Abstract
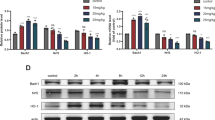
The endoplasmic reticulum (ER) stress inducers dithiothreitol (DTT) and sodium selenite (SS) were tested for effect on expression of ER selenoproteins and apoptosis markers in MCF7 breast adenocarcinoma cells. DTT used at 1 or 5 mM did not affect the survival of MCF7 cells. Based on the real-time PCR data and the protein expression levels of ER stress markers, ER stress was assumed to evolve along an adaptation pathway in MCF7 cells treated with 1 or 5 mM DTT, involving mainly the transcription factors IRE1 and ATF6 and the selenoproteins SELS, SELK, SELT, SELM, and SELN. Cell treatment with 0.01 μM SS decreases the mRNA levels of all genes examined. When the SS concentration was increased to 0.1 μM, an increase in expression was observed for key ER stress genes and apoptosis markers, including CHOP, GADD34, PUMA, BIM, ATF4, sXBP, uXBP, AKT1, BAX, and BAK. Higher SS concentrations were assumed to trigger the unfolded protein response (UPR) via a proapoptic signaling pathway involving PERK and an alternative IRE1 signaling pathway. Used at 1 μM, SS increased the mRNA levels of apoptosis markers, upregulated expression of a spliced form of XBP1, and substantially decreased the cell survival. SS (1 μM) was assumed to trigger apoptosis in MCF7 cells. The results indicate that both adaptive and proapoptic UPR signaling pathways are activated in cells, depending on the nature and concentration of the ER stress inducer.




Similar content being viewed by others
REFERENCES
Azamjah N., Soltan-Zadeh Y., Zayeri F. 2019. Global trend of breast cancer mortality rate: A 25-year study. Asian Pac. J. Cancer Prev. 20, 2015–2020.
Tang Y., Wang Y., Kiani M.F., Wang B. 2016. Classification, treatment strategy, and associated drug resistance in breast cancer. Clin. Breast Cancer. 16, 335–343.
Fisusi F.A., Akala E.O. 2019. Drug combinations in breast cancer therapy. Pharm. Nanotechnol. 7, 3–23.
Comşa Ş., Cîmpean A.M., Raica M. 2015. The story of MCF 7 breast cancer cell Line: 40 years of experience in research. Anticancer Res. 35, 3147–3154.
Hotamisligil G.S., Davis R.J. 2016. Cell signaling and stress responses. Cold Spring Harb. Perspect. Biol. 8, a006072.
Almanza A., Carlesso A., Chintha C., Creedican S., Doultsinos D., Leuzzi B., Luís A., McCarthy N., Montibeller L., More S., Papaioannou A., Püschel F., Sassano M. L., Skoko J., Agostinis P., et al. 2019. Endoplasmic reticulum stress signaling: From basic mechanisms to clinical applications. FEBS J. 286 (2), 241–278.
Lemmer I.L., Willemsen N., Hilal N., Bartelt A. 2021. A guide to understanding endoplasmic reticulum stress in metabolic disorders. Mol. Metab. 20, 101169.
Cui Z., Li C., Li X., Zhang Q., Zhang Y., Shao J., Zhou K. 2015. Sodium selenite (Na2SeO3) induces apoptosis through the mitochondrial pathway in CNE-2 nasopharyngeal carcinoma cells. Int. J. Oncol. 46 (6), 2506–2514.
Jiang Q., Li F., Shi K., Wu P., An J., Yang Y., Xu C. 2013. ATF4 activation by the p38MAPK–eIF4E axis mediates apoptosis and autophagy induced by selenite in Jurkat cells. FEBS Lett. 587 (15), 2420–2429.
Liu T., Sun Y., Yang S., Liang X. 2020. Inhibitory effect of selenium on esophagus cancer cells and the related mechanism. J. Nutr. Sci. Vitaminol. 66 (5), 456–461.
Bonfim N.E.S., Baranoski A., Mantovani M.S. 2020. Cytotoxicity of sodium selenite in HaCaT cells induces cell death and alters the mRNA expression of PUMA, ATR, and mTOR genes. J. Trace Elem. Med. Biol. 62, 126605.
Yang L., Cai Y.-S., Xu K., Zhu J.-L., Li Y.-B., Wu X.-Q., Sun J., Lu S.-M., Xu P. 2018. Sodium selenite induces apoptosis and inhibits autophagy in human synovial sarcoma cell line SW982 in vitro. Mol. Med. Rep. 17 (5), 6560–6568.
