Abstract
Introduction
Breast cancer is currently viewed as a heterogeneous disease made up of various subtypes, with distinct differences in prognosis. Our goal was to study the distribution and to characterize the clinical and biological factors that influence the behavior and clinical management of the different molecular breast cancer subtypes in premenopausal African-American women.
Methods
A retrospective analysis of Howard University Hospital tumor registry, for all premenopausal African-American women aged less than 50 years, diagnosed with breast cancer from 1998–2005, was performed.
Results
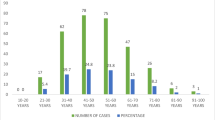
The luminal A subtype was the most prevalent (50.0%), vs basal-cell-like (23.2%), luminal B (14.1%), and HER-2/neu (12.7%). However when stratified by age groups, results showed that in the age group <35 years the basal-cell-like subtype was the most prevalent (55.6%), vs 25.9%, 14.8%, and 5.6% for luminal A, luminal B, and HER-2/neu subtypes, respectively (P < .000). P53 mutation was more prevalent in the basal-cell-like subtype compared to luminal A (48.0% vs 18.6%, P < .01).
The expression of the Bcl-2 gene differed by subtype, with the luminal A and luminal B subtypes more likely to overexpress the Bcl-2 gene (89.1% luminal A, 80.0% luminal B vs 47.6% basal-cell-like and 40.0% HER-2/neu, P < .000). Though not statistically significant, HER-2/neu and basal-cell-like subtypes had the shortest survival time (P < .31).
Conclusion
The high prevalence of the basal-cell-like subtype in young premenopausal African-American women aged <35 years may contribute to the poorer prognosis observed in this cohort of African-American women.


Similar content being viewed by others
References
Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Incidence—SEER 13 Regs Public-Use, Nov 2005 Sub (1992–2003), National Cancer Institute, DCCPS, Surveillance Research Program, Cancer Statistics Branch, released April 2006, based on the Nov 2005 submission
Swanson GM, Haslam SZ, Azzouz F. Breast cancer among young African-American women. Cancer 2003; 97(1 suppl):273–79
Clarke CA, West DW, Edwards BK, Figgs LW, Kerner J, Schwartz AG. Existing data on breast cancer in African-American women: what we know and what we need to know. Cancer 2003; 97(1 suppl):211–21
Ries LAG, Eisner MP, Kosary CL, et al. SEER Cancer Statistics Review, 1975–2002. Bethesda, MD: National Cancer Institute; 2005
Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Mortality—All COD, Public-Use With State, Total U.S. for Expanded Races/Hispanics (1990–2003), National Cancer Institute, DCCPS, Surveillance Research Program, Cancer Statistics Branch, released April 2006. Underlying mortality data provided by NCHS (www.cdc.gov/nchs)
Bain RP, Greenberg RS, Whitaker JP. Racial differences in survival of women with breast cancer. J Chronic Dis 1986; 39:631–42
Dayal HH, Power RN, Chiu C. Race and socio-economic status in survival from breast cancer. J Chronic Dis 1982; 35:675–83
Bassett MT, Krieger N. Social class and black-white differences in breast cancer. Am J Public Health 1986; 76:1400–3
Vastag B. Breast cancer racial gap examined: no easy answers to explain disparities in survival. JAMA 2003; 290:1838–42
Eley JW, Hill HA, Chen VW, et al. Racial differences in survival from breast cancer: results of the National Cancer Institute Black/White Cancer Survival Study. JAMA 1994; 272:947–54
Elledge RM, Clark GM, Chamness GC, et al. Tumor biologic factors and breast cancer prognosis among white, Hispanic, and black women in the United States. J Natl Cancer Inst 1994; 86:705–12
Polite BN, Olopade OI. Breast cancer and race: a rising tide does not lift all boats equally. Perspect Biol Med 2005; 48(1 Suppl):S166–S175
Furberg H, Millikan R, Dressler L, Newman B, Geradts J. Tumor characteristics in African American and white women. Breast Cancer Res Treat 2001; 68:33–43
Porter PL, Lund MJ, Lin MG, et al. Racial differences in the expression of cell cycle-regulatory proteins in breast carcinoma. Cancer 2004; 100:2533–42
Perou CM, Sorlie T, Eisen MB, et al. Molecular portraits of human breast tumours. Nature 2000; 406:747–52
