Abstract
Background
Breast cancer-related lymphedema (BCRL) is a source of postoperative morbidity for breast cancer survivors. Lymphatic microsurgical preventive healing approach (LYMPHA) is a technique used to prevent BCRL at the time of axillary lymph node dissection (ALND). We report the 5-year experience of a breast surgeon trained in LYMPHA and investigate the outcomes of patients who underwent LYMPHA following ALND for treatment of cT1–4N1–3M0 breast cancer.
Methods
A retrospective review of patients with cT1–4N1–3M0 breast cancer was performed in patients who underwent ALND with and without LYMPHA. Diagnosis of BCRL was made by certified lymphedema therapists. Descriptive statistics and lymphedema surveillance data were analyzed using results of Fisher’s exact or Wilcoxon rank-sum tests. Logistic regression and propensity matching were performed to assess the reduction of BCRL occurrence following LYMPHA.
Results
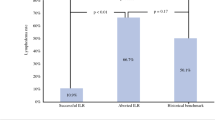
In a 5-year period, 132 patients met inclusion criteria with 76 patients undergoing LYMPHA at the time of ALND and 56 patients undergoing ALND alone. Patients who underwent LYMPHA at the time of ALND were significantly less likely to develop BCRL than those who underwent ALND alone (p = 0.045). Risk factors associated with BCRL development were increased patient age (p = 0.007), body mass index (BMI) (p = 0.003), and, in patients undergoing LYMPHA, number of positive nodes (p = 0.026).
Conclusions
LYMPHA may be successfully employed by breast surgeons trained in lymphatic–venous anastomosis at the time of ALND. While research efforts should continue to focus on prevention and surveillance of BCRL, LYMPHA remains an option to reduce BCRL and improve patient quality of life.

Similar content being viewed by others
References
DeSantis CE, Ma J, Gaudet MM, et al. Breast cancer statistics, 2019. CA Cancer J Clin. 2019;69(6):438–51.
American Cancer Society. Cancer facts & figures 2021. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2021/cancer-facts-and-figures-2021.pdf. Accessed 30 Mar 2021.
DiSipio T, Rye S, Newman B, Hayes S. Incidence of unilateral arm lymphoedema after breast cancer: a systematic review and meta-analysis. Lancet Oncol. 2013;14(6):500–15.
Fu MR. Breast cancer-related lymphedema: symptoms, diagnosis, risk reduction, and management. World J Clin Oncol. 2014;5(3):241–7.
Johnson AR, Kimball S, Epstein S, et al. Lymphedema incidence after axillary lymph node dissection: quantifying the impact of radiation and the lymphatic microsurgical preventive healing approach. Ann Plast Surg. 2019;82(4S Suppl 3):S234–41.
McDuff SGR, Mina AI, Brunelle CL, et al. Timing of lymphedema after treatment for breast cancer: when are patients most at risk? Int J Radiat Oncol Biol Phys. 2019;103(1):62–70.
Shaitelman SF, Chiang YJ, Griffin KD, et al. Radiation therapy targets and the risk of breast cancer-related lymphedema: a systematic review and network meta-analysis. Breast Cancer Res Treat. 2017;162(2):201–15.
Jammallo LS, Miller CL, Singer M, et al. Impact of body mass index and weight fluctuation on lymphedema risk in patients treated for breast cancer. Breast Cancer Res Treat. 2013;142(1):59–67.
Taghian NR, Miller CL, Jammallo LS, O’Toole J, Skolny MN. Lymphedema following breast cancer treatment and impact on quality of life: a review. Crit Rev Oncol Hematol. 2014;92(3):227–34.
Sharma A, Schwartz RA. Stewart-Treves syndrome: pathogenesis and management. J Am Acad Dermatol. 2012;67(6):1342–8.
Morgan PA, Franks PJ, Moffatt CJ. Health-related quality of life with lymphoedema: a review of the literature. Int Wound J. 2005;2(1):47–62.
Boyages J, Xu Y, Kalfa S, et al. Financial cost of lymphedema borne by women with breast cancer. Psychooncology. 2017;26(6):849–55.
Rockson SG. Lymphedema after Breast Cancer Treatment. N Engl J Med. 2019;380(7):694.
Davies C, Levenhagen K, Ryans K, Perdomo M, Gilchrist L. Interventions for breast cancer-related lymphedema: clinical practice guideline from the academy of oncologic physical therapy of APTA. Phys Ther. 2020;100(7):1163–79.
Penha TR, Ijsbrandy C, Hendrix NA, et al. Microsurgical techniques for the treatment of breast cancer-related lymphedema: a systematic review. J Reconstr Microsurg. 2013;29(2):99–106.