Goltyaev M.V., Varlamova E.G., Novoselov S.V., Fesenko E.E. 2020. Activation of signal pathways of apoptosis under conditions of prolonged ER-stress caused by exposure of mouse testicular teratoma cells to selenium-containing compounds. Dokl. Biochem. Biophys. 490, 9–11. https://doi.org/10.1134/S160767292001007X
Kuznetsova Yu.P., Goltyaev M.V., Gorbacheva O.S., Novoselov S.V., Varlamova E.G., Fesenko E.E. 2018. Influence of sodium selenite on the mRNA expression of the mammalian selenocysteine-containing protein genes in testicle and prostate cancer cells. Dokl. Biochem. Biophys. 480, 131–134. https://doi.org/10.1134/S1607672918030018
Takahashi K., Suzuki N., Ogra Y. 2017. Bioavailability comparison of nine bioselenocompounds in vitro and in vivo. Int. J. Mol. 18, 506.
Misra S., Boylan M., Selvam A., Spallholz J.E., Björnstedt M. 2015. Redox-active selenium compounds: From toxicity and cell death to cancer treatment. Nutrients. 7, 3536–3556.
Guan L., Han B., Li Z., Hua F., Huang F., Wei W., Yang Y., Xu C. 2009. Sodium selenite induces apoptosis by ROS-mediated endoplasmic reticulum stress and mitochondrial dysfunction in human acute promyelocytic leukemia NB4 cells. Apoptosis. 14, 218–225.
Okuno T., Honda E., Arakawa T., Ogino H., Ueno H. 2014. Glutathione-dependent cell cycle G1 arrest and apoptosis induction in human lung cancer A549 cells caused by methylseleninic acid: comparison with sodium selenite. Biol. Pharm. Bull. 37, 1831–1837.
Palsamy P., Bidasee K.R., Shinohara T. 2014. Selenite cataracts: Activation of endoplasmic reticulum stress and loss of Nrf2/Keap1-dependent stress protection. Biochim. Biophys. Acta. 1842 (9), 1794–1805.
Cui Z., Li C., Li X., Zhang Q., Zhang Y., Shao J., Zhou K. 2015. Sodium selenite (Na2SeO3) induces apoptosis through the mitochondrial pathway in CNE-2 nasopharyngeal carcinoma cells. Int. J. Oncol. 46 (6), 2506–2514.
Badr D.M., Hafez H.F., Agha A.M., Shouman S.A. 2016. The combination of α-tocopheryl succinate and sodium selenite on breast cancer: A merit or a demerit? Oxid. Med. Cell Longev. 2016, 4741694.
Tartier L., McCarey Y.L., Biaglow J.E., Kochevar I.E., Held K.D. 2000. Apoptosis induced by dithiothreitol in HL-60 cells shows early activation of caspase 3 and is independent of mitochondria. Cell Death Differ. 7 (10), 1002–1010.
Xiang X.Y., Yang X.C., Su J., Kang J.S., Wu Y., Xue Y.N., Dong Y.T., Sun L.K. 2016. Inhibition of autophagic flux by ROS promotes apoptosis during DTT-induced ER/oxidative stress in HeLa cells. Oncol. Rep. 35, 3471–3479.
Hughes D.J., Kunická T., Schomburg L., Liška V., Swan N., Souček P. 2018. Expression of selenoprotein genes and association with selenium status in colorectal adenoma and colorectal cancer. Nutrients. 10 (11), 1812.
Jia Y., Dai J., Zeng Z. 2020. Potential relationship between the selenoproteome and cancer. Mol. Clin. Oncol. 13 (6), 83.
Capone F., Polo A., Sorice A., Budillon A., Costantini S. 2020. Integrated analysis to study the relationship between tumor-associated selenoproteins: Focus on prostate cancer. Int. J. Mol. Sci. 21 (18), 6694.
Li J., Zhu Y., Zhou Y., Jiang H.G., Chen Z.H., Lu B.H., Shen X.N. 2020. The SELS rs34713741 polymorphism is associated with susceptibility to colorectal cancer and gastric cancer: a meta-analysis. Genet. Test. Mol. Biomarkers. 24 (12), 835–844.
Callejón-Leblic B., Arias-Borrego A., Rodríguez-Moro G., Roldán F.N., Pereira-Vega A., Gómez-Ariza J.L., García-Barrera T. 2021. Advances in lung cancer biomarkers: The role of (metal-) metabolites and selenoproteins. Adv. Clin. Chem. 100, 91–137.
