-
PDF
- Split View
-
Views
-
Cite
Cite
Shannon L. Okada, Jeff L. Ellsworth, Diane M. Durnam, Harald S. Haugen, James L. Holloway, Merideth L. Kelley, Katherine E. Lewis, Hongping Ren, Paul O. Sheppard, Harold M. Storey, Kimberly S. Waggie, Anitra C. Wolf, Lena Y. Yao, Philippa J. Webster, A Glycoprotein Hormone Expressed in Corticotrophs Exhibits Unique Binding Properties on Thyroid-Stimulating Hormone Receptor, Molecular Endocrinology, Volume 20, Issue 2, 1 February 2006, Pages 414–425, https://doi.org/10.1210/me.2005-0270
Close - Share Icon Share
Abstract
Corticotroph-derived glycoprotein hormone (CGH), also referred to as thyrostimulin, is a noncovalent heterodimer of glycoprotein hormone α 2 (GPHA2) and glycoprotein hormone β 5 (GPHB5). Here, we demonstrate that both subunits of CGH are expressed in the corticotroph cells of the human anterior pituitary, as well as in skin, retina, and testis. CGH activates the TSH receptor (TSHR); 125I-CGH binding to cells expressing TSHR is saturable, specific, and of high affinity. In competition studies, unlabeled CGH is a potent competitor for 125I-TSH binding, whereas unlabeled TSH does not compete for 125I-CGH binding. Binding and competition analyses are consistent with the presence of two binding sites on the TSHR transfected baby hamster kidney cells, one that can interact with either TSH or CGH, and another that binds CGH alone. Transgenic overexpression of GPHB5 in mice produces elevations in serum T4 levels, reductions in body weight, and proptosis. However, neither transgenic overexpression of GPHA2 nor deletion of GPHB5 produces an overt phenotype in mice. In vivo administration of CGH to mice produces a dose-dependent hyperthyroid phenotype including elevation of T4 and hypertrophy of cells within the inner adrenal cortex. However, the distinctive expression patterns and binding characteristics of CGH suggest that it has endogenous biological roles that are discrete from those of TSH.
CORTICOTROPH-DERIVED glycoprotein hormone (CGH) (1), also referred to as thyrostimulin (2), is the newest member of the glycoprotein hormone family, which includes TSH, FSH, LH, and chorionic gonadotropin (CG). FSH, LH, and TSH are produced in the anterior pituitary and are known to affect many aspects of thyroid and gonad growth and function, whereas production of CG in the placenta is important during pregnancy (3–5). The four previously identified hormones are heavily glycosylated heterodimers consisting of a common α subunit and a unique β subunit that determines hormone specificity (6). Elucidation of the crystal structures of FSH (7) and CG (8) has revealed that both the α and the β subunits are members of a larger family of cysteine-knot-containing proteins (reviewed in Ref.9). Glycoprotein hormones bind and activate a distinct set of G protein-coupled receptors: the TSH receptor (TSHR), the FSH receptor, and the LH/CG receptor (10).
Like the other glycoprotein hormone family members, CGH is a noncovalent heterodimer with a unique β subunit, GPHB5. Unlike the other family members, which share a common α subunit, CGH uses a novel α subunit, GPHA2. We discovered the subunits of CGH during the course of a bioinformatic analysis of cysteine-knot-containing proteins in human cDNA and genomic sequences. CGH was identified independently by Nakabayashi et al. (2). Stable association of the CGH subunits was confirmed by their coimmunoprecipitation with antibodies directed to either subunit (Okada, S. L., and P. J. Webster, unpublished observations; and Ref.2).
TSH, FSH, and LH are synthesized and stored in discrete subsets of cells in the anterior pituitary and released in response to specific releasing hormones secreted by the hypothalamus (4). Both FSH and LH are made in gonadotrophs and released in response to GnRH, whereas TSH is made in thyrotrophs and released in response to TRH. Upon its release, TSH binds TSHR in the thyroid, activating second messenger systems and leading predominantly to increased levels of cAMP. These, in turn, modulate gene expression to regulate the synthesis and secretion of thyroid hormone, which plays a critical role in the metabolism and physiological function of virtually all tissues. In a classical endocrine-negative feedback loop, excess thyroid hormone inhibits the release of TRH by the hypothalamus (11).
In the present study, we have localized CGH mRNA to a variety of tissues including corticotrophs, the cells in the anterior pituitary that produce and secrete ACTH in response to CRH. Although CGH is produced in a different subset of cells than TSH, we show that recombinant human (rh) CGH binds and activates the TSHR and is a more potent competitor for the TSHR than TSH itself. Analysis of rhCGH binding to TSHR expressing cells, moreover, suggests the presence of two separate binding domains, one that is recognized by and binds CGH but not TSH, and another that is recognized by and binds both hormones. The thyroid-stimulating properties of CGH were confirmed by the elevations of T4 observed after a single ip dose of rhCGH in mice.
RESULTS
Expression of CGH in Human Tissues
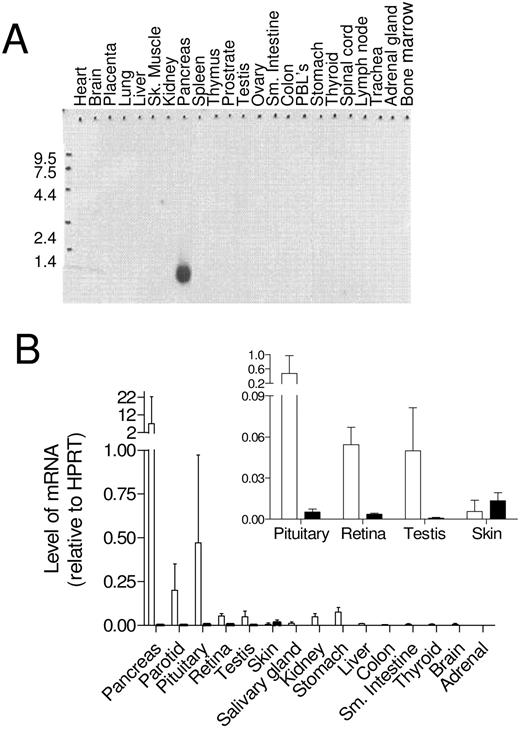
We performed a variety of analyses to identify the tissues and cell types where GPHA2 and GPHB5 were produced. By Northern analysis, GPHA2 mRNA was abundant in pancreas (Fig. 1A). Using quantitative real-time RT-PCR, we found the most abundant expression of GPHA2 mRNA in human pancreas and lower levels of expression in pituitary, parotid, retina, skin, testis, stomach, kidney, liver, colon, small intestine, and thyroid. GPHA2 mRNA was not detected in brain or the adrenal gland. By real-time RT-PCR, GPHB5 message was found in pituitary, retina, testis, and skin, but not in pancreas, parotid, kidney, stomach, liver, colon, small intestine, thyroid, brain, or adrenal gland. (Fig. 1B, inset). Similar results were obtained using real-time RT-PCR on RNA isolated from murine tissues using primers and probes specific for murine GPHA2 and GPHB5 (data not shown). Relative to expression of GPHA2, the levels of GPHB5 transcript were low in tissues that expressed both genes.
