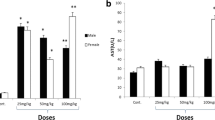
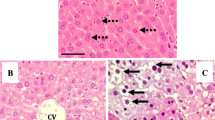
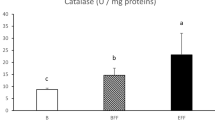
Abstract
The significant destructive changes in ultrastructure of hepatocytes from laboratory mice kept in different vivariums in Moscow and fed with dry laboratory animal diets acquired from different domestic manufacturers that were not standardized for initial products were demonstrated using electron microscopy. Furthermore, disruption in the ultrastructure of liver parenchymal cells occurred regardless of the animal status (SPF or conventional), conditions of various vivariums, as well as the feed manufacturer. At the same time, studies on ultrastructure of liver hepatocytes from mice kept in the Charles River Laboratory facilities in Germany and fed with the Altromin Spezialfutter laboratory animal diet (GmbH & Co., Germany) that was produced using quality control of ingredients did not reveal destructive changes in the internal ultrastructure of hepatocytes. However, if these mice were later fed with the food produced in local manufactures, changes in the structure of liver cells developed after 2 months. Thus, feeding with dry diet from the domestic producers of an unspecified composition causes significant changes in the ultrastructure of hepatocytes in control animals, reflecting the development of some pathological processes in the body.









Similar content being viewed by others
Abbreviations
- agEPR:
-
agranular EPR
- EPR:
-
endoplasmic reticulum
- grEPR:
-
granular EPR
- IVC:
-
individually ventilated cage
- NAFLD:
-
nonalcoholic fatty liver disease
- SPF status:
-
specific-pathogen-free animals
- VEC:
-
Complex of the Animal Facilities and the Research Unit Vivarium, Institute of Mitoengineering of Lomonosov Moscow State University
References
Dalton, A. J., Kahler, H., Striebich, M. J., and Lloyd, B. (1950) Finer structure of hepatic, intestinal and renal cells of the mouse as revealed by the electron microscope, J. Natl. Cancer Inst., 11, 439-461.
Fawcett, D. W. (1955) Observations on the cytology and electron microscopy of hepatic cells, J. Natl. Cancer Inst., 15, 1475-1503.
Bruni, C., and Porter, K. R. (1965) The fine structure of the parenchymal cell of the normal rat liver: I. General observations, Am. J. Pathol., 46, 691-755.
Jones, A. L., and Fawcett, D. W. (1966) Hypertrophy of the agranular endoplasmic reticulum in hamster liver induced by phenobarbital (with a review on the functions of this organelle in liver), J. Histochem. Cytochem., 14, 215-232.
Remmer, H., and Merker, H. J. (1965) Effect of drugs on the formation of smooth endoplasmic reticulum and drug-metabolizing enzymes, Ann. NY Acad. Sci., 123, 79-97.
Steiner, J. W., Carruthers, J. S., and Kalifat, S. R. (1962) Observations on the fine structure of rat liver cells in extrahepatic cholestasis, Z. Zellforsch. Mikrosk. Anat., 58, 141-159.
Wood, R. L. (1965) The fine structure of hepatic cells in chronic ethionine poisoning and during recovery, Am. J. Pathol., 46, 307-330.
Arhelger, R. B., Broom, J. S., and Boler, R. K. (1965) Ultrastructural hepatic alterations following tannic acid administration to rabbits, Am. J. Pathol., 46, 409-434.
Wieczorek, A., Stępień, P. M., Zarębska-Michaluk, D., Kryczka, W., Pabjan, P., and Król, T. (2017) Megamitochondria formation in hepatocytes of patient with chronic hepatitis C – a case report, Clin. Exp. Hepatol., 3, 169-175.