Varlamova E.G. 2020. Protein–protein interactions of ER-resident selenoproteins with their physiological partners. Biochimie. 171–172, 197–204. https://doi.org/10.1016/j.biochi.2020.03.012
Rocca C., Pasqua T., Boukhzar L., Anouar Y., Angelone T. 2019. Progress in the emerging role of selenoproteins in cardiovascular disease: Focus on endoplasmic reticulum-resident selenoproteins. Cell Mol. Life Sci. 76 (20), 3969–3985.
Zhang C., Ge J., Liv M., Zhang Q., Talukder M., Li J.L. 2020. Selenium prevents cadmium-induced hepatotoxicity through modulation of endoplasmic reticulum-resident selenoproteins and attenuation of endoplasmic reticulum stress. Environ. Pollut. 260, 113873.
Pitts M.W., Hoffmann P.R. 2018. Endoplasmic reticulum-resident selenoproteins as regulators of calcium signaling and homeostasis. Cell Calcium. 70, 76–86.
Wang P., Lu Z., He M., Shi B., Lei X., Shan A. 2020. The effects of endoplasmic-reticulum-resident selenoproteins in a nonalcoholic fatty liver disease pig model induced by a high-fat diet. Nutrients. 12 (3), 692.
Varlamova E.G. 2018. Participation of selenoproteins localized in the ER in the processes occurring in this organelle and in the regulation of carcinogenesis-associated processes. J. Trace Elem. Med. Biol. 48, 172–180.
Varlamova E.G., Goltyaev M.V., Fesenko E.E. 2019. Protein partners of selenoprotein SELM and the role of selenium compounds in regulation of its expression in human cancer cells. Dokl. Biochem. Biophys. 488, 300–303.
Goltyaev M.V., Mal’tseva V.N., Varlamova E.G. 2020. Expression of ER-resident selenoproteins and activation of cancer cells apoptosis mechanisms under ER-stress conditions caused by methylseleninic acid. Gene. 755, 144884.
Kim C., Kim B. 2018. Anti-cancer natural products and their bioactive compounds inducing ER stress-mediated apoptosis: a review. Nutrients. 10 (8), 1021.
Harnoss J., Thomas A., Reichelt M., Guttman O., Wu T.D., Marsters S.A., Shemorry A., Lawrence D., Kan D., Segal E., Merchant M., Totpal K., Crocker L.M., Mesh K., Dohse M., et al. 2020. IRE1α disruption in triple-negative breast cancer cooperates with antiangiogenic therapy by reversing ER stress adaptation and remodeling the tumor microenvironment. Cancer Res. 80 (11), 2368–2379.
Zhang Y., Roh Y.J., Han S.J., Park I., Lee H.M., Ok Y.S., Lee B.C., Lee S.R. 2020. Role of selenoproteins in redox regulation of signaling and the antioxidant system: a review. Antioxidants (Basel). 9 (5), 383.
Du S., Zhou J., Jia Y., Huang K. 2010. SelK is a novel ER stress-regulated protein and protects HepG2 cells from ER stress agent-induced apoptosis. Arch. Biochem. Biophys. 502 (2), 137–143.
Fan R.F., Cao C.Y., Chen M.H., Shi Q.X., Xu S.W. 2018. Gga–let–7f–3p promotes apoptosis in selenium deficiency-induced skeletal muscle by targeting selenoprotein K. Metallomics. 10 (7), 941–952.
Addinsall A.B., Wright C.R., Andrikopoulos S., Poel C., Stupka N. 2018. Emerging roles of endoplasmic reticulum-resident selenoproteins in the regulation of cellular stress responses and the implications for metabolic disease. Biochem J. 475 (6), 1037–1057.
Marciel M.P., Hoffmann P.R. 2019. Molecular mechanisms by which selenoprotein K regulates immunity and cancer. Biol. Trace Elem. Res. 192 (1), 60–68.
Lee J.H., Park K.J., Jang J.K., Jeon Y.H., Ko K.Y., Kwon J.H., Lee S.R., Kim I.Y. 2015. Selenoprotein S-dependent selenoprotein K binding to p97(VCP) protein is essential for endoplasmic reticulum-associated degradation. J. Biol. Chem. 290 (50), 29941–29952.
Shchedrina V.A., Everley R.A., Zhang Y., Gygi S.P., Hatfield D.L., Gladyshev V.N. 2011. Selenoprotein K binds multiprotein complexes and is involved in the regulation of endoplasmic reticulum homeostasis. J. Biol. Chem. 286 (50), 42937–42948.
