Abstract
Background
The survival of patients with stage IV gastric cancer is poor due to frequent peritoneal failure. The aim of this study was to investigate the impact of early postoperative intraperitoneal chemotherapy (EPIC) after cytoreductive surgery on the long-term survival of these patients, as determined by residual disease status.
Methods
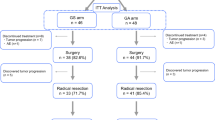
A total of 154 patients with stage IV gastric cancer were enrolled in our study. All patients underwent potentially curative or palliative resections. After surgery, the residual disease states of the patients were recorded. All patients received EPIC.
Results
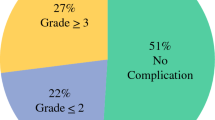
Of all 154 patients, R0 resection was achieved in 37, R1 in 56, and R2 in 61. All patients received a mean of 4.3 EPIC perfusions. After a mean followup period of 29 months, 14 patients remained alive. The median survival of all 154 patients was 11.4 months. Survival times were analyzed according to the type of residual tumor; the median survival time was 25.5 months in the R0 group, 15.6 months in the R1 group, and 7.2 months in the R2 group (p < .001). Upon multivariate analysis, the residual tumor states and the cycle of EPIC perfusion were found to be independent prognostic predictors (p < .001 and p = .018, respectively).
Conclusions
The residual tumor status is the most important predictor for the survival of very advanced gastric cancer patients who received cytoreductive surgery and EPIC. Therefore, complete cytoreductive surgery yielding R0 resection is mandatory for achieving the beneficial effects of EPIC.




Similar content being viewed by others
References
Jeung HC, Rha SY, Jang WI, Noh SH, Chung HC. Treatment of advanced gastric cancer by palliative gastrectomy, cytoreductive therapy and postoperative intraperitoneal chemotherapy. Br J Surg 2002; 89:460–466
Yoo CH, Noh SH, Shin DW, Choi SH, Min JS. Recurrence following curative resection for gastric carcinoma. Br J Surg 2000; 87:236–242
Yonemura Y, Kawamura T, Bandou E, Takahashi S, Sawa T, Matsuki N. Treatment of peritoneal dissemination from gastric cancer by peritonectomy and chemohyperthermic peritoneal perfusion. Br J Surg 2005; 92:370–375
Chu DZ, Lang NP, Thompson C, Osteen PK, Westbrook KC. Peritoneal carcinomatosis in nongynecologic malignancy. A prospective study of prognostic factors. Cancer 1989; 63:364–367
Hermans J, Bonenkamp JJ, Boon MC, Bunt AM, Ohyama S, Sasako M, Van de Velde CJ. Adjuvant therapy after curative resection for gastric cancer: meta-analysis of randomized trials. J Clin Oncol 1993; 11:1441–1447
Janunger KG, Hafstrom L, Glimelius B. Chemotherapy in gastric cancer: a review and updated meta-analysis. Eur J Surg. 2002;168:597–608
Dedrick RL, Myers CE, Bungay PM, DeVita VT Jr. Pharmacokinetic rationale for peritoneal drug administration in the treatment of ovarian cancer. Cancer Treat Rep 1978; 62:1–11
Yonemura Y, Ninomiya I, Kaji M, et al. Prophylaxis with intraoperative chemohyperthermia against peritoneal recurrence of serosal invasion-positive gastric cancer. World J Surg 1995; 19:450–455
Cunliffe WJ, Sugarbaker PH. Gastrointestinal malignancy: rationale for adjuvant therapy using early postoperative intraperitoneal chemotherapy. Br J Surg 1989; 76:1082–1090
Noh SH, Yoo CH, Chung HC, Roh JK, Shin DW, Min JS. Early postoperative intraperitoneal chemotherapy with mitomycin C, 5-fluorouracil and cisplatin for advanced gastric cancer. Oncology 2001; 60:24–30
Verwaal VJ, van Ruth S, Witkamp A, Boot H, van Slooten G, Zoetmulder FA. Long-term survival of peritoneal carcinomatosis of colorectal origin. Ann Surg Oncol 2005; 12:65–71
Yonemura Y, Fujimura T, Nishimura G, et al. Effects of intraoperative chemohyperthermia in patients with gastric cancer with peritoneal dissemination. Surgery 1996; 119:437–444
