Abstract
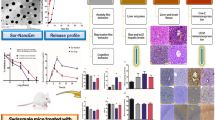
Minimal information is available on the oral bioavailability and liver-targeting properties of sorafenib solid lipid nanoparticles (SRF-SLNs) in rats. In this study, SRF-SLNs were prepared via the combined methods of high-speed shearing and ultrasonic treatment. SRF-SLN formulations were also optimized. Particle size, zeta potential, entrapment efficiency (EE), and drug loading (DL) were used as indices for the evaluation of the as-prepared SRF-SLNs. SRF concentration was determined by the high-performance liquid chromatography method. Results showed that the average EE and DL of SRF-SLNs were 89.87 and 5.39%. The average particle size, polydispersity index, and zeta potential of SRF-SLNs were 77.16 nm, 0.28, and − 18.1 mV, respectively. The results of the stability test showed that SRF-SLNs remained stable for more than 1 month at room temperature. After oral administration to rats (7.5 mg/kg), the liver-targeting evaluation results showed that the average drug selectivity index value of SRF-SLNs was 2.20 times higher, than that of the SRF-suspension. Furthermore, the area under the concentration–time curve of SRF increased by 66.7% in the SRF-SLN group comparing with that in the SRF-suspension. Our results suggested that SLNs were a promising approach for the oral delivery of SRF.





Similar content being viewed by others
References
Tang K, Li Y, Chen XG. Advance in the research of sorafenib, a multiple targeted antitutor agent. Chin New Drugs J. 2011;20(24):2434–41.
Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med. 2007;356(2):125–34.
Strumberg D, Clark JW, Awada A, Moore MJ, Richly H, Hendlisz A, et al. Safety, pharmacokinetics, and preliminary antitumor activity of sorafenib: a review of four phase I trials in patients with advanced refractory solid tumors. Oncologist. 2007;12(4):426–37.
Liu C, Chen Z, Chen Y, Lu J, Li Y, Wang S, et al. Improving Oral Bioavailability of sorafenib by optimizing the “Spring” and “Parachute” based on molecular interaction mechanisms. Mol Pharm. 2016;13(2):599–608.
Widemann BC, Kim A, Fox E, Baruchel S, Adamson PC, Ingle AM, et al. A phase I trial and pharmacokinetic study of sorafenib in children with refractory solid tumors or leukemias: a Children’s Oncology Group Phase I Consortium report. Clin Cancer Res. 2012;18(21):6011–22.
Wang XQ, Fan JM, Liu YO, Zhao B, Jia ZR, Zhang Q. Bioavailability and pharmacokinetics of sorafenib suspension, nanoparticles and nanomatrix for oral administration to rat. Int J Pharm. 2011;419(1–2):339–46.
Zhang ZW, Niu BH, Chen J, He XY, Bao XY, Zhu JH, et al. The use of lipid-coated nanodiamond to improve bioavailability and efficacy of sorafenib in resisting metastasis of gastric cancer. Biomaterials. 2014;35(15):4565–72.
Guo Y, Zhong T, Duan XC, Zhang S, Yao X, Yin YF, et al. Improving anti-tumor activity of sorafenib tosylate by lipid- and polymer-coated nanomatrix. Drug Deliv. 2017;24(1):270–7.
Muller RH, Runge SA, Ravelli V, Thunemann AF, Mehnert W, Souto EB. Cyclosporine-loaded solid lipid nanoparticles (SLN): drug-lipid physicochemical interactions and characterization of drug incorporation. Eur J Pharm Biopharm. 2008;68(3):535–44.
Hu L, Tang X, Cui F. Solid lipid nanoparticles (SLNs) to improve oral bioavailability of poorly soluble drugs. J Pharm Pharmacol. 2004;56(12):1527–35.
Kumar A, Kumar P, Gupta M, Kamath A, Maheshwari ASingh S. Profile of clients tested HIV positive in a voluntary counseling and testing center of a District Hospital, Udupi, South Kannada. Indian J Community Med. 2008;33(3):156–9.
Ding LX, Chai JL, Li H, Yang SS, Yu L, Li SJ, et al. New progress of study on solid lipid nanoparticles. Med J Chin People’s Health. 2014;26(20):69–71.
Jia W, Zhang H, Zhang Y. Preparation and in vitro drug release of sorafenib-Eudragit RS nanoparticles. China Pharm. 2015;18(04):541–544+554.
