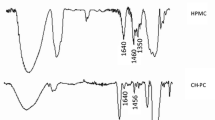
Abstract
Two groups of fluconazole mucoadhesive buccal discs were prepared: (a) Fluconazole buccal discs prepared by direct compression containing bioadhesive polymers, namely, Carbopol 974p (Cp), sodium carboxymethyl cellulose (SCMC), or sodium alginate (SALG) in combination with hydroxypropyl methylcellulose (HPMC) or hydroxyethyl cellulose (HEC). (b) Fluconazole buccal discs prepared by freeze drying containing different polymer combinations (SCMC/HPMC, Cp/HPMC, SALG/HPMC, and chitosan/SALG). The prepared discs were evaluated by investigating their release pattern, swelling capacity, mucoadhesion properties, and in vitro adhesion time. In vivo evaluation of the buccal disc and in vivo residence times were also performed. Fluconazole salivary concentration after application of fluconazole buccal systems to four healthy volunteers was determined using microbiological assay and high-performance liquid chromatography. SCMC/HPMC buccal disc prepared by direct compression could be considered comparatively superior mucoadhesive disc regarding its in vitro adhesion time, in vivo residence time, and in vitro/in vivo release rates of the drug. Determination of the amount of drug released in saliva after application of the selected fluconazole disc confirmed the ability of the disc to deliver the drug over a period of approximately 5 h and to reduce side effects and possibility of drug interaction encountered during systemic therapy of fluconazole, which would be beneficial in the case of oral candidiasis.






Similar content being viewed by others
References
R. Khanna, S. P. Agarwal, and A. Ahuja. Mucoadhesive buccal drug delivery: a potential alternative to conventional therapy. Ind. J. Pharm. Sci. 60:1–11 (1998).
Martindale; The Complete Drug Reference, 34th Edition. In S. C. Sweetman (ed.), The Pharmaceutical Press London 372 (2005).
C. Koks, P. Meenhorst, M. Hillebrand, and A. Bult. Pharmacokinetics of fluconazole in saliva and plasma after administration of an oral suspension and capsules. Antimicrob. Agents Chemother. 40:1935–1937 (1996).
A. E. Collins, and P. B. Deasy. Bioadhesive lozenge for the improved delivery of cetylpyredinium chloride. J. Pharm. Sci. 792:116–119 (1990).
S. Bouckaert, R. A. Lefebvre, and J. P. Remon. In vitro/in vivo correlation of the bioadhesive properties of a buccal bioadhesive miconazole slow-release tablet. Pharm. Res. 106:853–856 (1993).
T. S. Owens, R. J. Dansereau, and A. Sakr. Development and evaluation of extended release bioadhesive sodium fluoride tablets. Int. J. Pharm. 2881:109–122 (2005).
M. L. Vueba, L. A. E. B. deCarvalhob, F. Veigaa, J. J. Sousaa, and M. E. Pinaa. Influence of cellulose ether polymers on ketoprofen release from hydrophilic matrix tablets. Eur. J. Pharm. Biopharm. 58:51–59 (2004).
M. S. El-Samaligy, S. A. Yahia, and E. B. Basalious. Formulation and evaluation of diclofenac sodium buccoadhesive discs. Int. J. Pharm. 286:27–39 (2004).
R. W. Korsmeyer, R. Gurny, E. Doelker, P. Buri, and N. A. Peppas. Mechanisms of solute release from porous hydrophillic polymers. Int. J. Pharm. 15:25–35 (1983).
J. E. Mockel, and B. C. Lippold. Zero-order drug release from hydrocolloid matrices. Pharm. Res. 90:1066–1070 (1993).
N. A. Peppas, and J. J. Sahlin. A simple equation for the description of solute release. III. Coupling of diffusion and relaxation. Int. J. Pharm. 57:169–172 (1989).
M. Ozyazıcı, E. H. Gokce, and G. Ertan. Release and diffusional modeling of metronidazole lipid matrices. Eur. J. Pharm. Biopharm. 63:331–339 (2006).
B. Parodi, E. Russo, G. Caviglioli, S. Cafaggi, and G. Bignardi. Development and characterization of a buccoadhesive dosage form of oxycodone hydrochloride. Drug Dev. Ind. Pharm. 22:445–450 (1996).
K. Nam, J. Watanabe, and K. Ishihara. Modeling of swelling and drug release behavior of spontaneously forming hydrogels composed of phospholipid polymers. Int. J. Pharm. 2751–2:259–269 (2004).
R. Y. Han, J. Y. Fang, K. C. Sung, and O. Y. Hu. Mucoadhesive buccal disks for novel nalbuphine prodrug controlled delivery: effect of formulation variables on drug release and mucoadhesive performance. Int. J. Pharm. 1772:201–209 (1999).
T. Save, M. U. Stah, A. R. Ghamande, and P. Venkitachalam. Comparative study of buccoadhesive formulations and sublingual capsules of nifedipine. J. Pharm. Pharmacol. 463:192–195 (1994).
J. Ali, R. Khar, A. Ahuja, and R. Kalra. Buccoadhesive erodible disk for treatment of oro-dental infections: design and characterization. Int. J. Pharm. 238:93–103 (2002).
C. H. W. Koks, H. Rosing, P. L. Meenhorst, A. Bult, and J. H. Beijnen. High-performance liquid chromatographic determination of the antifungal drug fluconazole in plasma and saliva of human immunodeficiency virus-infected patients. J. Chromatogr. B. 663:345–351 (1995).
The British Pharmacopeia, British Pharmacopoeia Commission, HMSO, London, 2007: Electronic version.
J. Sujja-areevath, D. L. Munday, P. J. Cox, and K. A. Khan. Relationship between swelling, erosion and drug release in hydrophillic natural gum mini-matrix formulations. Eur. J. Pharm. Sci. 6:207–217 (1998).
D. S. Roy, and B. D. Rohera. Comparative evaluation of rate of hydration and matrix erosion of HEC and HPC and study of drug release from their matrices. Eur. J. Pharm. Sci. 16:193–199 (2002).
L. Perioli, V. Ambrogi, D. Rubini, S. Giovagnoli, M. Ricci, P. Blasi, and C. Rossi. Novel mucoadhesive buccal formulation containing metronidazole for the treatment of periodontal disease. J. Control. Rel. 95:521–533 (2004).
T. Nagai, and R. Konishi. Buccal/gingival drug delivery systems. J. Control. Rel. 61:353–360 (1987).
S. A. Yehia, O. N. El-Gazayerly, E. B. Basalious, and M. S. El-Din. Preparation and evaluation of fluconazole mucoadhesive buccal films. AAPS J. 8S2 (2006) Abstract T3172.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yehia, S.A., El-Gazayerly, O.N. & Basalious, E.B. Design and In Vitro/In Vivo Evaluation of Novel Mucoadhesive Buccal Discs of an Antifungal Drug: Relationship Between Swelling, Erosion, and Drug Release. AAPS PharmSciTech 9, 1207–1217 (2008). https://doi.org/10.1208/s12249-008-9166-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-008-9166-1