Abstract
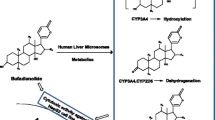
3-Acetyl-11-keto-β-boswellic acid (AKBA) and 11-keto-β-boswellic acid (KBA) are widely used in the clinic as anti-inflammatory drugs. However, these drugs have the poor bioavailability, which may be caused by their extensive metabolism. In this study, we systemically characterized both phase I and II metabolism of AKBA and KBA in vitro. In total, four major metabolites were firstly biosynthesized and identified using 1D and 2D NMR spectroscopy. Among them, three metabolites were novel. The kinetic parameters (K m , V max , CL int, and K i ) were also analyzed systematically in various biological samples. Finally, the deacetylation of AKBA and hydroxylation of KBA were confirmed to be the major metabolic pathways based on their large CL int and the high amounts of KBA (46.7%) and hydroxylated KBA (50.8%) along with a low amount of AKBA (2.50%) in human primary hepatocytes. Carboxylesterase 2 (CE2) selectively catalyzed the deacetylation of AKBA to form KBA. Although CYP3A4, CYP3A5, and CYP3A7 catalyzed the metabolism of KBA, CYP3A4 played a predominant role in the hydroxylation reaction of KBA in human. Notably, deacetylation and regioselective hydroxylation exhibited considerable species differences. Deacetylation was only observed in human liver microsomes and primary human hepatocytes; 21- and 20-mono-hydroxylation of KBA were primarily observed in human, monkey, and dog; and 16- and 30-mono-hydroxylation were observed in other species. More importantly, all four mono-hydroxylation metabolites exhibited a moderate anti-inflammatory activity. The 21- and 20-hydroxylation metabolites inhibited the expression of iNOS, the LPS-induced activation of IkBα and p65 phosphorylation, and suppressed p65 nuclear translocation in RAW264.7 cells.







Similar content being viewed by others
Abbreviations
- AKBA:
-
3-Acetyl-11-keto-β-boswellic acid
- KBA:
-
11-Keto-β-boswellic acid
- HLM:
-
Human liver microsomes
- HIM:
-
Human intestine microsomes
- CyLM:
-
Monkey liver microsomes
- RLM:
-
SD rat liver microsomes
- AChE:
-
Acetylcholinesterase
- BuChE:
-
Butyrylcholinesterase
- HSA:
-
Human serum albumin
- RSA:
-
Rat serum albumin
- PLM:
-
Pig liver microsomes
- DLM:
-
Dog liver microsomes
- MLM:
-
Mouse liver microsomes
- RaLM:
-
Rabbit liver microsomes
- GLM:
-
Guinea pig liver microsomes
- CEs:
-
Carboxylesterases
- NADPH:
-
β-Nicotinamide adenine dinucleotide phosphate disodium salt
- M-2:
-
21β-Hydroxy-11-keto-β-boswellic acid
- M-3:
-
16β-Hydroxy-11-keto-β-boswellic acid
- M-4:
-
30-Hydroxy-11-keto-β-boswellic acid
- M-5:
-
20β-Hydroxy-11-keto-β-boswellic acid
- HA:
-
Huperzine A
- iso-OMPA:
-
Tetraisopropylpyrophosphoramide
- BNPP:
-
Bis-p-nitrophenyl phosphate
- LPA:
-
Loperamide
- LPS:
-
Lipopolysaccharide
- TEPA:
-
Triethylenethiophosphoramide
References
Hamidpour R, Hamidpour S, Hamidpour M, Shahlari M. Frankincense (ru xiang; boswellia species): from the selection of traditional applications to the novel phytotherapy for the prevention and treatment of serious diseases. J Tradit Complement Med. 2013;3(4):221–6. doi:10.4103/2225-4110.119723.
Singh S, Khajuria A, Taneja SC, Johri RK, Singh J, Qazi GN. Boswellic acids: a leukotriene inhibitor also effective through topical application in inflammatory disorders. Phytomedicine. 2008;15(6-7):400–7. doi:10.1016/j.phymed.2007.11.019.
Zhang YS, Xie JZ, Zhong JL, Li YY, Wang RQ, Qin YZ, et al. Acetyl-11-keto-beta-boswellic acid (AKBA) inhibits human gastric carcinoma growth through modulation of the Wnt/beta-catenin signaling pathway. Biochim Biophys Acta. 2013;1830(6):3604–15. doi:10.1016/j.bbagen.2013.03.003.
Assimopoulou AN, Zlatanos SN, Papageorgiou VP. Antioxidant activity of natural resins and bioactive triterpenes in oil substrates. Food Chem. 2005;92(4):721–7. doi:10.1016/j.foodchem.2004.08.0333.
