Abstract
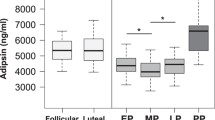
The purpose of this study was to examine circulating maternal follistatin-like 3 (FSTL-3) by gestational age and obesity in pregnancy and preeclampsia. FSTL-3 was quantified in maternal plasma collected in each trimester from prepregnancy body mass index-determined groups: 15 lean and 24 obese controls and 20 obese women who developed preeclampsia. Repeated measures mixed models and logistic regression were conducted (P ≤ .05). FSTL-3 was not related to maternal adiposity. FSTL-3 changed across pregnancy in lean controls and obese preeclampsia but not in obese controls. FSTL-3 was higher in preeclampsia in the second trimester compared to lean controls and in the third trimester compared to both control groups. Elevated FSTL-3 at mid-gestation was associated with an increased odds of preeclampsia (odds ratio 3.15; 95% confidence interval 1.19-8.36; P = .02). Elevated FSTL-3 concentrations were attributable to preeclampsia and were associated with increased likelihood of later developing preeclampsia, suggesting further study as a biomarker prior to clinically evident disease.
Similar content being viewed by others
References
American College of Obstetricians and Gynecologists. ACOG practice bulletin. Diagnosis and management of preeclampsia and eclampsia. Number 33, January 2002. Obstet Gynecol. 2002;99(1):159–167.
Sibai B, Dekker G, Kupferminc M. Pre-eclampsia. Lancet. 2005;365(9461):785–799.
World Health Organization. Maternal and perinatal health. http://www.who.int/reproductivehealth/topics/maternal_perinatal/epidemiology/en/index.html. Accessed March 10, 2013.
Founds S, Conley YP, Lyons-Weiler JF, Jeyabalan A, Hogge WA, Conrad KP. Global gene expression in first trimester placentas of women destined to develop preeclampsia. Placenta. 2009;30(1):15–24.
Founds SA, Terhorst LA, Conrad KP, Hogge WA, Jeyabalan A, Conley YP. Gene expression of eight candidates in first trimester preeclampsia placenta. Biol Res Nurs. 2011;13(2):134–139.
Tortoriello DV, Sidis Y, Holtzman DA, Holmes WE, Schneyer AL. Human follistatin-related protein: a structural homologue of follistatin with nuclear localization. Endocrinology. 2001;142(8):3426–3434.
Schneyer A, Sidis Y, Xia Y, et al. Differential actions of follistatin and follistatin-like 3. Molec Cell Endocr. 2004;225(1–2):25–28.
Maguer Satta V, Rimokh R. FLRG, member of the follistatin family, a new player in hematopoiesis. Mol Cell Endocrinol. 2004;225(1–2):109–118.
Hill JJ, Davies MV, Pearson AA, et al. The myostatin propeptide and the follistatin-related gene are inhibitory binding proteins of myostatin in normal serum. J Biol Chem. 2002;277(43):40735–40741.
Sidis Y, Tortoriello DV, Holmes WE, Pan Y, Keutmann HT, Schneyer AL. Follistatin-related protein and follistatin differentially neutralize endogenous vs. exogenous activin. Endocrinology. 2002;143(5):1613–1624.
Guzeloglu-Kayisli O, Kayisli UA, Taylor HS. The role of growth factors and cytokines during implantation: endocrine and paracrine interactions. Semin Reprod Med. 2009;27(1):62–79.
Caniggia I, Winter J, Lye SJ, Post M. Oxygen and placental development during the first trimester: implications for the pathophysiology of pre-eclampsia. Placenta. 2000;21(suppl A):S25–S30.
Brown ML, Bonomi L, Ungerleider N, et al. Follistatin and follistatin like-3 differentially regulate adiposity and glucose homeostasis. Obesity. 2011;19(10):1940–1949.
Allen DL, Cleary AS, Speaker KJ, et al. Myostatin, activin receptor IIb, and follistatin-like-3 gene expression are altered in adipose tissue and skeletal muscle of obese mice. Am J Physiol Endocrinol Metab. 2008;294(5):E918–E927.
Mukherjee A, Sidis Y, Mahan A, et al. FSTL3 deletion reveals roles for TGF-beta family ligands in glucose and fat homeostasis in adults. Proc Natl Acad Sci U S A. 2007;104(4):1348–1353.
Ciarmela P, Florio P, Toti P, et al. Human placenta and fetal membranes express follistatin-related gene mRNA and protein. J Endocrinol Invest. 2003;26(7):641–645.
Khong T, De Wolf F, Robertson WB, Brosens I. Inadequate maternal vascular response to placentation in pregnancies complicated by pre-eclampsia and by small-for-gestational age infants. Br J Obstet Gynaecol. 1986;93(10):1049–1059.
Meekins J, Pijnenborg R, Hanssens M, McFadyen IR, van Asshe A. A study of placental bed spiral arteries and trophoblast invasion in normal and severe pre-eclamptic pregnancies. Br J Obstet Gynaecol. 1994;101(8):669–674.
Thadhani R, Powe CE, Tjoa ML, Khankin E, Ye J, Ecker J, Schneyer A, Karumanchi SA. First trimester follistatin-like-3 levels in pregnancies complicated by subsequent gestational diabetes mellitus. Diabetes Care. 2010;33(3):664–669.
Miron P, Lambert J, Marcil A, Cowans NJ, Stamatopoulou A, Spencer K. Maternal plasma levels of follistatin-related gene protein in the first trimester of pregnancies with Down syndrome. Prenat Diagn. 2010;30(3):224–228.
Pryor-Koishi K, Nishizawa H, Kato T, et al. Overproduction of the follistatin-related gene protein in the placenta and maternal serum of women with pre-eclampsia. Br J Obstet Gynaecol. 2007;114(9):1128–1137.
