Abstract
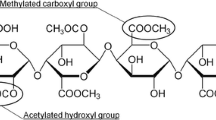
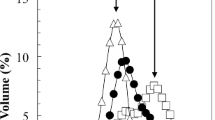
The effects of temperature on the intrinsic viscosity and on the conformation of pectin obtained from Citrus, Apple and Sunflower in a 0.17M NaCl solution were studied. Mark-Houwink plots for Orange, Apple and Sunflower pectin were obtained using HPSEC with online light scattering and viscosity detection. The intrinsic viscosity and flow activation energy E a of pectin from the sources studied were measured over the temperature range 20–60°C. E a values were 0.67, 0.69, 1.34, and 1.44 × 107 J/(kmol) for commercial Citrus, Orange, Sunflower and Apple pectin, respectively. Intrinsic viscosity decreased linearly with increasing temperature, for all pectins except Apple one. These results clearly indicated that Apple pectin underwent structural changes that were more drastic than those that occurred for pectin from the other sources. E a increased with decreasing weight average molar mass M w indicating that pectin with low M w were more asymmetric than pectin with higher values of M w. Changes in the Huggins coefficients K h with temperature for pectin from the various sources were attributed to the ability of pectin to aggregate, disaggregate and re-aggregate according to the temperature at which it was stored.
Similar content being viewed by others
References
D. Mohnen, Curr. Opin. Plant Biol. 11, 266 (2008).
D. G. Oakenfull, in The Chemistry and Technology of Pectin, Ed. by R. H. Walter (Acc., San Diego, 1991).
M. A. V. Axelos and J.-F. Thibault, in The Chemistry and Technology of Pectin, Ed. by R. H. Walter (Acc., San Diego, 1991).
M. P. Tombs and S. E. Harding, An Introduction to Polysaccharide Biotechnology (Taylor and Francis, London, 1998).
T. M. C. C. Filisetti-Cozzi and N. C. Carpita, Anal. Biochem. 197, 157 (1991).
C. P. Kelco, Control Methods. Determination of Pectin DE, 3 (2001).
M. L. Fishman, H. K. Chau, F. Kolpak, and J. J. Brady, Agric. Food Chem. 49, 4494 (2001).
Z. K. Muhidinov, M. L. Fishman, R. M. Gorshkova, et al., Kazakh. Chem. J. 21, 60 (2008).
L. E. Nielsen, Polymer Rheology (Marcel Dekker, New York, 1977).
R. H. Chen and W. C. Lin, J Fish. Soc. Taiwan 19, 299 (1992).
M. Rinaudo and A. Domard, in Chitin and Chitosan: Sources, Chemistry, Biochemistry, Physical Properties and Applications, Ed. by G. Skiak-Braek, T. Anthonsen, and P. Sandford (Elsevier Applied Science, London, 1989), p. 71.
B. Launay, J. L. Doublier, and G. Cuvelier, in Functional Properties of Food Macromolecules, Ed. by J. R. Mitchell and D. A. Ledward (Elsevier Applied Science, London, 1986), p. 1.
M. Bohdanecky and J. Kovar, Viscosity of Polymer Solutions (Elsevier, Amsterdam, 1982).
M. L. Fishman, H. K. Chau, P. D. Hoagland, and K. Ayyad, Carbohydr. Res. 323, 126 (2000).
M. L. Fishman, H. K. Chau, P. D. Hoagland, and A. T. Hotchkiss, Food Hydrocollloids 20, 1170 (2006).
M. L. Fishman, P. H. Cooke, H. K. Chau, et al., Biomacromolecules 8, 573 (2007).
M. L. Fishman, P. H. Cooke, B. Levaj, et al., Arch. Biochem. Biophys. 294, 253 (1992).
M. L. Fishman, P. H. Walker, H. K. Chau, and A. T. Hotchkiss, Biomacromolecules 4, 880 (2003).
M. L. Fishman, P. H. Cooke, A. T. Hotchkiss, and W. Damert, Carbohydr. Res. 248, 303 (1993).
G. A. Morris, T. J. Foster and S. E. Harding, Carbohydr. Polym. 48, 361 (2002).
M. A. V. Axelos and M. Branger, Food Hydrocolloids 7, 91(1993).
M. A. Rao, in Rheology of Fluid and Semisolid Foods: Principal and Application (Chapman and Hall Food Science Book, New York, 1999), p. 12.
R. Berger, Macromol. Chem. 102, 24 (1967).
W. Wang and D. Xu, Int. J. Biol. Macromol. 16, 149 (1994).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Muhidinov, Z.K., Fishman, M.L., Avloev, K.K. et al. Effect of temperature on the intrinsic viscosity and conformation of different pectins. Polym. Sci. Ser. A 52, 1257–1263 (2010). https://doi.org/10.1134/S0965545X10120035
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0965545X10120035