Expression of GPHA2 and GPHB5 mRNA by Northern and Quantitative Real-Time RT-PCR Analysis A, Northern analysis of GPHA2 in a selection of mRNAs from human tissues. B, Real-time RT-PCR analysis of human tissues. GPHA2 (open bars) and GPHB5 (solid bars) mRNA levels are expressed relative to that of human hypoxanthine guanine physphoribosyl transferase (HPRT). Each bar represents the mean ± sd of n = 3 measurements. The number of tissues from separate individuals analyzed is described in Materials and Methods. Inset, Real-time RT-PCR mRNA measurements from selected tissues shown on an expanded scale.
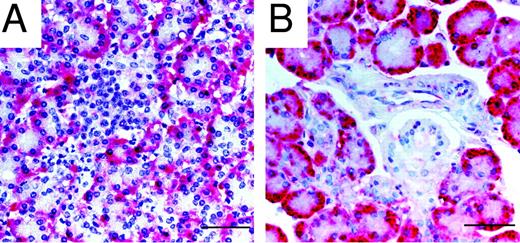
By in situ hybridization, expression of GPHA2 was detected within the acinar cells but not the islets of the human pancreas (Fig. 2A) and the acinar cells of the salivary glands (Fig. 2B). Expression of GPHB5 was not evaluated by in situ hybridization in pancreas because no GPHB5 mRNA was detected within this tissue by quantitative RT-PCR analysis (Fig. 1B).
Localization of GPHA2 mRNA in Human Pancreas and Salivary Glands by in Situ Hybridization A, Human pancreatic tissue; positive staining (red) is seen within acinar cells. The islet cells are negative. B, Human salivary gland; positive staining (red) is observed within acinar cells. ×40,. Scale bar, 50 μm.
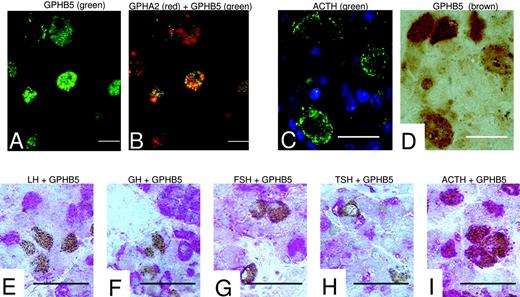
To evaluate expression of CGH in the anterior pituitary, we used double in situ hybridization and double immunohistochemistry to identify the specific cell types in the anterior pituitary that express GPHA2 and GPHB5. As is shown in Fig. 3, A and B, GPHA2 and GPHB5 mRNAs are coexpressed in the same subset of cells in the anterior pituitary. The identity of the cells expressing GPHA2 and GPHB5 mRNA was evaluated using double immunohistochemical methods to stain for the localization of GPHB5 protein relative to protein markers for different cell populations in the anterior pituitary (Fig. 3, C–I). These markers included GH (a marker for somatotrophs), FSH (gonadotrophs), LH (gonadotrophs), TSH (thyrotrophs), and ACTH (corticotrophs). Scattered cells in the human anterior pituitary showed staining with a GPHA2 antibody, however, the quality of staining and background level of staining was not suitable for colocalization of GPHA2 protein with other pituitary hormone markers (data not shown). GPHB5 protein was found to colocalize with ACTH, but not with any of the other markers. These data strongly support the conclusion that CGH is produced in the pituitary by corticotrophs.
Expression of GPHA2 and GPHB5 in Human Pituitary A, Cells in human anterior pituitary are positive for GPHB5 mRNA (green staining). B, GPHA2 (red staining) and GPHB5 (green staining) mRNAs colocalize in the human anterior pituitary (resulting in yellow staining). Panels A and B are taken in the same field on the same section. C and D, Immunohistochemical colocalization of ACTH (green staining, C) and GPHB5 (brown staining, D) in corticotrophs in the human anterior pituitary. Panels C and D are taken in the same field on the same section. E–I, Immunohistochemical localization of GPHB5 (brown staining) compared with a variety of cell-specific markers (red staining) in the human anterior pituitary. E, GPHB5 and LH (a marker for gonadotrophs) do not colocalize. F, GPHB5 and GH (a marker for somatotrophs) do not colocalize. G, GPHB5 and FSH (a marker for gonadotrophs) do not colocalize. H, GPHB5 and TSH (a marker for thyrotrophs) do not colocalize. I, GPHB5 and ACTH (a marker for corticotrophs) colocalize in corticotrophs. ×100, Scale bar, 17 μm for panels A and B. ×1000, Scale bar, 20 μm for panels C and D. ×400, Scale bar, 50 μm for panels E–I.
Expression and Purification of Recombinant Human CGH
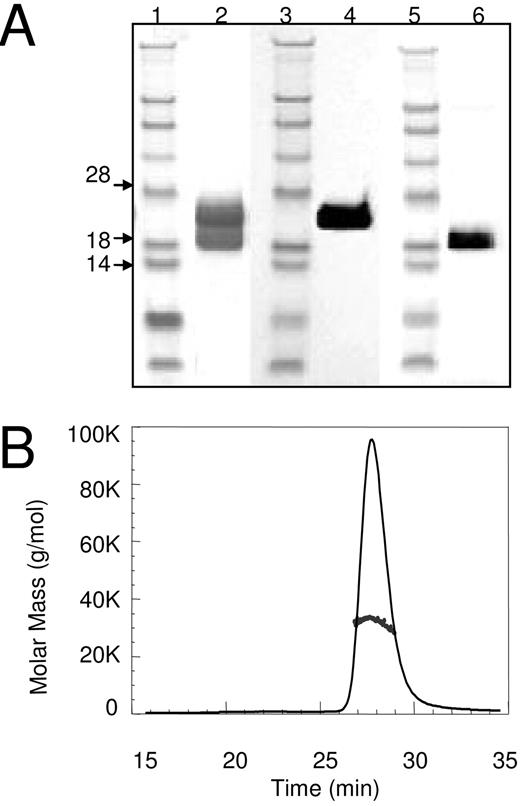
We prepared rhCGH from the conditioned media of Chinese hamster ovary (CHO) cells transfected with expression constructs for GPHA2 and GPHB5. CGH was secreted primarily as noncovalent heterodimer at a concentration of approximately 20 mg/liter. Reducing SDS-PAGE and Western blotting analyses of the purified protein confirmed the presence of both GPHA2 and GPHB5 subunits (Fig. 4A). Because nonreducing SDS-PAGE dissociated the heterodimer, CGH structure was also analyzed by size-exclusion chromatography-multiangle light scattering to give us an accurate determination of its size (Fig. 4B). Ultraviolet Radiation/Light Scattering/Refractive Index (UV/LS/RI) analysis detected one peak with a monodisperse molecular mass distribution at 34 kDa (±5%), with 24 kDa attributable to the core protein and 10 kDa to glycan (∼29%).