Shen P, Hawksworth J, Lovato J, et al. Cytoreductive surgery and intraperitoneal hyperthermic chemotherapy with mitomycin C for peritoneal carcinomatosis from nonappendiceal colorectal carcinoma. Ann Surg Oncol 2004; 11:178–186
McQuellon RP, Loggie BW, Lehman AB, et al. Long-term survivorship and quality of life after cytoreductive surgery plus intraperitoneal hyperthermic chemotherapy for peritoneal carcinomatosis. Ann Surg Oncol 2003; 10:155–162
Elias D, Delperro JR, Sideris L, et al. Treatment of peritoneal carcinomatosis from colorectal cancer: impact of complete cytoreductive surgery and difficulties in conducting randomized trials. Ann Surg Oncol 2004; 11:518–521
Glehen O, Mohamed F, Gilly FN. Peritoneal carcinomatosis from digestive tract cancer: new management by cytoreductive surgery and intraperitoneal chemohyperthermia. Lancet Oncol 2004; 5:219–228
Hagiwara A, Takahashi T, Kojima O, et al. Prophylaxis with carbon-adsorbed mitomycin against peritoneal recurrence of gastric cancer. Lancet 1992; 339:629–631
Armstrong DK, Bundy B, Wenzel L, et al. Intraperitoneal cisplatin and paclitaxel in ovarian cancer. N Engl J Med 2006; 354:34–43
Cannistra SA. Intraperitoneal chemotherapy comes of age. N Engl J Med 2006; 354:77–79
Sobin LH, Wittekind C, eds. (1997) TNM Classification of Malignant Tumours. 5th ed. New York: John Wiley & Sons.
Harmon RL, Sugarbaker PH. Prognostic indicators in peritoneal carcinomatosis from gastrointestinal cancer. Int Semin Surg Oncol 2005; 2:3
Hall JJ, Loggie BW, Shen P, et al. Cytoreductive surgery with intraperitoneal hyperthermic chemotherapy for advanced gastric cancer. J Gastrointest Surg 2004; 8:454–463
Glehen O, Schreiber V, Cotte E, et al. Cytoreductive surgery and intraperitoneal chemohyperthermia for peritoneal carcinomatosis arising from gastric cancer. Arch Surg 2004; 139:20–26
Sugarbaker PH. Intraperitoneal chemotherapy and cytoreductive surgery for the prevention and treatment of peritoneal carcinomatosis and sarcomatosis. Semin Surg Oncol 1998;14:254–261
van Ruth S, Verwaal VJ, Hart AA, van Slooten GW, Zoetmulder FA. Heat penetration in locally applied hyperthermia in the abdomen during intra-operative hyperthermic intraperitoneal chemotherapy. Anticancer Res 2003; 23:1501–1508
Sugarbaker PH, Chang D, Koslowe P. Prognostic features for peritoneal carcinomatosis in colorectal and appendiceal cancer patients when treated by cytoreductive surgery and intraperitoneal chemotherapy. Cancer Treat Res 1996; 81:89–104
Colleoni M, Litman HJ, Castiglione-Gertsch M, et al. Duration of adjuvant chemotherapy for breast cancer: a joint analysis of two randomized trials investigating three versus six courses of CMF. Br J Cancer 2002; 86:1705–1714
Smith IE, O’Brien ME, Talbot DC, et al. Duration of chemotherapy in advanced non-small-cell lung cancer: a randomized trial of three versus six courses of mitomycin, vinblastine, and cisplatin. J Clin Oncol 2001; 19:1336–1343
Markman M, Liu PY, Wilczynski S, et al. Phase III randomized trial of 12 versus 3 months of maintenance paclitaxel in patients with advanced ovarian cancer after complete response to platinum and paclitaxel-based chemotherapy: a Southwest Oncology Group and Gynecologic Oncology Group trial. J Clin Oncol 2003; 21:2460–2465
Acknowledgments
This work was supported by the Korea Science and Engineering Foundation (KOSEF) through the Cancer Metastasis Research Center (CMRC) at Yonsei University College of Medicine.
Author information
Authors and Affiliations
Corresponding author
Additional information
J.-H. Cheong and J. Y. Shen contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cheong, JH., Shen, J.Y., Song, C.S. et al. Early Postoperative Intraperitoneal Chemotherapy Following Cytoreductive Surgery in Patients with Very Advanced Gastric Cancer. Ann Surg Oncol 14, 61–68 (2007). https://doi.org/10.1245/s10434-006-9205-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-006-9205-7