Abstract
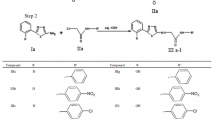
A series of novel metronidazole derivatives were designed via introduction of pharmacologically active 1,3,4-thiadiazole and Schiff base fragments into the 5-nitroimidazole skeleton of metronidazole at the 2-position without modifying the nitro group. The designed compounds were synthesized as per literature method with little modification and were isolated in moderate to good yield (68–92%) in the analytically pure form without resorting to chromatographic purification. The antibacterial activity of the newly synthesized compounds was assessed against three gram-positive bacterial strains (S. aureus, B. cereus, and S. epidermidis) and three gram-negative strains (E. coli, S. typhi, and K. pneumoniae) by standard disc diffusion method. The synthesized compounds were also tested for anthelmintic activity at a concentration of 2 mg/mL against two worm species, Pheretima posthuma and Perionyx excavatus. Three of the twelve new derivatives showed comparable antibacterial activity to that of standard but none of them exhibited anthelmintic potential.


Similar content being viewed by others
REFERENCES
Ceruelos, A.H., Romero-Quezada, L.C., Ledezma, J.C.R., and Contreras, L.L., Eur. Rev. Med. Pharmacol. Sci., 2019, vol. 23, p. 397. https://doi.org/10.26355/eurrev_201901_16788
Congiu, C., Cocco, M.T., and Onnis, V., Bioorg Med Chem Lett., 2008, vol. 18, p. 989. https://doi.org/10.1016/j.bmcl.2007.12.023
Yurttaş, L., Ertaş, M., Çiftçi, G.A., Temel, H.E., and Demirayak, Ş., Acta. Pharm. Sci., 2017, vol. 55, p. 39. https://doi.org/10.23893/1307-2080.APS.0553
Lin, Y.I., Peterson, P.J., Yang, Y., Weiss, W.J., and Shales, D.M., Bioorg. Med. Chem., 2008 vol. 16, p. 1890. https://doi.org/10.1016/j.bmc.2007.11.006
Nakamura, T., Kakinuma, H., Umemiya, H., Amada, H., and Miyata, N., Bioorg. Med. Chem. Lett., 2004, vol. 14, p. 333. https://doi.org/10.1016/j.bmcl.2003.11.005
Han, M.S. and Kim, D.H., Bioorg. Med. Chem. Lett., 2001, vol. 11, p. 1425. https://doi.org/10.1016/s0960-894x(01)00226-8
Roman, G., Riley, J.G., Vlahakis, J.Z., Kinobe, R.T., Brien, J.F., Nakatsu, K., and Szarek, W.A., Bioorg. Med. Chem., 2007, vol. 15, p. 3225. https://doi.org/10.1016/j.bmc.2007.02.034
Babizhayev, M.A., Life. Sci., 2006, vol. 78, p. 2343. https://doi.org/10.1016/j.lfs.2005.09.054
Nantermet, P.G., Barrow, J.C., Lindsley, S.R., Young, M.B., Mao, Sh.-Sh., Carroll, S., Bailey, C., Bosserman, M., Colussi, D., McMasters, D.R., Vacca, J.P., and Selnick, H.G., Bioorg. Med. Chem. Lett., 2004, vol. 14, p. 2141. https://doi.org/10.1016/j.bmcl.2004.02.033
Ganguly, S., Vithlani, V., Kesharwani, A., Kuhu, R., Baskar, L., Mitramazumder, P., Sharon, A., and Dev, A. Acta Pharm., 2011, vol. 61, p. 187. https://doi.org/10.2478/v10007-011-0018-2
Husain, A., Drabu, S., and Kumar, N., Acta. Pol. Pharm., 2009, vol. 66, p. 243. https://ptfarm.pl/pub/File/Acta_Poloniae/2009/3/243.pdf
Mallemula, V.R., Sanghai, N.N., Himabindu, V., and Chakravarthy, A.K., Res. Chem. Intermed., 2013, vol. 41, p. 2125. https://doi.org/10.1007/s11164-013-1335-5
Bhinge, S.D., Chature, V., and Sonawane, L.V., Pharm. Chem. J., 2015, vol. 49, p. 367. https://doi.org/10.1007/s11094-015-1287-8
Munguía, B., Mendina, P., Espinosa, R., Lanz, A., Saldaña, J., Andina, M.J., Ures, X., López, A., Manta, E., and Domínguez, L., Lett. Drug Des. Discovery, 2013, vol. 10, p. 1007. https://doi.org/10.2174/15701808113109990028
Sadek, B. and Fahelelbom, K.M.S., Molecules, 2011, vol. 16, p. 4339. doi.org/10.3390/molecules16064339
Varshney, M.M., Husain, A., and Parcha, V., Med. Chem. Res., 2014, vol. 23, p. 4034. https://doi.org/10.1007/s00044-014-0982-4
Husain, A., Varshney, M.M., Parcha, V., Ahmad, A., and Khan, S.A., Lett. Drug Des. Discovery, 2018, vol. 15, p. 103. https://doi.org/10.2174/1570180814666170710160751
Balaji, P.N., Ranganayakulu, D., and Subba Reddy, G.V., Asian J. Pharm. Pharmacol., 2017, vol. 3, p. 9. http://www.ajpp.in/uploaded/p48.pdf
Rao, R., Reddy, K.R., and Mahendra, K.N., Bulg. Chem. Commun., 2014, vol. 46, p. 11. http://bcc.bas.bg/BCC_Volumes/Volume_46_Number_1_2014/2.pdf
Baluja, S. and Chanda, S., Rev. Colomb. Cienc. Quím. Farm., 2016, vol. 45, p. 201–218. https://doi.org/10.15446/rcciquifa.v45n2.59936
Atia, A.J.Kh., Molecules, 2009, vol. 14, p. 2431. https://doi.org/10.3390/molecules14072431
Norris, J.F., Experimental Organic Chemistry, New York: McGraw-Hill, 1924, 2nd ed., p. 173. https://library.sciencemadness.org/library/books/norris_experimental_organic_chemistry.pdf
Sharba, A.H.K., Al-Bayati, R.H., Rezki, N., Aouad, M.R., Molecules, 2005, vol. 10, p. 1153. https://doi.org/10.3390/10091153
Sahoo, P.K., Sharma, R., Pattanayak, P., Med. Chem. Res., 2010, vol. 19, p. 127. https://doi.org/10.1007/s00044-009-9178-8
Husain, A., Varshney, M.M., Parcha, V., Ahmad, A., and Khan, S.A., Bangladesh J. Pharmacol., 2015, vol. 10, p. 555. https://doi.org/10.3329/bjp.v10i3.23381
Dahiya, R. and Pathak, D., Eur. J. Med. Chem., 2007, vol. 42, p. 772. https://doi.org/10.1016/j.ejmech.2006.11.015
ACKNOWLEDGMENTS
The authors are thankful to the Faculty of Pharmacy, Bhagwant University, Ajmer, and Sri Jaydeva College of Pharmaceutical Sciences, Naharkanta, Bhubaneswar, for providing the necessary support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Pattanayak, P., Saravanan, K. Synthesis and Biological Activity of Some Novel Metronidazole Derivatives Containing a 1,3,4-Thiadiazole Schiff Base Moiety. Russ J Org Chem 58, 99–105 (2022). https://doi.org/10.1134/S1070428022010146
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428022010146