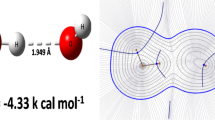
Competitive kinetic experiments were performed for the reactions of H atoms with HCOOH and DCOOD in aqueous solution. Excellent Arrhenius behavior is represented by log(k/M-1 s-1) = (11.6 ± 0.2) - (34.0 ± 1.1) kJ mol-1/2.303RT ) for HCOOH, log(k/M-1 s-1) = (11.8 ± 0.2) - (39.8 ± 1.2) kJ mol-1/2.303RT ) for DCOOD. Kinetic isotope effects are discussed in terms of transition state theory, supported by ab initio calculations, and in comparison with previous results for the abstraction from the formate ion.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lossack, A.M., Bartels, D.M. & ‡, E.R.1. Rate constants and kinetic isotope effects in hydrogen abstractions by H from formic acid. Res Chem Intermediat 27, 475–483 (2001). https://doi.org/10.1163/156856701104202129
Published:
Issue Date:
DOI: https://doi.org/10.1163/156856701104202129