Analysis of Recombinant Human CGH by SDS-PAGE and Multiangle Light Scattering. A, SDS-PAGE, Coomassie Blue-stained gel (lanes 1 and 2) illustrates subunit structure of rhCGH (lane 2) by Coomassie, anti-GPHA2 immunoreactivity (lane 4) and anti-GPHB5 immunoreactivity (lane 6). Lanes 1, 3, and 5 are Invitrogen SeeBlue MW Markers. B, SEC-MALS Mass Distribution Plot (size-exclusion chromatography-multiangle light scattering) of CGH demonstrates the expected mass of 34 kDa, with 24 kDa attributable to the core protein and 10 kDa to glycan (∼29%).
Activation of Cells Expressing the TSHR by CGH
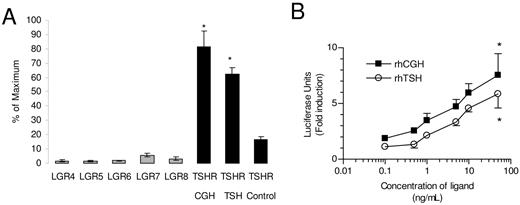
To identify a receptor for CGH, we measured the ability of rhCGH to activate a cAMP response element (CRE)-luciferase reporter construct in the TM3, TM4, and NCI-H295R cell lines. These cells express endogenous LH/CG receptors (12), FSH receptors (12), and TSHRs (13, 14), respectively. Only NCI-H295R, an adrenal cortex carcinoma cell line, showed an increase in luciferase activity in response to CGH treatment (data not shown), suggesting that CGH did not activate the LH/CG or FSH receptors, but might activate TSHR. To evaluate this hypothesis, baby hamster kidney (BHK) cells were stably transfected with the human TSHR and a CRE-luciferase reporter construct (BHK-hTSHR cells). Incubation of BHK-hTSHR cells with 100 ng/ml of either rhCGH- or rhTSH-induced luciferase activity (Fig. 5A). To compare the activity of rhTSH and rhCGH, BHK-TSHR cells were treated with each hormone over a range of concentrations. rhCGH was approximately 4-fold more potent than rhTSH in stimulating cAMP-driven luciferase activity in these cells (Fig. 5B). The GPHB5 subunit alone also activated the BHK-hTSHR, but at a concentration 100-fold higher than the CGH heterodimers (data not shown). Under identical conditions, the GPHA2 subunit alone did not induce luciferase activity in the BHK-hTSHR cells (data not shown). To evaluate whether CGH could bind and activate other orphan members of the glycoprotein hormone receptor family, BHK cells were stably transfected with one of each of five glycoprotein hormone receptors in the leucine-rich G protein-coupled receptor (LGR) family (15, 16). CGH did not activate any of the LGR receptors tested (LGR4–8; Fig. 5A).
CGH Is a Ligand for the TSHR A, BHK cells containing the TSHR are represented by the black bars. rhCGH and rhTSH activate luciferase activity in BHK cells transfected with the TSHR. rhCGH does not activate BHK cells transfected with the LGRs 4–8 (gray bars). Each bar represents the mean ± sd of five to six determinations. Differences were significant (*, P = 0.0086) relative to treatment with vehicle alone. B, BHK cells containing the TSHR were treated with the indicated concentration of either rhTSH or rhCGH. Each data point represents the mean ± sd of six determinations. Differences were significant (*, P < 0.0001) relative to treatment with vehicle alone.
Binding of 125I-CGH and 125I-TSH Human TSHR
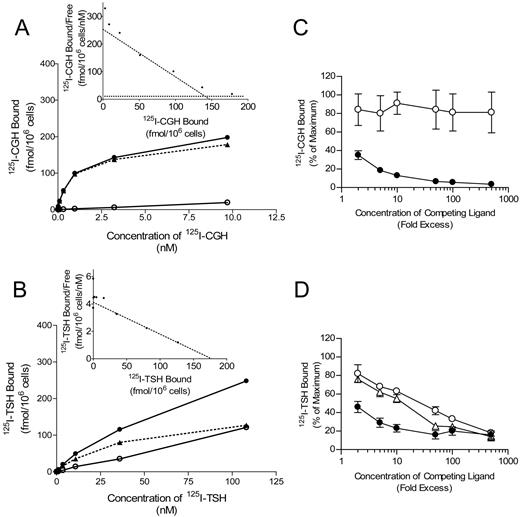
Both 125I-CGH and 125I-TSH bound to BHK-TSHR cells in a time-dependent manner and reached an apparent steady state within 2 h at 4 C (data not shown). Binding of both radiolabeled ligands was saturable, specific, and of high affinity (Fig. 6, A and B). Scatchard plots of 125I-CGH binding were concave upward, indicating either that 125I-CGH bound to at least two sites in these cells or that binding exhibited negative cooperativity (Fig. 6A, inset). Hill plots of the 125I-CGH-specific binding data were linear with a Hill slope of 1.08; however, a finding consistent with multiple binding sites for 125I-CGH under these conditions. The 125I-CGH-specific binding data were directly fitted to a two-site model by nonlinear regression to yield the equilibrium dissociation constant (Kd) and the maximum amount bound (Bmax) to each of the sites at steady state. The Kd and Bmax for the high-affinity site were 1.19 ± 0.68 nm and 139 ± 74 fmol/106 cells or 86,000 high-affinity sites per cell, respectively. The low-affinity site exhibited a Kd and Bmax of 2.7 ± 2.2 mm and 1.6 ± 1.4 x106 fmol/106 cells, respectively. Scatchard plots for 125I-TSH-specific binding were linear, suggesting the presence of a single binding site (Fig. 6B, inset). Using a single site model of nonlinear regression, the Kd and Bmax for 125I-TSH binding to BHK-TSHR cells were estimated at 41 nm and 178 fmol/106 cells or about 107,000 sites per cell. Little or no binding of 125I-TSH or 125I-CGH was observed, in contrast, to parental BHK cells lacking the TSHR (data not shown). Addition of increasing amounts of unlabeled rhCGH reduced the amount of 125I-CGH bound to BHK-TSHR cells (Fig. 6C). In contrast, over an identical concentration range, the addition of neither unlabeled rhTSH (Fig. 6C) nor unlabeled rhGPHA2 nor rhGPHB5 (data not shown) reduced the amount of 125I-CGH bound. However, rhCGH was about 10–20 times more potent as a competitor than either bovine or human TSH in reducing the binding of 125I-TSH to BHK-TSHR cells (Fig. 6D).