Sorlie T, Perou CM, Tibshirani R, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 2001; 98:10869–74
Sorlie T, Tibshirani R, Parker J, et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci USA 2003; 100:8418–23
Nielsen TO, Hsu FD, Jensen K, et al. Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res 2004; 10:5367–74
Carey LA, Perou CM, Livasy CA, et al. Race, Breast Cancer Subtypes, and Survival in the Carolina Breast Cancer Study. JAMA 2006; 295:2492–2502
Fitzgibbons PL, Page DL, Weaver D, et al. Prognostic factors in breast cancer, college of American Pathologists Consensus Statement 1999. Arch Pathol Lab Med 2000; 124:966–78
Carey LA, Dees EC, Sawyer LR, et al. The triple negative paradox: Primary tumor chemosensitivity of Breast Cancer Subtypes. Clin Cancer Res 2007; 13(8):2329–34
Livasy CA, Karaca G, Nanda R, et al. Phenotypic evaluation of the basal-like subtype of invasive breast carcinoma. Mod Pathol 2006; 2:264–71
Thor AD, Moore DM, Edgerton SM, et al. Accumulation of p53 tumor suppressor gene protein: an independent marker of prognosis in breast cancers. J Natl Cancer Inst 1992; 84:845–55
Davidoff AM, Humphrey PA, Iglehart JD, Marks JR. Genetic basis for p53 overexpression in human breast cancer. Proc Natl Acad Sci USA 1991; 88:5006–10
Banks L, Matlashewski G, Crawford L. Isolation of human-p53-specific monoclonal antibodies and their use in the studies of human p53 expression. Eur J Biochem 1986; 159:529–34
Bartek J, Bartkova J, Vojtesek B, et al. Patterns of expression of p53 tumour suppressor in human breast tissues and tumours in situ and in vitro. Int J Cancer 1990; 46:839–44
Histological Typing of Breast Tumours, 2nd ed. World Health Organization, Geneva, Switzerland, 1981). International Classification of Tumours
Fitzgibbons PL, Connolly JL, Page DL. Updated protocol for the examination of specimens from patients with carcinomas of the breast, a basis for checklists. Arch Pathol Lab Med 2000; 124:1026–33
Rolland P, Spendlove I, Madjid Z, Rakha EA, Patel P, Ellis IO, Durrant L. The p53 positive Bcl-2 negative phenotype is an independent marker of prognosis in breast cancer. Int J Cancer 2007; 120:1311–7
Horita K, Yamaguchi A, Hirose K, Ishida M, Noriki S, Imamura Y, Fukuda M. Prognostic factors affecting disease-free survival rate following surgical resection of primary breast cancer. Eur J Histochem 2001; 45:73–84
Silvestrini R, Benini E, Veneroni S, Daidone MG, Tomasic G, Squicciarini P, Salvadori B. p53 and bcl-2 expression correlates with clinical outcome in a series of node-positive breast cancer patients. J Clin Oncol 1996; 14:1604–10
Fisher B, Anderson S, Bryant J, et al. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med 2002; 347(16):1233–41
Veronesi U, Cascinelli N, Mariani L, et al. Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N Engl J Med 2002; 347(16):1227–32
Haffty BG, Yang Q, Reiss M, et al. Locoregional and distant metastasis in conservatively managed triple negative early-stage breast cancer. J Clin Oncol 2006; 24:5652–7
Romond EH, Perez EA, Bryant J, et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Eng J Med 2005; 353:1673–84
Guarneri V, Broglio K, Kau SW, et al. Prognostic value of pathologic complete response after primary chemotherapy in relation to hormone receptor status and other factors. J Clin Oncol 2006; 24:1037–44
Urruticoechea A, Smith IE, Dowsett M. Proliferation marker Ki-67 in early breast cancer. J Clin Oncol 2005; 23:7212–20
Rouzier R, Perou CM, Symmans WF, et al. Breast cancer molecular subtypes respond differently to preoperative chemotherapy. Clin Cancer Res 2005; 11:5678–85
Acknowledgment
The Susan G. Komen for the cure, for establishing the Dr LaSalle D. Leffall Jr Komen Fellowship in Health Disparities. Dr C. Okasi for her editorial contribution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ihemelandu, C.U., Leffall, L.D., Dewitty, R.L. et al. Molecular Breast Cancer Subtypes in Premenopausal African-American Women, Tumor Biologic Factors and Clinical Outcome. Ann Surg Oncol 14, 2994–3003 (2007). https://doi.org/10.1245/s10434-007-9477-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-007-9477-6