Raja AF, Ali F, Khan IA, Shawl AS, Arora DS. Acetyl-11-keto-beta-boswellic acid (AKBA); targeting oral cavity pathogens. BMC Res Notes. 2011;4:406. doi:10.1186/1756-0500-4-406.
Raja AF, Ali F, Khan IA, Shawl AS, Arora DS, Shah BA, et al. Antistaphylococcal and biofilm inhibitory activities of acetyl-11-keto-beta-boswellic acid from Boswellia serrata. BMC Microbiol. 2011;11:54. doi:10.1186/1471-2180-11-54.
Poeckel D, Werz O. Boswellic acids: biological actions and molecular targets. Curr Med Chem. 2006;13(28):3359–69.
Ernst E. Frankincense: systematic review. BMJ. 2008;337:a2813. doi:10.1136/bmj.a2813.
Gerhardt H, Seifert F, Buvari P, Vogelsang H, Repges R. Therapy of active Crohn disease with Boswellia serrata extract H 15. Z Gastroenterol. 2001;39(1):11–7. doi:10.1055/s-2001-10708.
Gupta I, Parihar A, Malhotra P, Singh GB, Ludtke R, Safayhi H, et al. Effects of Boswellia serrata gum resin in patients with ulcerative colitis. Eur J Med Res. 1997;2(1):37–43.
Cuaz-Perolin C, Billiet L, Bauge E, Copin C, Scott-Algara D, Genze F, et al. Antiinflammatory and antiatherogenic effects of the NF-kappaB inhibitor acetyl-11-keto-beta-boswellic acid in LPS-challenged ApoE-/- mice. Arterioscler Thromb Vasc Biol. 2008;28(2):272–7. doi:10.1161/ATVBAHA.107.155606.
Takada Y, Ichikawa H, Badmaev V, Aggarwal BB. Acetyl-11-keto-beta-boswellic acid potentiates apoptosis, inhibits invasion, and abolishes osteoclastogenesis by suppressing NF-kappa B and NF-kappa B-regulated gene expression. J Immunol. 2006;176(5):3127–40.
Altmann A, Poeckel D, Fischer L, Schubert-Zsilavecz M, Steinhilber D, Werz O. Coupling of boswellic acid-induced Ca2+ mobilisation and MAPK activation to lipid metabolism and peroxide formation in human leucocytes. Br J Pharmacol. 2004;141(2):223–32. doi:10.1038/sj.bjp.0705604.
Sailer ER, Subramanian LR, Rall B, Hoernlein RF, Ammon HP, Safayhi H. Acetyl-11-keto-beta-boswellic acid (AKBA): structure requirements for binding and 5-lipoxygenase inhibitory activity. Br J Pharmacol. 1996;117(4):615–8.
Poeckel D, Tausch L, Kather N, Jauch J, Werz O. Boswellic acids stimulate arachidonic acid release and 12-lipoxygenase activity in human platelets independent of Ca2+ and differentially interact with platelet-type 12-lipoxygenase. Mol Pharmacol. 2006;70(3):1071–8. doi:10.1124/mol.106.024836.
Safayhi H, Rall B, Sailer ER, Ammon HP. Inhibition by boswellic acids of human leukocyte elastase. J Pharmacol Exp Ther. 1997;281(1):460–3.
Tausch L, Henkel A, Siemoneit U, Poeckel D, Kather N, Franke L, et al. Identification of human cathepsin G as a functional target of boswellic acids from the anti-inflammatory remedy frankincense. J Immunol. 2009;183(5):3433–42. doi:10.4049/jimmunol.0803574.
Siemoneit U, Koeberle A, Rossi A, Dehm F, Verhoff M, Reckel S, et al. Inhibition of microsomal prostaglandin E2 synthase-1 as a molecular basis for the anti-inflammatory actions of boswellic acids from frankincense. Br J Pharmacol. 2011;162(1):147–62. doi:10.1111/j.1476-5381.2010.01020.x.
Siemoneit U, Hofmann B, Kather N, Lamkemeyer T, Madlung J, Franke L, et al. Identification and functional analysis of cyclooxygenase-1 as a molecular target of boswellic acids. Biochem Pharmacol. 2008;75(2):503–13. doi:10.1016/j.bcp.2007.09.010.
Wang C, Dong PP, Zhang LY, Huo XK, Zhang BJ, Wang CY, et al. Regio- and stereo-selective oxidation of beta-boswellic acids transformed by filamentous fungi. RSC Adv. 2015;5(17):12717–25. doi:10.1039/c4ra16459h.