Binding of 125I-Labeled Human CGH and TSH to BHK Cells Expressing the Human TSHR Saturation binding studies (A and B) were carried out as described in Materials and Methods. Cells were incubated with the indicated concentration of 125I-CGH (A) or 125I-TSH (B) in the absence (closed circles, total binding) or presence (open circles, nonspecific binding) of 25 μg/ml of unlabeled rhCGH (A) or 10 μg/ml unlabeled rhTSH (B). Specific binding (closed triangles) was calculated as described in Materials and Methods. Scatchard plots of 125I-CGH and 125I-TSH-specific binding are shown in the insets to panels A and B, respectively. The dotted lines correspond to the nonlinear regression fits. Competition studies (C and D) were performed as described described in Materials and Methods. Cells were incubated with 0.1 nm125I-CGH (C) or 1.0 nm125I-TSH (D) in the absence or presence of increasing amounts of unlabeled rhCGH (closed circles), rhTSH (open circles), or bovine TSH (open triangles).
Overexpression of GPHA2 and GPHB5 in Transgenic Mice
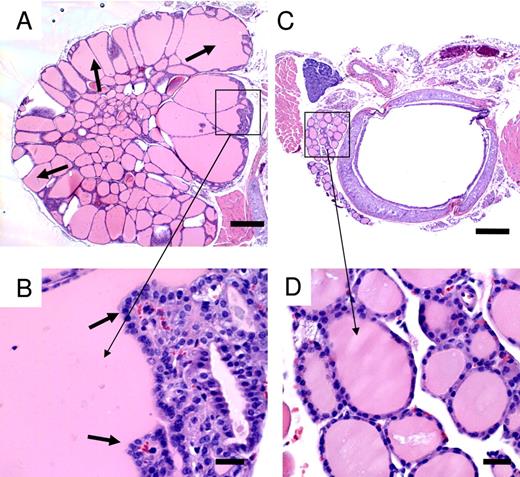
We established transgenic mice overexpressing either GPHA2 or GPHB5 under control of the metallo-thionein-I promoter. Expression analysis on liver biopsies confirmed high levels of the transgenic transcripts. Although the GPHA2 overexpressing mice showed no overt phenotype, overexpression of GPHB5 had profound effects on thyroid function and morphology. After 6–8 wk of age, many of these animals developed proptosis, or bulging eyes, a characteristic of Grave’s disease in humans (data not shown). The animals had serum T4 levels that were elevated 2- to 3-fold above littermate controls and had significantly reduced body weights. Male GPHB5 transgenic mice at 6 wk of age had an average body weight of 23.3 ± 2.8 g compared with wild-type animals with an average body weight of 29.6 ± 4.1 g (P < 0.008). Female GPHB5 transgenic mice at 6 wk of age had an average body weight of 17.8 g ± 2.8 g compared with wild-type animals with an average body weight of 23.7 ± 1.9 g (P < 0.001). Histological analysis of the thyroid revealed hypertrophy of the thyroid follicles (Fig. 7A) and hyperplasia of the thyroid follicular epithelial cells (Fig. 7B) of the transgenic mice but not in littermate controls (Fig. 7, C and D). Hypertrophy of cells within the inner adrenal cortex was also observed only in the transgenic animals (data not shown).
Hypertrophy and Hyperplasia of Thyroid from Transgenic Overexpression of GPHB5 A, Thyroid from GPHB5 transgenic mouse. The arrows point to hypertrophied follicles. H&E, ×4. B, Magnified view of boxed area in panel A. The arrows point to hyperplasia of the thyroid follicular epithelial cells. H&E, ×40. C, Thyroid of wild-type control mouse. H&E, ×4. D, Magnified view of boxed area in A. H&E, ×40. Scale bar, 300 μm for panels A and C. Scale bar, 30 μm for panels B and D.
Our results are similar to those described in a recent report (17) where GPHB5 transgenic animals were established by inserting the GPHB5 coding sequence into the Rosa26 locus. Macdonald et al. (17) observed an approximately 2-fold elevation in serum T4 levels; however, they found no overt changes in the thyroid glands of transgenic animals.
Systemic Delivery of CGH Induced a Hyperthyroid Phenotype in Normal Mice
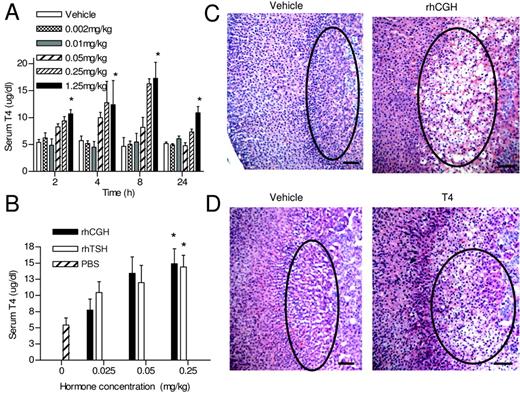
C57BL/6 female mice were given a single ip injection with rhCGH at doses ranging from 0.002 mg/kg to 1.25 mg/kg, and serum T4 levels were measured over a 24-h period. Significant increases in serum T4 levels were observed at 2 h after injection for doses of 0.05 mg/kg and higher, changes that persisted for more than 24 h at higher doses of rhCGH (Fig. 8A). Subsequently, C57BL/6 female mice were given an ip injection once daily with 0.25 mg/kg rhCGH for a period of 2 wk. Histological examination of the adrenal glands after 2 wk of treatment revealed profound hypertrophy and fine intracytoplasmic vacuolation of the cells in the inner adrenal cortex of rhCGH-treated animals but not in vehicle-treated controls (Fig. 8C). The adrenal gland weights normalized to body weights were also significantly increased compared with vehicle-treated animals. Mice treated with 0.25 mg/kg of rhCGH had an average adrenal weight per 100 g of body weight of 22.26 mg ± 0.99 mg compared with vehicle-treated animals with a value of 15.32 mg ± 2.21 mg (P < 0.0015). The cytoplasm of the hypertrophied cells in the inner adrenal cortex stained positive with Oil red O confirming the cells were filled with neutral fat (data not shown.) Because changes in the inner adrenal cortex were previously documented for hyperthyroid mice (18, 19), we also compared chronic rhCGH treatment with exogenous T4 treatment. T4 treatment at a dose of 0.125 mg/kg produced similar changes in the adrenal cortex as were observed with rhCGH administration (Fig. 8D).