Guo J, Tian T, Lu D, Xia G, Wang H, Dong M. Alterations of maternal serum and placental follistatin-like 3 and myostatin in pre-eclampsia. J Obstet Gynaecol Res. 2012;38(7):988–996.
Yamamoto S, Douchi T, Yoshimitsu N, Nakae M, Nagata Y. Waist to hip circumference ratio as a significant predictor of preeclampsia, irrespective of overall adiposity. J Obstet Gynaecol Res. 2001;27(1):27–31.
Founds SA, Powers RW, Patrick TE, et al. A comparison of circulating TNF-alpha in obese and lean women with and without preeclampsia. Hypertens Pregnancy. 2008;27(1):39–48.
Roberts JM, Pearson G, Cutler J, Lindheimer M. NHLBI working group. Summary of the NHLBI working group on research on hypertension during pregnancy. Hypertension. 2003;41(3):437–445.
Lukaski HC, Siders WA, Nielsen EJ, Hall CB. Total body water in pregnancy: assessment by using bioelectrical impedance. Am J Clin Nutr. 1994;59(3):578–585.
van Raaij JM, Peek ME, Vermaat-Miedema SH, Schonk CM, Hautvast JG. New equations for estimating body fat mass in pregnancy from body density or total body water. Am J Clin Nutr. 1988;48(1):24–29.
McCarthy EA, Strauss BJ, Walker SP, Permezel M. Determination of maternal body composition in pregnancy and its relevance to perinatal outcomes. Obstet Gynecol Surv. 2004;59(10):731–742.
Bergman RN, Stefanovski D, Buchanan TA, et al. A better index of body adiposity. Obesity (Silver Spring). 2011;19(5):1083–1089.
Hayette S, Gadoux M, Martel S, et al. FLRG (follistatin-related gene), a new target of chromosomal rearrangement in malignant blood disorders. Oncogene. 1998;16(22):2949–2954.
Biron-Shental T, Schaiff WT, Rimon E, Shim TL, Nelson DM, Sadovsky Y. Hypoxia enhances the expression of follistatin-like 3 in term human trophoblasts. Placenta. 2008;29(1):51–57.
Roberts J, Taylor RN, Musci TJ, Rodgers GM, Hubel CA, McLaughlin MK. Preeclampsia: an endothelial cell disorder. Am J Obstet Gynecol. 1989;161(5):1200–1204.
Roberts JM, Hubel CA. Oxidative stress in preeclampsia. Am J Obstet Gynecol. 2004;190(5):1177–1178.
Myatt L, Clifton RG, Roberts JM, et al. First-trimester prediction of preeclampsia in nulliparous women at low risk. Obstet Gynecol. 2012;119(6):1234–1242.
Eskenazi B, Fenster L, Sidney S. A multivariate analysis of risk factors for preeclampsia. JAMA. 1991;266(2):237–241.
Kaaja R, Laivuori H, Laakso M, Tikkanen MJ, Ylikorkala O. Evidence of a state of increased insulin resistance in preeclampsia. Metabolism. 1999;48(7):892–896.
Bartholin L, Maguer-Satta V, Hayette S, et al. Transcription activation of FLRG and follistatin by activin A, through Smad proteins, participates in a negative feedback loop to modulate activin A function. Oncogene. 2002;21(14):2227–2235.
Ogawa K, Funaba M, Chen Y, Tsujimoto M. Activin A functions as a Th2 cytokine in the promotion of the alternative activation of macrophages. J Immunol. 2006;177(10):6787–6794.
Hu D, Tian T, Guo J, Wang H, Chen D, Dong M. Decreased maternal and placental concentrations of follistatin-like 3 in gestational diabetes. Clin Chim Acta. 2012;413(5–6):533–536.
Samuel VT, Petersen KF, Shulman GI. Lipid-induced insulin resistance: unravelling the mechanism. Lancet. 2010;375(9733):2267–2277. doi: 10.1016/S0140-6736(10)60408-4.
Catalano PM. Obesity, insulin resistance, and pregnancy outcome. Reproduction. 2010;140(3):365-371. doi: 10.1530/REP-10-0088.
Butte NF. Carbohydrate and lipid metabolism in pregnancy: normal compared with gestational diabetes mellitus. Am J Clin Nutr. 2000;71(5 suppl):1256S-1261S.
Catalano PM, Roman-Drago NM, Amini SB, Sims EA. Longitudinal changes in body composition and energy balance in lean women with normal and abnormal glucose tolerance during pregnancy. Am J Obstet Gynecol. 1998;179(1):156–165.
Homko CJ, Sivan E, Reece EA, Boden G. Fuel metabolism during pregnancy. Semin Reprod Endocrinol. 1999;17(2):119–125.
Oshima Y, Ouchi N, Shimano M, et al. Activin A and follistatinlike 3 determine the susceptibility of heart to ischemic injury. Circulation. 2009;120(16):1606–1615. doi: 10.1161/CIRCULATIONAHA.109.872200.
Levario-Carrillo M, Avitia M, Tufiño-Olivares E, Trevizo E, Corral-Terrazas M, Reza-López S. Body composition of patients with hypertensive complications during pregnancy. Hypertens Pregnancy. 2006;25(3):259–269.
da Silva EG, Carvalhaes MA, Hirakawa HS, da Silva EG, Peraçoli JC. Bioimpedance in pregnant women with preeclampsia. Hypertens Pregnancy. 2010;29(4):357–365.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Founds, S.A., Ren, D., Roberts, J.M. et al. Follistatin-Like 3 Across Gestation in Preeclampsia and Uncomplicated Pregnancies Among Lean and Obese Women. Reprod. Sci. 22, 402–409 (2015). https://doi.org/10.1177/1933719114529372
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719114529372