Huang J.Q., Ren F.Z., Jiang Y.Y., Lei X. 2016. Characterization of selenoprotein M and its response to selenium deficiency in chicken brain. Biol. Trace Elem. Res. 170 (2), 449–458.
Gong T., Hashimoto A.C., Sasuclark A.R., Khadka V.S., Gurary A., Pitts M.W. 2019. Selenoprotein M promotes hypothalamic leptin signaling and thioredoxin antioxidant activity. Antioxid. Redox Signal. https://doi.org/10.1089/ars.2018.7594
Jiang H., Shi Q.Q., Ge L.Y., Zhuang Q.F., Xue D., Xu H.Y., He X.Z. 2019. Selenoprotein M stimulates the proliferative and metastatic capacities of renal cell carcinoma through activating the PI3K/AKT/mTOR pathway. Cancer Med. 8 (10), 4836–4844.
Varone E., Pozzer D., Di Modica S., Chernorudskiy A., Nogara L., Baraldo M., Cinquanta M., Fumagalli S., Villar-Quiles R.N., De Simoni M.G., Blaauw B., Ferreiro A., Zito E. 2019. SELENON (SEPN1) protects skeletal muscle from saturated fatty acid-induced ER stress and insulin resistance. Redox Biol. 24, 101176.
AlRasheed M.M., AlAnzi A., AlShalhoub R., Abanmy N., Bakheet D. 2019. A study of the role of DIO1 and DIO2 polymorphism in thyroid cancer and drug response to therapy in the Saudi population. Saudi Pharm. J. 27 (6), 841–845.
Zevenbergen C., Groeneweg S., Swagemakers S.M.A., de Jong A., Medici-Van den Herik E., Rispens M., Klootwijk W., Medici M., de Rijke Y.B., Meima M.E., Larsen P.R., Chavatte L., Venter D., Peeters R.P., Van der Spek P.J., Visser W.E. 2019. Functional analysis of genetic variation in the SECIS element of thyroid hormone activating type 2 deiodinase. J. Clin. Endocrinol. Metab. 104 (5), 1369–1377.
Sovolyova N., Healy S., Samali A., Logue S.E. 2014. Stressed to death – mechanisms of ER stress-induced cell death. Biol. Chem. 395 (1), 1–13.
Hetz C., Zhang K., Kaufman R.J. 2020. Mechanisms, regulation and functions of the unfolded protein response. Nat. Rev. Mol. Cell. Biol. 21 (8), 421–438.
Sano R., Reed J.C. 2013. ER stress-induced cell death mechanisms. Biochim. Biophys. Acta. 1833 (12), 3460–3470.
Ma urel M., Chevet E., Tavernier J., Gerlo S. 2014. Getting RIDD of RNA: IRE1 in cell fate regulation. Trends Biochem. Sci. 39 (5), 245–254.
ACKNOWLEDGMENTS
We are grateful to the Department of Optical Microscopy and Spectrophotometry (Pushchino Biological Research Center, Russian Academy of Sciences, http://www.ckp-rf.ru/ckp/670266/).
Funding
This work was supported by State Contract no. 0191-2019-0019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflict of interest.
Statement on the welfare of humans or animals. This article does not contain any studies involving humans or animals performed by any of the authors.
Additional information
Translated by T. Tkacheva
Abbreviations: ASK1, apoptosis signal-regulating kinase 1; ATF4 and ATF6, activating transcription factors 4 and 6, respectively; Bak, Bax, and Bim, apoptosis inducers of the Bcl-2 family; CHOP, CCAAT enhancer-binding protein homologous protein; DIO2, iodothyronine deiodinase 2; IRE1, inositol-requiring enzyme 1; GADD34, growth arrest and DNA damage-inducible protein 34; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; PUMA, p53 upregulated modulator of apoptosis; SELK, SELN, SELS, SELM, SELT, SELI, and SEP15, selenoproteins K, N, S, M, T, I, and 15, respectively; UPR, unfolded protein response; XBP1, X-box binding protein 1; ROS, reactive oxygen species; DTT, dithiothreitol; SS, sodium selenite; REL, relative expression level; PCR, polymerase chain reaction; ER, endoplasmic reticulum.
Rights and permissions
About this article
Cite this article
Mal’tseva, V.N., Goltyaev, M.V., Novoselov, S.V. et al. Effects of Sodium Selenite and Dithiothreitol on Expression of Endoplasmic Reticulum Selenoproteins and Apoptosis Markers in MSF7 Breast Adenocarcinoma Cells. Mol Biol 56, 97–106 (2022). https://doi.org/10.1134/S0026893322010058
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893322010058