Administration of rhCGH Produces a Hyperthyroid Phenotype A, Mice received a single ip injection of the indicated amount of rhCGH and serum levels of T4 were analyzed as described in Materials and Methods. Each bar represents the mean values ± sd for n = 4 mice. Differences were significant (*, P < 0.0001) relative to mice treated with vehicle alone. B, Mice received a single ip injection of the indicated amounts of vehicle alone, rhCGH, or rhTSH and the serum T4 levels were measured 4 h later. Each bar represents the mean ± sd for n = 8–12 mice per group. Differences were significant (*, P < 0.0001) relative to mice treated with vehicle alone. C, H&E-stained section of the adrenal glands of mice that received a single ip injection of vehicle (left panel) or 0.25 mg/kg rhCGH (right panel). The areas of comparison are circled for clarification, ×10. D, H&E-stained section of the adrenal glands from mice that received a single ip injection of vehicle alone (left panel) or 0.125 mg/kg T4 (right panel), ×10. Scale bar, 100 μm for panels C and D.
Comparison of T4 Induction by Systemic Delivery of rhCGH or rhTSH in Normal Mice
To compare in vivo activity of TSH and CGH, serum T4 levels were measured 4 h after female C57BL/6 mice were injected ip with either rhCGH, rhTSH, or PBS. Significant increases (P > 0.001) in serum T4 levels were observed in mice treated with both rhCGH and rhTSH over the entire dose range tested compared with PBS-injected mice (Fig. 8B). Overall, there was little difference in T4 levels after single injections of rhCGH compared with rhTSH over the dose range administered except at the lowest dose tested. With a dose of 0.025 mg/kg, rhTSH produced a modest, but statistically significant, increase in T4 levels above that observed after injection of the same dose of rhCGH.
Deletion of GPHB5 Produced No Overt Phenotype in Mice
We established GPHB5 knockout mice and subjected them to a wide variety of physiological assays. In brief, this analysis included body weight, clinical blood chemistry, and general observations of live animals at various ages between 4 and 59 wk of age. Animals were killed at various ages, and the animals were x-rayed, body and tissue weights were collected, and any gross abnormalities were noted. Thirty-three tissues from major organ systems were collected and microscopically evaluated by routine methods. No overt differences were found in the GPHB5 homozygous knockouts relative to heterozygous littermates or wild-type controls.
DISCUSSION
CGH, the fifth member of the glycoprotein hormone family, is similar in both sequence and structure to other family members. It is a noncovalent heterodimer composed of GPHA2 and GPHB5, novel α and β subunits. In its activity it appears to be related to TSH, serving as an alternate ligand for the TSHR. However, here we report that CGH is expressed in corticotrophs, a site of production in the anterior pituitary that is distinct from that of TSH, which is expressed in thyrotrophs. The difference in localization suggests that the two hormones are also secreted under different control mechanisms. In addition, we find that CGH has binding characteristics for TSHR that are also different from those of TSH. Both the distinct localization and binding properties suggest that the biological role of CGH is discrete from that of TSH.
It is intriguing that endogenous CGH is expressed in corticotrophs, cells that were not previously believed to have a direct role in thyroid activation. In a system known as the hypothalamus-pituitary-adrenal (HPA) axis, corticotrophs produce and store ACTH, which is secreted under conditions of stress in response to the release of CRH by the hypothalamus. ACTH in turn stimulates the adrenal cortex to secrete cortisol. If CGH is stored in the same secretion granules as ACTH, it may be coreleased as a component of stress-response through the HPA axis. CGH could affect adrenal function directly, via TSHR expressed in adrenal tissue, or indirectly through the thyroid, via elevations in thyroid hormone. Alternatively, the secretion of CGH from corticotrophs may not be in response to stress and/or may not serve to modulate adrenal activity. Future experiments will be critical to determine whether or not CGH is released along with ACTH in response to stimulation by CRH. Elucidation of the mechanism controlling the release of CGH from corticotrophs may provide some insight into a physiological role of this hormone.
Nakabayashi and colleagues (2) also reported colocalization of GPHA2 and GPHB5 to the same cells in the anterior pituitary; they did not, however, observe colocalization of GPHA2 with any of the markers for cell types in the anterior pituitary, including GH, PRL, LH-β, TSH-β, and ACTH. The reason for this discrepancy is unclear but may be related to antibody specificity as anti-GPHA2 antibodies were used in the previous study, whereas anti-GPHB5 antibodies were used in the present report. Alternatively, species differences (rat vs. human tissues) may also account for these differences.
We report both subunits of CGH are also found in tissues outside of the HPA and hypothalamus-pituitary-thyroid (HPT) axes, including skin, retina, and testis. Our results are similar to those described by Nakabayashi et al. (2) where by endpoint RT-PCR analysis of a panel of rat tissue mRNAs, they also found GPHA2 and GPHB5 transcripts in diverse tissues outside of the HPA and HPT axes. However, in the previous study, GPHB5 expression was observed in the brain, thyroid, and the adrenal gland, but not in the testis. The difference in expression pattern for GPHB5 between the previous report and the current report is unclear but could be explained by the different sensitivities of endpoint RT-PCR vs. real-time RT-PCR. Our results are also similar to those described in another report where GPHA2 and GPHB5 were colocalized in the rat pituitary, central nervous system, adrenal gland, stomach, duodenum, pancreas, and testis using immunohistochemistry (20).
Expression of TSH and TSHR in a variety of tissues outside of the HPA and HPT axes is also well documented (21). The wide distribution of CGH, TSH, and TSHR suggests that they have additional roles outside of adrenal or thyroid activation. For example, TSH may be an important mediator of immune function in the intestine (22, 23). When small intestinal epithelial cells are stimulated with TRH, they secrete TSH, which can activate TSHR expressed in intestinal T cells. In addition, TSH immunocytochemical staining was increased after acute enteric virus infection in mice suggesting that TSH pays a role in the process of local immunity during viral infection (24). This localized function for TSH is entirely separate from the normal regulation of thyroid activity. The distribution of CGH expression supports the idea that CGH also has distinct, localized activities.
CGH or TSH may have localized activity within the pituitary. TSHR expression has been reported in the folliculo-stellate (FS) cells in the anterior pituitary (25, 26). FS cells have long cytoplasmic processes that are interdigitated between the endocrine cells, including corticotrophs and thyrotrophs, in the anterior pituitary (27). FS cells produce cytokines and growth factors and are involved in endocrine function and regulation. The proximity of TSH- and CGH-producing cells and TSHR-expressing FS cells in the anterior pituitary suggests a paracrine interaction that has yet to be evaluated. Again, the difference between the cell types in the pituitary expressing CGH and TSH is consistent with the idea that the two hormones function under distinct circumstances. Interestingly, GPHA2 is also highly expressed in exocrine pancreas and salivary gland where GPHB5 is not detected, suggesting that this protein may have additional independent functions outside of its role in CGH. Similarly, autonomous functions have also been suggested for the common glycoprotein hormone α subunit (28, 29).