Wang Y, Sun Y, Wang C, Huo X, Liu P, Wang C, et al. Biotransformation of 11-keto-beta-boswellic acid by Cunninghamella blakesleana. Phytochemistry. 2013;96:330–6. doi:10.1016/j.phytochem.2013.07.018.
Sun Y, Liu D, Xi R, Wang X, Wang Y, Hou J, et al. Microbial transformation of acetyl-11-keto-beta-boswellic acid and their inhibitory activity on LPS-induced NO production. Bioorg Med Chem Lett. 2013;23(5):1338–42. doi:10.1016/j.bmcl.2012.12.086.
Bleif S, Hannemann F, Zapp J, Hartmann D, Jauch J, Bernhardt R. A new Bacillus megaterium whole-cell catalyst for the hydroxylation of the pentacyclic triterpene 11-keto-beta-boswellic acid (KBA) based on a recombinant cytochrome P450 system. Appl Microbiol Biotechnol. 2012;93(3):1135–46. doi:10.1007/s00253-011-3467-0.
Brill E, Hannemann F, Zapp J, Bruning G, Jauch J, Bernhardt R. A new cytochrome P450 system from Bacillus megaterium DSM319 for the hydroxylation of 11-keto-beta-boswellic acid (KBA). Appl Microbiol Biotechnol. 2014;98(4):1701–17. doi:10.1007/s00253-013-5029-0.
Gerbeth K, Husch J, Fricker G, Werz O, Schubert-Zsilavecz M, Abdel-Tawab M. In vitro metabolism, permeation, and brain availability of six major boswellic acids from Boswellia serrata gum resins. Fitoterapia. 2013;84:99–106. doi:10.1016/j.fitote.2012.10.009.
Buchele B, Simmet T. Analysis of 12 different pentacyclic triterpenic acids from frankincense in human plasma by high-performance liquid chromatography and photodiode array detection. J Chromatogr B Analyt Technol Biomed Life Sci. 2003;795(2):355–62.
Abdel-Tawab M, Werz O, Schubert-Zsilavecz M. Boswellia serrata: an overall assessment of in vitro, preclinical, pharmacokinetic and clinical data. Clin Pharmacokinet. 2011;50(6):349–69. doi:10.2165/11586800-000000000-00000.
Sharma S, Thawani V, Hingorani L, Shrivastava M, Bhate VR, Khiyani R. Pharmacokinetic study of 11-Keto beta-Boswellic acid. Phytomedicine. 2004;11(2-3):255–60.
Kruger P, Daneshfar R, Eckert GP, Klein J, Volmer DA, Bahr U, et al. Metabolism of boswellic acids in vitro and in vivo. Drug Metab Dispos: Biol Fate Chem. 2008;36(6):1135–42. doi:10.1124/dmd.107.018424.
Senter PD, Beam KS, Mixan B, Wahl AF. Identification and activities of human carboxylesterases for the activation of CPT-11, a clinically approved anticancer drug. Bioconjug Chem. 2001;12(6):1074–80.
Humerickhouse R, Lohrbach K, Li L, Bosron WF, Dolan ME. Characterization of CPT-11 hydrolysis by human liver carboxylesterase isoforms hCE-1 and hCE-2. Cancer Res. 2000;60(5):1189–92.
Oda S, Fukami T, Yokoi T, Nakajima M. A comprehensive review of UDP-glucuronosyltransferase and esterases for drug development. Drug Metab Pharmacokinet. 2015;30(1):30–51. doi:10.1016/j.dmpk.2014.12.001.
Zuber R, Anzenbacherova E, Anzenbacher P. Cytochromes P450 and experimental models of drug metabolism. J Cell Mol Med. 2002;6(2):189–98.
Thummel KE, Wilkinson GR. In vitro and in vivo drug interactions involving human CYP3A. Annu Rev Pharmacol Toxicol. 1998;38:389–430. doi:10.1146/annurev.pharmtox.38.1.389.
Guengerich FP. Cytochrome P-450 3A4: regulation and role in drug metabolism. Annu Rev Pharmacol Toxicol. 1999;39:1–17. doi:10.1146/annurev.pharmtox.39.1.1.
Leeder JS, Gaedigk R, Marcucci KA, Gaedigk A, Vyhlidal CA, Schindel BP, et al. Variability of CYP3A7 expression in human fetal liver. J Pharmacol Exp Ther. 2005;314(2):626–35. doi:10.1124/jpet.105.086504.
Domanski TL, Finta C, Halpert JR, Zaphiropoulos PG. cDNA cloning and initial characterization of CYP3A43, a novel human cytochrome P450. Mol Pharmacol. 2001;59(2):386–92.