Zhang H, Zhang FM, Yan SJ. Preparation, in vitro release, and pharmacokinetics in rabbits of lyophilized injection of sorafenib solid lipid nanoparticles. Int J Nanomedicine. 2012;7:2901–10.
Grillone A, Riva ER, Mondini A, Forte C, Calucci L, Innocenti C, et al. Active targeting of sorafenib: preparation, characterization, and in vitro testing of drug-loaded magnetic solid lipid nanoparticles. Adv Healthc Mater. 2015;4(11):1681–90.
Cai Z, Wang Y, Zhu LJ, Liu ZQ. Nanocarriers: a general strategy for enhancement of oral bioavailability of poorly absorbed or pre-systemically metabolized drugs. Curr Drug Metab. 2010;11(2):197–207.
Xiao L, Yi T, Liu Y. A new self-microemulsifying mouth dissolving film to improve the oral bioavailability of poorly water soluble drugs. Drug Dev Ind Pharm. 2013;39(9):1284–90.
Wang XQ, Zhang Q. pH-sensitive polymeric nanoparticles to improve oral bioavailability of peptide/protein drugs and poorly water-soluble drugs. Eur J Pharm Biopharm. 2012;82(2):219–29.
Kim DH, Kim MD, Choi CW, Chung CW, Ha SH, Kim CH, et al. Antitumor activity of sorafenib-incorporated nanoparticles of dextran/poly(dl-lactide-co-glycolide) block copolymer. Nanoscale Res Lett. 2012;7(1):91.
Lee EQ, Kuhn J, Lamborn KR, Abrey L, DeAngelis LM, Lieberman F, et al. Phase I/II study of sorafenib in combination with temsirolimus for recurrent glioblastoma or gliosarcoma: North American Brain Tumor Consortium study 05-02. Neuro-Oncology. 2012;14(12):1511–8.
Kelley RK, Nimeiri HS, Munster PN, Vergo MT, Huang Y, Li CM, et al. Temsirolimus combined with sorafenib in hepatocellular carcinoma: a phase I dose-finding trial with pharmacokinetic and biomarker correlates. Ann Oncol. 2013;24(7):1900–7.
Lathia C, Lettieri J, Cihon F, Gallentine M, Radtke M, Sundaresan P. Lack of effect of ketoconazole-mediated CYP3A inhibition on sorafenib clinical pharmacokinetics. Cancer Chemother Pharmacol. 2006;57(5):685–92.
Shimada M, Okawa H, Maejima T, Yanagi T, Hisamichi K, Matsuura M, et al. A quantitative HPLC-UV method for determination of serum sorafenib and sorafenib N-oxide and its application in hepatocarcinoma patients. Tohoku J Exp Med. 2014;233(2):103–12.
Haouala A, Zanolari B, Rochat B, Montemurro M, Zaman K, Duchosal MA, et al. Therapeutic drug monitoring of the new targeted anticancer agents imatinib, nilotinib, dasatinib, sunitinib, sorafenib and lapatinib by LC tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2009;877(22):1982–96.
Sparidans RW, Vlaming ML, Lagas JS, Schinkel AH, Schellens JHBeijnen JH. Liquid chromatography-tandem mass spectrometric assay for sorafenib and sorafenib-glucuronide in mouse plasma and liver homogenate and identification of the glucuronide metabolite. J Chromatogr B Analyt Technol Biomed Life Sci. 2009;877(3):269–76.
Heinz WJ, Kahle K, Helle-Beyersdorf A, Schirmer D, Lenker U, Keller D, et al. High-performance liquid chromatographic method for the determination of sorafenib in human serum and peritoneal fluid. Cancer Chemother Pharmacol. 2011;68(1):239–45.
Acknowledgements
The work was supported by a grant from the National Natural Science Foundation of China (No. 81560631), Distinguished Young Scholars Foundation of Jiangxi Province (No. 20162BCB23022) and the Innovation Fund Designated for Graduate Students of Nanchang University (No. cx2016296 and No. cx2016297).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The animal study was conducted in accordance with the Guidelines for Animal Experiments and approved by the Animal Ethics Committee of Nanchang University.
Conflict of Interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Wang, H., Wang, H., Yang, W. et al. Improved Oral Bioavailability and Liver Targeting of Sorafenib Solid Lipid Nanoparticles in Rats. AAPS PharmSciTech 19, 761–768 (2018). https://doi.org/10.1208/s12249-017-0901-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-017-0901-3