The biological actions of CGH appear to be mediated by binding and activation of the TSHR. The unexpected finding that TSH fails to compete for 125I-CGH binding suggests that the two hormones bind distinct sites in the TSHR-transfected BHK cells used in the present study. It is unlikely that this result is due to differences in ligand affinities for the TSHR, as a 500-fold excess of TSH fails to reduce the amount of 125I-CGH bound. On the other hand, the potent competition by CGH for 125I-TSH binding suggests that a portion of the CGH binding sites overlap with those of TSH. Studies of TSH and TSHR mutations in the human population coupled with site-directed mutagenesis of both TSH and its receptor have revealed a complex set of interactions that regulate ligand binding to the TSHR (30). Similar to other GPCRs, the TSHR has been shown to dimerize and may exist as higher order oligomers within the plasma membrane (30, 31). Further complexity is evident from observations that TSHRs can undergo posttranslational processing, producing receptors with variable ligand binding (31). Thus, the present results could be explained by CGH binding to a specific oligomeric form or open confirmation (30, 31) of the TSHR. In support of this is the similarity in receptor numbers measured with both hormones in the TSHR-BHK cells. Whether these results using the TSHR-BHK cells can be extrapolated to endogenous TSHR in target cells is not clear at this present time. Whatever the nature of this binding site, the present data demonstrate the presence of a discrete site on TSHR-BHK cells that binds CGH but not TSH, as well as a second domain that can bind either hormone.
Surprisingly, transgenic animals overexpressing GPHB5 alone show thyroid activation, an effect also recently reported by Macdonald et al. (17). This result may be explained by our in vitro data demonstrating that, at high concentrations, GPHB5 alone can stimulate the TSHR. However, neither GPHA2 nor GPHB5 competes for binding of 125I-CGH to BHK-TSHR cells. Alternatively, because the metallothionein-1 promoter drives expression in a wide range of tissues, it is possible that the transgenic GPHB5 is being coexpressed with native GPHA2 to form active CGH protein.
The function of CGH in vivo is currently unknown. No overt phenotypes were observed in knockout mice lacking the GPHB5 gene, suggesting that CGH is not critical for survival under normal laboratory conditions. CGH and TSH are alternate ligands for the same receptor, and CGH administered in vivo clearly has an effect similar to TSH in stimulating thyroid metabolism. However, as the two ligands are expressed in discrete locations and have quite different binding properties for TSHR, it seems unlikely that the biological function of CGH is entirely redundant with that of TSH. Future experiments comparing the activities of TSH and CGH should help to provide a more comprehensive understanding of the similarities and differences of their physiological roles.
MATERIALS AND METHODS
Cloning of Human and Mouse GPHA2 and GPHB5
GPHA2 and GPHB5 were identified by sequence analysis looking for open reading frames encoding putative cysteine-knot-containing proteins and cloned using standard techniques. Briefly, human GPHA2 was cloned from a human pancreatic islet cell cDNA library. Human GPHB5 was cloned from a human testis cDNA library. Mouse GPHA2 was ordered from the I.M.A.G.E. Consortium (LLNL) cDNA Clones where it was cloned from a C57BL/6J mouse thymus library. Two exons from mouse GPHB5 were amplified from a positive clone from the BAC 129Sv Mouse Release II library (Genome Systems, St. Louis, MO).
Identification of Cells Expressing GPHA2 and GPHB5 by in Situ Hybridization
Human pituitaries were screened for GPHA2 and GPHB5 expression by in situ hybridization. The tissues were fixed in 10% buffered formalin and embedded in paraffin blocks using standard techniques. Tissues were sectioned at 4–8 μm, and the sections were prepared using a standard protocol. Briefly, tissue sections were deparaffinized with HistoClear (National Diagnostics, Atlanta, GA) and then rehydrated with graded ethanol. Enzyme digestion was performed with Proteinase K (50 μg/ml) (Roche, Indianapolis, IN) at 37 C for 3–10 min. Genomic DNA was removed with deoxyribonuclease (5 U/μl) pretreatment (Roche, Indianapolis, IN) at 37 C for 30 min. This step was followed by acetylation and dehydration of the tissues.
Using oligonucleotides specific for GPHB5 sequences, a PCR-based in situ method was used to visualize GPHB5 mRNA with a fluorescein isothiocyanate (FITC) detection system, which gives a green signal. After this reaction, the same slide was subjected to a standard in situ hybridization protocol using a probe designed against the human GPHA2 sequence. T7 RNA polymerase was used with a linearized plasmid template containing the entire coding domain and the 3′-untranslated region of GPHA2 to generate an antisense probe. The probe was labeled with digoxigenin (Roche, Indianapolis, IN) using an In Vitro Transcription System kit (Promega, Madison, WI) following the manufacturer’s instructions. The digoxigenin-labeled GPHA2 probe was added to the slides at a concentration of 1–5 pmol/ml for 12–16 h at 60 C. Slides were subsequently washed in 2× SSC (saline sodium citrate) and 0.1× SSC at 55 C. The signals were amplified using tyramide signal amplification (in situ indirect kit; NEN, PerkinElmer Life Sciences, Boston, MA) and visualized with Texas Red following the manufacturer’s instructions. No counterstain was performed due to lack of nucleic stain caused by deoxyribonuclease digestion as described earlier.