Martignoni M, Groothuis GM, de Kanter R. Species differences between mouse, rat, dog, monkey and human CYP-mediated drug metabolism, inhibition and induction. Expert Opin Drug Metab Toxicol. 2006;2(6):875–94. doi:10.1517/17425255.2.6.875.
Grubic Z, Sket D, Brzin M. Iso-OMPA-induced potentiation of soman toxicity in rat correlates with the inhibition of plasma carboxylesterases. Arch Toxicol. 1988;62(5):398–9.
Feng L, Liu ZM, Hou J, Lv X, Ning J, Ge GB, et al. A highly selective fluorescent ESIPT probe for the detection of human carboxylesterase 2 and its biological applications. Biosens Bioelectron. 2014;65C:9–15. doi:10.1016/j.bios.2014.10.002.
Kurokawa T, Fukami T, Nakajima M. Characterization of species differences in tissue diltiazem deacetylation identifies Ces2a as a rat-specific diltiazem deacetylase. Drug Metab Dispos: Biol Fate Chem. 2015;43(8):1218–25. doi:10.1124/dmd.115.064089.
Tian X, Liang S, Wang C, Wu B, Ge G, Deng S, et al. Regioselective glucuronidation of andrographolide and its major derivatives: metabolite identification, isozyme contribution, and species differences. AAPS J. 2015;17(1):156–66. doi:10.1208/s12248-014-9658-8.
Ning J, Yu ZL, Hu LH, Wang C, Huo XK, Deng S, et al. Characterization of phase I metabolism of resibufogenin and evaluation of the metabolic effects on its antitumor activity and toxicity. Drug Metab Dispos: Biol Fate Chem. 2015;43(3):299–308. doi:10.1124/dmd.114.060996.
Dinger J, Meyer MR, Maurer HH. Development of an in vitro cytochrome P450 cocktail inhibition assay for assessing the inhibition risk of drugs of abuse. Toxicol Lett. 2014;230(1):28–35. doi:10.1016/j.toxlet.2014.08.004.
Walsky RL, Obach RS, Hyland R, Kang P, Zhou S, West M, et al. Selective mechanism-based inactivation of CYP3A4 by CYP3cide (PF-04981517) and its utility as an in vitro tool for delineating the relative roles of CYP3A4 versus CYP3A5 in the metabolism of drugs. Drug Metab Dispos: Biol Fate Chem. 2012;40(9):1686–97. doi:10.1124/dmd.112.045302.
Ge GB, Ning J, Hu LH, Dai ZR, Hou J, Cao YF, et al. A highly selective probe for human cytochrome P450 3A4: isoform selectivity, kinetic characterization and its applications. Chem Commun. 2013;49(84):9779–81. doi:10.1039/c3cc45250f.
Zhu L, Ge G, Zhang H, Liu H, He G, Liang S, et al. Characterization of hepatic and intestinal glucuronidation of magnolol: application of the relative activity factor approach to decipher the contributions of multiple UDP-glucuronosyltransferase isoforms. Drug Metab Dispos: Biol Fate Chem. 2012;40(3):529–38. doi:10.1124/dmd.111.042192.
Yu Z, Guo W, Ma X, Zhang B, Dong P, Huang L, et al. Gamabufotalin, a bufadienolide compound from toad venom, suppresses COX-2 expression through targeting IKKbeta/NF-kappaB signaling pathway in lung cancer cells. Mol Cancer. 2014;13:203. doi:10.1186/1476-4598-13-203.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (Nos. 81274047, 81473334, and 81503201), the Distinguished Professor of Liaoning Province, Dalian Outstanding Youth Science and Technology Talent program (2014J11JH132 and 2015J12JH201), and the Liaoning Bai Qian Wan Talent Program and Innovation Team of Dalian Medical University.
Author Contributions
Xiaochi Ma, Yonglei Cui, and Xiangge Tian designed the experiments. Yonglei Cui and Jing Ning performed the experiments. Chao Wang, Zhenlong Yu, and Yan Wang analyzed the data. Xiaokui Huo, Lingling Jin, Sa Deng, and Baojing Zhang prepared the figures. Yonglei Cui and Xiangge Tian wrote the main text. All authors reviewed the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Yonglei Cui and Xiangge Tian contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 1430 kb)
Rights and permissions
About this article
Cite this article
Cui, Y., Tian, X., Ning, J. et al. Metabolic Profile of 3-Acetyl-11-Keto-β-Boswellic Acid and 11-Keto-β-Boswellic Acid in Human Preparations In Vitro, Species Differences, and Bioactivity Variation. AAPS J 18, 1273–1288 (2016). https://doi.org/10.1208/s12248-016-9945-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-016-9945-7