Abstract
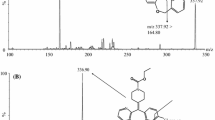
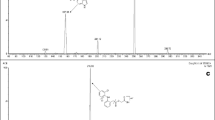
A simple, sensitive and high throughput liquid chromatography-tandem mass spectrometry method has been developed for the determination of lafutidine in human plasma. Sample clean-up involved solid phase extraction of lafutidine along with ranitidine as the internal standard from 100 μL of human plasma. The chromatographic separation is achieved within 2.5 min on a Grace Denali C18 (50 × 4.6 mm, 5 μ) column using 2 mM ammonium acetate, pH 3.0 adjusted with acetic acid and acetonitrile (20: 80, v/v) as the mobile phase. The precursor → product ion transitions for lafutidine (m/z 432.2 → 351.4) and IS (m/z 315.3 → 176.3) were monitored on a triple quadrupole mass spectrometer, operating in the multiple reaction monitoring and positive ion mode. The method is validated over a wide dynamic concentration range of 0.25–1000 ng/mL. The mean relative recovery for lafutidine across quality controls is 97.9%. The relative matrix effect between eight different plasma lots, expressed as coefficient of variation of the slopes of the calibration lines is 1.94. The method is applied to a bioequivalence study of 10 mg lafutidine tablet formulation in 26 healthy Indian male subjects under fasting condition. The reproducibility of study data is demonstrated by analysis of 93 incurred samples.
Similar content being viewed by others
References
Sato, H., Kawashima, K., Yuki, M., Kazumori, H., Azharul, M., Rumi, K., Ortega-Cava, C.F., Ishihara, S., and Kinoshita, Y., J. Lab. Clin. Med., 2003, vol. 141, p. 102.
Scarpignato, C., Drug Dis. Today: Ther. Strategies 2007, vol. 4, p. 155.
Ikawa, K., Shimatani, T., Hayato, S., Morikawa, N., and Tazuma, S., Biol. Pharm. Bull., 2007, vol. 30, p. 1003.
Onodera, S., Shibata, M., Tanaka, M., Inaba, N., Yamaura, T., and Ohnishi, H., Jpn. J. Pharmacol., 1995, vol. 68, p. 161.
Shibata, M., Yamaura, T., Inaba, N., Onodera, S., Chiba, Y., and Ohnishi, H., Eur. J. Pharmacol., 1993, vol. 235, p. 245.
Kato, S., Tanaka, A., Kunikata, T., Umeda, M., and Takeuchi, K., Digestion, 2000, vol. 61, p. 39.
Yamagishi, H., Koike, T., Ohara, S., Horii, T., Kikuchi, R., Kobayashi, S., Abe, Y., Iijima, K., Imatani, A., Suzuki, K., Hishinuma, T., Goto, J., and Shimosegawa, T., World J. Gastroenterol., 2008, vol. 14, p. 2406.
Inaba, N., Shibata, M., Onodera, S., Tanaka, M., Suzuki, T., Yamaura, T., and Ohnishi, H., Folia Pharmacol. Jpn., 1995, vol. 105, p. 231.
Inamori, M., Togawa, J., Iwasaki, T., Ozawa, Y., Kikuchi, T., Muramatsa, K., Chiguchi, G., Mastumoto, S., Kawamura, H., Abe, Y., Kirikoshi, H., Kobayashi, N., Shimamura, T., Kunota, K., Sakaguchi, T., Saito, S., Ueno, N., and Nakajima, A., J. Gastroenterol., 2005, vol. 40, 453.
Nakano, M., Kitano, S., Nanri, M., and Kiniwa, M., Eur. J. Pharmacol., 2011, vol. 658, p. 236.
Itoh, H., Naito, T., and Takeyama, M., Biol. Pharm. Bull., 2002, vol. 25, p. 379.
Wu, L., Zhang, Z., Tian, Y., Li, W., Xu, F., Chen, Y., and Wei, H., J. Mass Spectrom., 2005, vol. 40, p. 1637.
Chen, W.D., Liang, Y., Li, H., Xiong, Y., Liu, X.D., Wang, G.J., and Xie, L., J. Pharm. Biomed. Anal., 2006, vol. 41, p. 256.
Dewan, B. and Chimata, R., World J. Gastrointest. Pharmacol. Ther., 2010, vol. 1, p. 112.
Sun, X., Tian, Y., Zhang, Z., and Chen Y., J. Chromatogr. B, 2009, vol 877, p. 3953.
Guidance for Industry, Bionanlytical Method Validation, US Department of Health and Human Services, Food and Drug Administration Centre for Drug Evaluation and Research (CDER), Centre for Veterinary Medicine (CVM), May 2001.
Viswanathan, C.T., Bansal, S., Booth, B., DeStefano, A.J., Rose, M.J., Sailstad, J., Shah, V.P., Skelly, J.P., Swann, P.G., and Weiner R., AAPS J., 2007, vol. 9, p. E30.
King, R., Bonfiglio, R., Fernandez-Metzler, C., Miller-Stein, C., and Olah, T., J. Am. Soc. Mass Spectrom., 2000, vol. 11, p. 942.
Matuszewski, B.K., Constanzer, M.L., and Chavez-Eng, C.M., Anal. Chem., 2003, vol. 75, p. 3019.
Matuszewski, B.K., J. Chromatogr. B, 2006, vol. 830, p. 293.
Guidance for Industry: ICH E6 Good Clinical Practice, U.S. Department of Health and Human Services, Food and Drug Administration, Centre for Drug Evaluation and Research (CDER), Centre for Biologics Evaluation and Research (CBER), April 1996.
Yadav, M. and Shrivastav, P., Bioanalysis, 2011, vol. 3, p. 1007.
Author information
Authors and Affiliations
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Yadav, M., Trivedi, V., Upadhyay, V. et al. Sensitive, selective and rapid determination of lafutidine in human plasma by solid phase extraction-liquid chromatography-tandem mass spectrometry. J Anal Chem 69, 448–460 (2014). https://doi.org/10.1134/S1061934814050116
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934814050116