Double Immunohistochemical Staining of GPHB5 and Markers for Cell Types in the Human Anterior Pituitary
Human anterior pituitaries were screened using antibodies against GPHB5 and a variety of cell type-specific markers to determine which cell types express GPHB5 protein. Double immunostains were performed for GPHB5 vs. GH, FSH, LH, TSH, ACTH, PRL, and S-100 protein. Although scattered cells in the human anterior pituitary showed staining with a GPHA2 antibody, the quality of staining and background level of staining was not suitable for colocalization of GPHA2 protein with other pituitary hormone markers. Sandwich technique immunohistochemistry was used using two primary antibodies (anti-GPHB5 and antibodies against one of the marker proteins) and two detection systems: immunoperoxidase (IP) with diaminobenzidine (DAB) (Ventana Bio Tek Systems, Tucson, AZ), leading to a brown signal indicating the presence of GPHB5, and alkaline phosphatase (AP) with BioTek Red (Ventana Bio Tek Systems), leading to a red signal for the marker protein in question. A second detection system was used to confirm the costaining of GPHB5 and ACTH. This detection system included IP with DAB leading to a brown signal for the presence of GPHB5, and FITC-conjugated secondary antibody leading to a green signal indicating the presence of ACTH. Sections of a human pituitary gland taken from a 24-yr-old male were fixed in 10% buffered formalin for 24–48 h, processed, embedded in paraffin, and sectioned at 4–8 μm using standard histology techniques. The tissue sections were stained with the TechMate 500 autoimmunstainer (Ventana, Tucson, AZ) using modified IP-AP and IP-FITC protocols (Ventana). The basic staining sequence for the IP-AP protocol was as follows: 1) citrate buffer antigen retrieval (LabVision, Fremont, CA), 20 min steam; 2) avidin/biotin blocking (Zymed, South San Francisco, CA), 10 min each; 3) 5% normal goat serum blocking (Vector, Burlingame, CA), 20 min; 4) rabbit antihuman GPHB5 protein (produced in-house, internal reference number E3039), working dilution: 1:3200, 25 min; 5) biotinylated goat antirabbit IgG (Vector), working solution: 7.5 μg/ml, diluted in PBS with 2% normal goat serum, 30 min; 6) 3% HP blocking (Ventana), 7 min × 3; 7) ABC, 25 min; 8) DAB, 4 min × 3; 9) rabbit antihuman GH (Zymed), working dilution: 1:25; mouse antihuman FSH (Zymed), working dilution: 1:50; mouse antihuman LH (Zymed), working dilution: 1:50; mouse antihuman TSH (Zymed), working dilution: 1:50; rabbit antihuman ACTH (Zymed), working dilution: 1:50; rabbit anti-PRL (Zymed), working dilution: 1:50; or rabbit anti-S-100 protein (Zymed), working dilution: 1:1000 and 1:2000; 10) biotinylated goat antirabbit IgG (Vector), working solution: 7.5 μg/ml, diluted in PBS with 2% normal goat serum; or biotinylated goat antimouse IgG (Vector), working solution: 7.5 μg/ml, diluted in PBS with 2% normal goat serum and 2% nonfat dried milk (NFDM, Mid-American Farms, Kansas City, MO); 11) AP, 25 min; 12) CHROM GEN, 7 min × 3; and 13) hematoxylin nuclear counter stain, 1 min. The basic staining sequence for the IP-FITC protocol was the same as the IP-AP from steps 1–9, followed by: 10) FITC-conjugated goat antirabbit IgG (KPL, Gaithersburg, MD), working solution: 6.7 μg/ml, diluted in PBS with 2% normal goat serum; and 11) DAPI nuclear counter stain (Vectashield, Vector). Immunostaining in the tissue samples was observed using a Nikon Eclipse E600 microscope (Nikon Inc., Melville, NY) with an Y-FL EPI-Fluorescence Attachment. Images were captured using a charge-coupled device (3CCD) video camera (Dage-MTI, Michigan City, IN) mounted in the Nikon Eclipse 600 microscope.
Quantitative Real-Time PCR
Expression of GPHA2 and GPHB5 mRNA was measured with a multiplex real-time quantitative RT-PCR method (TaqMan) and the ABI PRISM 7900 sequence detection system (PE Applied Biosystems, Foster City, CA). GPHA2 and GPHB5 mRNA levels were normalized to the expression of hypoxanthine guanine physphoribosyl transferase mRNA and determined by the comparative threshold cycle method (User Bulletin 2; PE Applied Biosystems). The primer pairs and probes were designed to span an intron to avoid signal from genomic DNA. The primers and probe for human GPHA2 included forward primer 5′-TCTCAGTGCTGCACCATCAGT, reverse primer 5′-TCCGGCTCCCCACACA and probe 5′-TGCAGCTGTACTTTGACCTTCTTCAGG. The primers and probe for human GPHB5 included forward primer 5′-CCTGGGAGAAACCCATTCTG, reverse primer 5′-GTTGGGCAGCTTGACAGTCA and probe 5′-CCCTATATTGAAGCCCATCATCGAGTCTGTACCT. Two individual human normal tissue samples were analyzed for retina, salivary gland, stomach, liver, colon brain, and parotid. Three individual tissue samples were analyzed for pituitary, skin, and adrenal gland. Four individual tissue samples were analyzed for testis and kidney. Nine individual tissue samples were analyzed for pancreas and thyroid.
Expression and Purification of Recombinant Human CGH
CHO DG44 cells were transfected with expression constructs containing GPHA2, GPHB5, and the drug resistance dihydrofolate reductase under the control of the cytomegalovirus promoter. The resulting pool was selected and amplified using methotrexate. When early analysis indicated overexpression of GPHA2 and low-level GPHB5 expression, a second construct expressing GPHB5 from the cytomegalovirus promoter and Zeocin resistance from the simian virus 40 promoter was transfected into the selected, amplified pool by electroporation. After Zeocin selection, the final pool expressed significant levels of both GPHA2 and GPHB5, secreted primarily as the noncovalent heterodimer CGH (estimated at 20 mg/liter by analytical RP-HPLC.) CGH was purified from CHO culture conditioned media after adjusting the conditioned media to pH 6.0 and loading the sample at 55 cm/h onto a POROS HS50 column previously equilibrated in 20 mm 4-morpholine-ethanesulfonic acid (pH 6.0). The column was washed with equilibration buffer containing 300 mm NaCl, and the CGH was eluted with a linear gradient from 300–800 mm NaCl in 20 mm 4-morpholine-ethanesulfonic acid (pH 6.0). Samples containing CGH were combined, adjusted to 1.0 m (NH4)2SO4 and pH 6.9 with NaOH and were loaded at 100 cm/h onto a TOSOH Butyl 650S column equilibrated in 50 mm sodium phosphate, 1.0 m (NH4)2SO4 pH 6.9. The column was washed with decreasing concentrations of (NH4)2SO4 and the CGH was eluted at 200 cm/h by reducing the (NH4)2SO4 to 0.3 m and collecting 5 CV. The CGH-containing fractions were combined, concentrated by ultrafiltration, and applied to a Superdex 200 column equilibrated in 50 mm sodium phosphate, 109 mm NaCl (pH 7.2). Homogeniety of the preparation was confirmed by SDS-PAGE, N-terminal sequencing, mass spectrometry, and light scattering analyses.
Activation of BHK Cells Expressing the Human TSHR
BHK cells were cultured in DMEM (high glucose) with l-glutamine (DMEM; Invitrogen, Carlsbad, CA) supplemented with 1 mm sodium pyruvate and 5% fetal bovine serum (FBS) at 37 C in a humidified atmosphere of 95% air, 5% CO2. Transfection of the vector containing KZ55, a CRE-driven luciferase reporter cassette was performed using Lipofectamine (Invitrogen). A similar procedure was used for transfection of expression vectors containing the orphan glycoprotein hormone receptors LGR4–8 and TSHR. Two days before assaying, cells were seeded at 5,000 cells per well in a 96-well white opaque/clear bottom plate (BD Biosciences, Bedford, MA). One day before assay, cells were switched to serum-free media containing 0.5% BSA and incubated with vehicle alone, 10 μm forskolin, or the indicated concentrations of rhCGH. After 4 h at 37 C, the plates were washed with PBS and cells were processed using the Luciferase Assay System (Promega, Madison, WI) according to the manufacturers instructions. Luciferase activity was measured on a microplate luminometer (PerkinElmer Life Sciences, Inc.) after automated injection of luciferase assay substrate.
