primary Merkel cell carcinoma in the United States. J Am Acad Dermatol. 2003;49:832–41.
Hodgson NC. Merkel cell carcinoma: changing incidence trends. J Surg Oncol. 2005;89:1–4.
Ramahi E, Choi J, Fuller CD, Eng TY. Merkel cell carcinoma. Am J Clin Oncol. 2011 (in press).
Ott MJ, Tanabe KK, Gadd MA, et al. Multimodality management of Merkel cell carcinoma. Arch Surg. 1999; 134:388–92; discussion 92–3
Medina-Franco H, Urist MM, Fiveash J, Heslin MJ, Bland KI, Beenken SW. Multimodality treatment of Merkel cell carcinoma: case series and literature review of 1024 cases. Ann Surg Oncol. 2001;8:204-8.
Warner RE, Quinn MJ, Hruby G, Scolyer RA, Uren RF, Thompson JF. Management of merkel cell carcinoma: the roles of lymphoscintigraphy, sentinel lymph node biopsy and adjuvant radiotherapy. Ann Surg Oncol. 2008;15:2509–18.
Muller A, Keus R, Neumann N, Lammering G, Schnabel T. Management of Merkel cell carcinoma: case series of 36 patients. Oncol Rep. 2003;10:577–85.
Allen PJ, Zhang ZF, Coit DG. Surgical management of Merkel cell carcinoma. Ann Surg. 1999;229:97–105.
Boyer JD, Zitelli JA, Brodland DG, D’Angelo G. Local control of primary Merkel cell carcinoma: review of 45 cases treated with Mohs micrographic surgery with and without adjuvant radiation. J Am Acad Dermatol. 2002;47:885–92.
Gillenwater AM, Hessel AC, Morrison WH, et al. Merkel cell carcinoma of the head and neck: effect of surgical excision and radiation on recurrence and survival. Arch Otolaryngol Head Neck Surg. 2001;127:149–54.
Meeuwissen JA, Bourne RG, Kearsley JH. The importance of postoperative radiation therapy in the treatment of Merkel cell carcinoma. Int J Radiat Oncol Biol Phys. 1995;31:325–31.
Morrison WH, Peters LJ, Silva EG, Wendt CD, Ang KK, Goepfert H. The essential role of radiation therapy in securing locoregional control of Merkel cell carcinoma. Int J Radiat Oncol Biol Phys. 1990;19:583–91.
Bajetta E, Celio L, Platania M, et al. Single-institution series of early-stage Merkel cell carcinoma: long-term outcomes in 95 patients managed with surgery alone. Ann Surg Oncol. 2009;16:2985–93.
Lewis KG, Weinstock MA, Weaver AL, Otley CC. Adjuvant local irradiation for Merkel cell carcinoma. Arch Dermatol. 2006;142:693–700.
Mojica P, Smith D, Ellenhorn JD. Adjuvant radiation therapy is associated with improved survival in Merkel cell carcinoma of the skin. J Clin Oncol. 2007;25:1043–7.
Edge SB BD, Compton CC, et al. american joint committee on cancer: AJCC cancer staging manual. 7th ed. pp 315–23. New York, NY: Springer; 2010.
Allen PJ, Bowne WB, Jaques DP, Brennan MF, Busam K, Coit DG. Merkel cell carcinoma: prognosis and treatment of patients from a single institution. J Clin Oncol. 2005;23:2300–9.
Hui AC, Stillie AL, Seel M, Ainslie J. Merkel cell carcinoma: 27-year experience at the Peter MacCallum cancer centre. Int J Radiat Oncol Biol Phys. 2010;80(5):1430–5.
Fields RC, Busam KJ, Chou JF, et al. Five hundred patients with Merkel cell carcinoma evaluated at a single institution. Ann Surg.254:465–73; discussion 73–5.
Toker C. Trabecular carcinoma of the skin. Arch Dermatol. 1972;105:107–10.
Tang CK, Toker C. Trabecular carcinoma of the skin: further clinicopathologic and ultrastructural study. Mt Sinai J Med. 1979;46:516–23.