Ngo QD, Munot S, Mackie H, et al. Vascularized lymph node transfer for patients with breast cancer-related lymphedema can potentially reduce the burden of ongoing conservative management. Lymphat Res Biol. 2020;18(4):357–64.
Jatoi I, Benson JR, Toi M. De-escalation of axillary surgery in early breast cancer. Lancet Oncol. 2016;17(10):e430–41.
McLaughlin SA, Staley AC, Vicini F, et al. Considerations for clinicians in the diagnosis, prevention, and treatment of breast cancer-related lymphedema: recommendations from a multidisciplinary expert ASBrS panel: part 1: definitions, assessments, education, and future directions. Ann Surg Oncol. 2017;24(10):2818–26.
Boccardo F, Casabona F, De Cian F, et al. Lymphedema microsurgical preventive healing approach: a new technique for primary prevention of arm lymphedema after mastectomy. Ann Surg Oncol. 2009;16(3):703–8.
Boccardo FM, Casabona F, Friedman D, et al. Surgical prevention of arm lymphedema after breast cancer treatment. Ann Surg Oncol. 2011;18(9):2500–5.
Boccardo F, Casabona F, De Cian F, et al. Lymphatic microsurgical preventing healing approach (LYMPHA) for primary surgical prevention of breast cancer-related lymphedema: over 4 years follow-up. Microsurgery. 2014;34(6):421–4.
Feldman S, Bansil H, Ascherman J, et al. Single institution experience with lymphatic microsurgical preventive healing approach (LYMPHA) for the primary prevention of lymphedema. Ann Surg Oncol. 2015;22(10):3296–301.
Hahamoff M, Gupta N, Munoz D, et al. A lymphedema surveillance program for breast cancer patients reveals the promise of surgical prevention. J Surg Res. 2019;244:604–11.
Schwarz GS, Grobmyer SR, Djohan RS, et al. Axillary reverse mapping and lymphaticovenous bypass: lymphedema prevention through enhanced lymphatic visualization and restoration of flow. J Surg Oncol. 2019;120(2):160–7.
Spiguel L, Shaw C, Katz A, et al. Fluorescein isothiocyanate: a novel application for lymphatic surgery. Ann Plast Surg. 2017;78(6S Suppl 5):S296–8.
Koelmeyer LA, Borotkanics RJ, Alcorso J, et al. Early surveillance is associated with less incidence and severity of breast cancer-related lymphedema compared with a traditional referral model of care. Cancer. 2019;125(6):854–62.
Stout NL, Pfalzer LA, Springer B, et al. Breast cancer-related lymphedema: comparing direct costs of a prospective surveillance model and a traditional model of care. Phys Ther. 2012;92(1):152–63.
Mehrara BJ, Greene AK. Lymphedema and obesity: is there a link? Plast Reconstr Surg. 2014;134(1):154e–60e.
Chaput G, Ibrahim M, Towers A. Cancer-related lymphedema: clinical pearls for providers. Curr Oncol. 2020;27(6):336–40.
Halsted WSI. The results of operations for the cure of cancer of the breast performed at the Johns Hopkins Hospital from June, 1889, to January, 1894. Ann Surg. 1894;20(5):497–555.
Ghossain A, Ghossain MA. History of mastectomy before and after Halsted. J Med Liban. 2009;57(2):65–71.
Fisher B, Jeong JH, Anderson S, Bryant J, Fisher ER, Wolmark N. Twenty-five-year follow-up of a randomized trial comparing radical mastectomy, total mastectomy, and total mastectomy followed by irradiation. N Engl J Med. 2002;347(8):567–75.
Giuliano AE, Kirgan DM, Guenther JM, Morton DL. Lymphatic mapping and sentinel lymphadenectomy for breast cancer. Ann Surg. 1994;220(3):391–8; discussion 398–401.
Krag DN, Anderson SJ, Julian TB, et al. Sentinel-lymph-node resection compared with conventional axillary-lymph-node dissection in clinically node-negative patients with breast cancer: overall survival findings from the NSABP B-32 randomised phase 3 trial. Lancet Oncol. 2010;11(10):927–33.
Veronesi U, Viale G, Paganelli G, et al. Sentinel lymph node biopsy in breast cancer: ten-year results of a randomized controlled study. Ann Surg. 2010;251(4):595–600.
McLaughlin SA, Wright MJ, Morris KT, et al. Prevalence of lymphedema in women with breast cancer 5 years after sentinel lymph node biopsy or axillary dissection: objective measurements. J Clin Oncol. 2008;26(32):5213–9.