Binding of 125I-CGH and 125I-TSH to BHK Cells Expressing the TSHR
Radiolabeled rhCGH and human pituitary-derived TSH (BiosPacific, Emeryville, CA) were prepared with 125I-Bolton Hunter Reagent (Amersham Pharmacia Biotech, Buckinghamshire, UK) according to the manufacturer’s instructions. Fifty micrograms of protein were radiolabeled to specific activities of 9,000–14,000 cpm/ng with 89–97% of the radioactivity precipitating with 10% trichloroacetic acid. Bioactivity of each preparation of 125I-CGH and 125I-TSH was measured using BHK cells transfected with a CRE-luciferase reporter construct and the human TSHR. There were no significant differences in the bioactivity of 125I-CGH or 125I-TSH compared with their unlabeled counterparts. BHK cells expressing murine or human TSHRs were cultured in DMEM (high glucose) containing 5% fetal bovine serum, 1% Glutamax, 1% sodium pyruvate, 250 nm methotrexate, and Geneticin (1/100). Cells were seeded on d 0 at a density of 4000 cells/cm2 and were used on d 4–5 by which time the cell number had increased to about 1 × 105 cells/cm2. Plates of cells were placed on ice and washed twice with 1.0–2.0 ml of ice-cold PBS. The washes were discarded and each well was incubated with the indicated concentration of 125I-CGH or 125I-TSH in 0.1 ml/cm2 of Medium B [DMEM (no bicarbonate), 0.02 m HEPES (pH 7.4), 1.0 mg BSA/ml]. Specific binding was determined in the presence (nonspecific binding) and absence (total binding) of unlabeled rhCGH (25.0 μg/ml) or TSH (10 μg/ml). Binding reactions were terminated by removing the binding medium, and the cell monolayers were washed three times with 1.0–2.0 ml of ice-cold PBS containing 1.0 mg/ml BSA and twice with 1.0–2.0 ml of ice-cold albumin-free PBS. Cells were solubilized in 1.0 ml of 1.0 n NaOH for 10 min at room temperature and cell-associated radioactivity was determined in a γ counter. The measured radioactivity was normalized to cell number that was determined on plates of cells cultured in parallel. Binding data were analyzed by nonlinear regression using Prism 3.0 (GraphPad Software, San Diego, CA).
Generation of GPHB5 Transgenic Mice
The human GPHB5 open reading frame was inserted into the expression vector pTG12–8, containing the metallothionein-1 promoter. A consensus translation initiation sequence was placed upstream of the GPHB5 ATG. Introns from rat insulin II and a human GH polyadenylation signal sequence were cloned 5′ and 3′ of the GPHB5 ORF to enhance transgene expression. Fertilized ova from mating of B6C3F1Tac mice were microinjected with the transgene insert by standard procedures. Founders were identified by PCRs on genomic tail DNA, and transgenic lines were established by breeding founders with C57BL/6 mice (Taconic, Germantown, NY). Transgene expression was performed as described (32). Mice were housed in ventilated microisolator racks in a barrier facility, and the colony routinely produced negative results in tests for common murine pathogens. The ZymoGenetics Institutional Animal Care and Use Committee approved all animal procedures.
Targeted Deletion of GPHB5 in Mice
GPHB5−/− mice were generated at Lexicon Genetics Inc. (The Woodlands, TX) as described (33). A replacement-type targeting vector with 10-kb homologous arms of the GPHB5 genomic was used to delete a 179-bp genomic region at Exon 3 of the GPHB5 gene. Thus, approximately half of the GPHB5 open reading frame was deleted. The deleted region was replaced by an IRESLacZ/MC1-Neo selection cassette. The linearized targeting vector was electroporated into 129S5-derived Lex-1 ES cells. Correctly targeted ES clones were identified by Southern blotting. Chimeras were generated by injection of targeted ES cells into C57BL/6-Tyrc-Brd blastocysts. Their progeny were genotyped by PCRs detecting the wild-type and neo alleles.
Systemic Delivery of rhCGH in Normal Mice
Increasing doses of rhCGH protein, rhTSH protein (Genzyme Therapeutics, Cambridge, MA), or T4 (Sigma-Aldrich, St. Louis, MO) were injected one time or once daily for 2 wk into the ip cavity of 8- to 12-wk-old female C57BL/6 mice. rhCGH or rhTSH protein was delivered at doses ranging from 0.025–1.25 mg/kg. T4 was delivered at a dose of 0.125 mg/kg. Blood was collected by cardiac puncture (terminal only) under isoflurane anesthesia and serum total T4 levels were measured using the Vet ACE serum chemistry analyzer (Alfa Wassermann, West Caldwell, NJ). The ZymoGenetics Institutional Animal Care and Use Committee approved all animal procedures.
Histologic Analysis of Murine Tissues
Mouse tissues were fixed in 10% neutral buffered formalin and routinely processed into hematoxylin and eosin (H&E)-stained sections. A subset of adrenal glands was frozen in OCT, cryosectioned, and stained for lipid with Oil red O. Tissues were evaluated by an ACVP board-certified veterinary pathologist. Representative sections were images with a CoolSnap digital camera (Roper Scientific, Inc., Tucson, AZ) mounted on a Nikon E600 light microscope (Nikon Inc. Instrument Group).
Statistical analysis
Results are expressed as mean ± sd. Differences were analyzed by use of one-way ANOVA. Two-group comparisons were evaluated by the Mann-Whitney test using the Bonferroni correction for multiple comparisons. Statistical significance was considered when P < 0.05.
Acknowledgments
We thank our colleagues at ZymoGenetics, including S. Jaspers and S. Dillon for helpful discussions; B. Stamm, K. Kim, K. Bontadelli, D. Cutler, L. Lockwood, K. Foley, R. Schreckhise, and Z. Chen for help with the generation and analysis of the GPHB5 transgenic and knockout mice; C. Zuvela, M. Dixon-Brooks, D. Christensen, S. Nicholson, E. Chadwick, and L. Mayr for animal husbandry; David Taft for Northern analysis; and Megan Lantry for SEC-MALS.
Present address for P.J.W.: Nanostring Technologies, 201 Elliott Avenue West, Seattle, Washington 98119.
Abbreviations
- AP
Alkaline phosphatase;
- BHK
baby hamster kidney;
- CG
chorionic gonadotropin;
- CGH
corticotroph-derived glycoprotein hormone;
- CHO
Chinese hamster ovary;
- CRE
cAMP response element;
- DAB
diaminobenzidine;
- FITC
fluorescein isothiocyanate;
- FS
folliculo-stellate;
- GPHA2
glycoprotein hormone α 2;
- GPHB5
glycoprotein hormone β 5;
- H&E
hematoxylin and eosin;
- HPA
hypothalamus-pituitary-adrenal;
- HPT
hypothalamus-pituitary-thyroid;
- IP
immunoperoxidase;
- LGR
leucine-rich G protein-coupled receptor;
- rh
recombinant human;
- TSHR
TSH receptor.