Toker C. Trabecular carcinoma of the skin. A question of title. Am J Dermatopathol. 1982;4:497–500.
Moll R, Lowe A, Laufer J, Franke WW. Cytokeratin 20 in human carcinomas. A new histodiagnostic marker detected by monoclonal antibodies. Am J Pathol. 1992;140:427–47.
Scott MP, Helm KF. Cytokeratin 20: a marker for diagnosing Merkel cell carcinoma. Am J Dermatopathol. 1999;21:16–20.
Hanly AJ, Elgart GW, Jorda M, Smith J, Nadji M. Analysis of thyroid transcription factor-1 and cytokeratin 20 separates merkel cell carcinoma from small cell carcinoma of lung. J Cutan Pathol. 2000;27:118–20.
Allen PJ, Busam K, Hill AD, Stojadinovic A, Coit DG. Immunohistochemical analysis of sentinel lymph nodes from patients with Merkel cell carcinoma. Cancer. 2001;92:1650–5.
Andea AA, Coit DG, Amin B, Busam KJ. Merkel cell carcinoma: histologic features and prognosis. Cancer. 2008;113:2549–58.
Heath M, Jaimes N, Lemos B, et al. Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol. 2008;58:375–81.
Guler-Nizam E, Leiter U, Metzler G, Breuninger H, Garbe C, Eigentler TK. Clinical course and prognostic factors of Merkel cell carcinoma of the skin. Br J Dermatol. 2009;161:90–4.
Lemos BD, Storer BE, Iyer JG, et al. Pathologic nodal evaluation improves prognostic accuracy in Merkel cell carcinoma: analysis of 5823 cases as the basis of the first consensus staging system. J Am Acad Dermatol. 2010;63:751–61.
Pfeifer T, Weinberg H, Brady MS. Lymphatic mapping for Merkel cell carcinoma. J Am Acad Dermatol. 1997;37:650–1.
Ames SE, Krag DN, Brady MS. Radiolocalization of the sentinel lymph node in Merkel cell carcinoma: a clinical analysis of seven cases. J Surg Oncol. 1998;67:251–4.
Hill AD, Brady MS, Coit DG. Intraoperative lymphatic mapping and sentinel lymph node biopsy for Merkel cell carcinoma. Br J Surg. 1999;86:518–21.
Gupta SG, Wang LC, Penas PF, Gellenthin M, Lee SJ, Nghiem P. Sentinel lymph node biopsy for evaluation and treatment of patients with Merkel cell carcinoma: The Dana-Farber experience and meta-analysis of the literature. Arch Dermatol. 2006;142:685–90.
Stokes JB, Graw KS, Dengel LT, et al. Patients with Merkel cell carcinoma tumors < or = 1.0 cm in diameter are unlikely to harbor regional lymph node metastasis. J Clin Oncol. 2009;27:3772–7.
Fields RC, Busam KJ, Chou JF, et al. Recurrence and survival in patients undergoing sentinel lymph node biopsy for Merkel cell carcinoma: analysis of 153 patients from a single institution. Ann Surg Oncol. 2011.
Schwartz JL, Griffith KA, Lowe L, et al. Features predicting sentinel lymph node positivity in Merkel cell carcinoma. J Clin Oncol. 2011;29:1036-41.
Veness MJ. Merkel cell carcinoma: improved outcome with the addition of adjuvant therapy. J Clin Oncol. 2005;23:7235-6; author reply 7–8.
Clark JR, Veness MJ, Gilbert R, O’Brien CJ, Gullane PJ. Merkel cell carcinoma of the head and neck: is adjuvant radiotherapy necessary? Head Neck. 2007;29:249–57.
Foote M, Harvey J, Porceddu S, et al. Effect of radiotherapy dose and volume on relapse in Merkel cell cancer of the skin. Int J Radiat Oncol Biol Phys. 2010;77:677–84.
Poulsen M, Round C, Keller J, Tripcony L, Veness M. Factors influencing relapse-free survival in Merkel cell carcinoma of the lower limb–a review of 60 cases. Int J Radiat Oncol Biol Phys. 2010;76:393–7.