Giuliano AE, Ballman KV, McCall L, et al. Effect of axillary dissection vs no axillary dissection on 10-year overall survival among women with invasive breast cancer and sentinel node metastasis: the ACOSOG Z0011 (Alliance) randomized clinical trial. JAMA. 2017;318(10):918–26.
Donker M, van Tienhoven G, Straver ME, et al. Radiotherapy or surgery of the axilla after a positive sentinel node in breast cancer (EORTC 10981–22023 AMAROS): a randomised, multicentre, open-label, phase 3 non-inferiority trial. Lancet Oncol. 2014;15(12):1303–10.
Galimberti V, Cole BF, Zurrida S, et al. Axillary dissection versus no axillary dissection in patients with sentinel-node micrometastases (IBCSG 23–01): a phase 3 randomised controlled trial. Lancet Oncol. 2013;14(4):297–305.
Solá M, Alberro JA, Fraile M, et al. Complete axillary lymph node dissection versus clinical follow-up in breast cancer patients with sentinel node micrometastasis: final results from the multicenter clinical trial AATRM 048/13/2000. Ann Surg Oncol. 2013;20(1):120–7.
Sávolt Á, Péley G, Polgár C, et al. Eight-year follow up result of the OTOASOR trial: the optimal treatment of the axilla - surgery or radiotherapy after positive sentinel lymph node biopsy in early-stage breast cancer: a randomized, single centre, phase III, non-inferiority trial. Eur J Surg Oncol. 2017;43(4):672–9.
Boughey JC, Suman VJ, Mittendorf EA, et al. Sentinel lymph node surgery after neoadjuvant chemotherapy in patients with node-positive breast cancer: the ACOSOG Z1071 (Alliance) clinical trial. JAMA. 2013;310(14):1455–61.
Kuehn T, Bauerfeind I, Fehm T, et al. Sentinel-lymph-node biopsy in patients with breast cancer before and after neoadjuvant chemotherapy (SENTINA): a prospective, multicentre cohort study. Lancet Oncol. 2013;14(7):609–18.
Boileau JF, Poirier B, Basik M, et al. Sentinel node biopsy after neoadjuvant chemotherapy in biopsy-proven node-positive breast cancer: the SN FNAC study. J Clin Oncol. 2015;33(3):258–64.
Comparison of axillary lymph node dissection with axillary radiation for patients with node-positive breast cancer treated with chemotherapy. Alliance A11202 Trial, NCT01872975. https://clinicaltrials.gov/ct2/show/NCT01901094. Accessed 10 April 2021.
Moo TA, Morrow M. ASO author reflections: low-volume sentinel node disease after neoadjuvant chemotherapy is still an indication for axillary dissection. Ann Surg Oncol. 2018;25(Suppl 3):685–6.
Thompson M, Korourian S, Henry-Tillman R, et al. Axillary reverse mapping (ARM): a new concept to identify and enhance lymphatic preservation. Ann Surg Oncol. 2007;14(6):1890–5.
Suami H, Scaglioni MF. Anatomy of the lymphatic system and the lymphosome concept with reference to lymphedema. Semin Plast Surg. 2018;32(1):5–11.
Ochoa D, Korourian S, Boneti C, Adkins L, Badgwell B, Klimberg VS. Axillary reverse mapping: five-year experience. Surgery. 2014;156(5):1261–8.
Han C, Yang B, Zuo WS, Zheng G, Yang L, Zheng MZ. The feasibility and oncological safety of axillary reverse mapping in patients with breast cancer: a systematic review and meta-analysis of prospective studies. PLoS ONE. 2016;11(2):e0150285.
Axillary reverse mapping in preventing lymphedema in patients with breast cancer undergoing axillary lymph node dissection. Alliance 221702 trial, NCT 03927027. https://clinicaltrials.gov/ct2/show/NCT03927027. Accessed 10 April 2021.
Acknowledgment
Research is supported by the University of Florida Ann and Ira Horowitz Fund for Breast Cancer Research. Dr. Herremans and Dr. Riner are supported by the National Human Genome Research Institute T32 HG008958. Melany Ailin Vidret assisted in data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
The authors have no disclosures.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Herremans, K.M., Cribbin, M.P., Riner, A.N. et al. Five-Year Breast Surgeon Experience in LYMPHA at Time of ALND for Treatment of Clinical T1–4N1–3M0 Breast Cancer. Ann Surg Oncol 28, 5775–5787 (2021). https://doi.org/10.1245/s10434-021-10551-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-021-10551-8