Veness M, Foote M, Gebski V, Poulsen M. The role of radiotherapy alone in patients with Merkel cell carcinoma: reporting the Australian experience of 43 patients. Int J Radiat Oncol Biol Phys. 2010;78:703–9.
Merkel cell carcinoma. NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. http://www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed 4/23/2011.
Fields RC, Busam KJ, Chou JF, et al. Recurrence after complete resection and selective use of adjuvant therapy for stage I through III Merkel cell carcinoma. Cancer;118(13):3311–20. doi:10.1002/cncr.26626.
Voog E, Biron P, Martin JP, Blay JY. Chemotherapy for patients with locally advanced or metastatic Merkel cell carcinoma. Cancer. 1999;85:2589–95.
Tai PT, Yu E, Winquist E, et al. Chemotherapy in neuroendocrine/Merkel cell carcinoma of the skin: case series and review of 204 cases. J Clin Oncol. 2000;18:2493–9.
Poulsen M, Rischin D, Walpole E, et al. Analysis of toxicity of Merkel cell carcinoma of the skin treated with synchronous carboplatin/etoposide and radiation: a Trans-Tasman Radiation Oncology Group study. Int J Radiat Oncol Biol Phys. 2001;51:156–63.
Poulsen M, Rischin D, Walpole E, et al. High-risk Merkel cell carcinoma of the skin treated with synchronous carboplatin/etoposide and radiation: a Trans-Tasman Radiation Oncology Group Study–TROG 96:07. J Clin Oncol. 2003;21:4371–6.
Poulsen MG, Rischin D, Porter I, et al. Does chemotherapy improve survival in high-risk stage I and II Merkel cell carcinoma of the skin? Int J Radiat Oncol Biol Phys. 2006;64:114–9.
Poulsen M, Walpole E, Harvey J, et al. Weekly carboplatin reduces toxicity during synchronous chemoradiotherapy for Merkel cell carcinoma of skin. Int J Radiat Oncol Biol Phys. 2008;72:1070–4.
Feng H, Shuda M, Chang Y, Moore PS. Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science. 2008;319:1096–100.
Kassem A, Schopflin A, Diaz C, et al. Frequent detection of Merkel cell polyomavirus in human Merkel cell carcinomas and identification of a unique deletion in the VP1 gene. Cancer Res. 2008;68:5009–13.
Becker JC, Houben R, Ugurel S, Trefzer U, Pfohler C, Schrama D. MC polyomavirus is frequently present in Merkel cell carcinoma of European patients. J Invest Dermatol. 2009;129:248–50.
Varga E, Kiss M, Szabo K, Kemeny L. Detection of Merkel cell polyomavirus DNA in Merkel cell carcinomas. Br J Dermatol. 2009;161:930–2.
Duncavage EJ, Zehnbauer BA, Pfeifer JD. Prevalence of Merkel cell polyomavirus in Merkel cell carcinoma. Mod Pathol. 2009;22:516–21.
Busam KJ, Jungbluth AA, Rekthman N, et al. Merkel cell polyomavirus expression in merkel cell carcinomas and its absence in combined tumors and pulmonary neuroendocrine carcinomas. Am J Surg Pathol. 2009;33:1378–85.
Nardi V, Song Y, Santamaria-Barria JA, et al. Activation of PI3K signaling in merkel cell carcinoma. Clin Cancer Res. 18:1227–36. doi:1078-0432.CCR-11-2308.
Hafner C, Houben R, Baeurle A, et al. Activation of the PI3 K/AKT Pathway in Merkel Cell Carcinoma. PLoS One. 2012;7(2):e31255.
Disclosures
The authors have no potential conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Santamaria-Barria, J.A., Boland, G.M., Yeap, B.Y. et al. Merkel Cell Carcinoma: 30-Year Experience from a Single Institution. Ann Surg Oncol 20, 1365–1373 (2013). https://doi.org/10.1245/s10434-012-2779-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2779-3