Falcón, V., Acosta-Rivero, N., Chinea, G., Gavilondo, J., de la Rosa, M. C., Menéndez, I., Dueñas-Carrera, S., Viña, A., García, W., Gra, B., Noa, M., Reytor, E., Barceló, M. T., Alvarez, F., and Morales-Grillo, J. (2003) Ultrastructural evidences of HCV infection in hepatocytes of chronically HCV-infected patients, Biochem. Biophys. Res. Commun., 305, 1085-1090.
Zhang, X. Q., Xu, C. F., Yu, C. H., Chen, W. X., and Li, Y. M. (2014) Role of endoplasmic reticulum stress in the pathogenesis of nonalcoholic fatty liver disease, World J. Gastroenterol., 20, 1768-1776.
Cali, A. M., Zern, T. L., Taksali, S. E., de Oliveira, A. M., Dufour, S., Otvos, J. D., and Caprio, S. (2007) Intrahepatic fat accumulation and alterations in lipoprotein composition in obese adolescents: a perfect proatherogenic state, Diabetes Care, 30, 3093-3098.
Fon Tacer, K., and Rozman, D. (2011) Nonalcoholic fatty liver disease: focus on lipoprotein and lipid deregulation, J. Lipids, 2011, 783976.
Dallak, M. A., Bin-Jaliah, I., Albawardi, A., Haidara, M. A., Sakr, H. F., Eid, R. A., Hassan, W. N., and Al-Ani, B. (2018) Swim exercise training ameliorates hepatocyte ultrastructural alterations in rats fed on a high fat and sugar diet, Ultrastruct. Pathol., 42, 155-161.
Abo El-Khair, S. M., Ghoneim, F. M., Shabaan, D. A., and Elsamanoudy, A. Z. (2020) Molecular and ultrastructure study of endoplasmic reticulum stress in hepatic steatosis: role of hepatocyte nuclear factor 4α and inflammatory mediators, Histochem. Cell Biol., 153, 49-62.
Silva-Veiga, F. M., Rachid, T. L., de Oliveira, L., Graus-Nunes, F., Mandarim-de-Lacerda, C. A., and Souza-Mello, V. (2018) GW0742 (PPAR-beta agonist) attenuates hepatic endoplasmic reticulum stress by improving hepatic energy metabolism in high-fat diet fed mice, Mol. Cell Endocrinol., 474, 227-237.
GOST R 50258-92 (1992) Combined Full-Rationed Feeds for Laboratory Animals, Technical Conditions, Standard Izd. (in Russian).
Reynolds, E. S. (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy, J. Cell Biol., 17, 208-212.
Silva, G. H., Hessel, G., Coelho, K. I., and Escanhoela, C. A. (2011) Steatosis of indeterminate cause in a pediatric group: is it a primary mitochondrial hepatopathy? Sao Paulo Med. J., 129, 217-223.
Aragno, M., Tomasinelli, C. E., Vercellinatto, I., Catalano, M. G., Collino, M., Fantozzi, R., Danni, O., and Boccuzzi, G. (2009) SREBP-1c in nonalcoholic fatty liver disease induced by Western-type high-fat diet plus fructose in rats, Free Radic. Biol. Med., 47, 1067-1074.
Meli, R., MattaceRaso, G., Irace, C., Simeoli, R., Di Pascale, A., Paciello, O., Pagano, T. B., Calignano, A., Colonna, A., and Santamaria, R. (2013) High fat diet induces liver steatosis and early dysregulation of iron metabolism in rats, PLoS One, 8, e66570.
Pevzner, M. I. (1958) Principles of Dietotherapy (Acharkyan, A. I., and Marshak, M. S., eds.) Medgiz, Moscow (in Russian).
Sihali-Beloui, O., Aroune, D., Benazouz, F., Hadji, A., El-Aoufi, S., and Marco, S. A. (2019) A hypercaloric diet induces hepatic oxidative stress, infiltration of lymphocytes, and mitochondrial reshuffle in Psammomys obesus, a murine model of insulin resistance, C R Biol., 342, 209-219, doi: https://doi.org/10.1016/j.crvi.2019.04.003 .
Funding
This work was supported by the Russian Foundation for Basic Research (project no. 19-04-00578) and performed with support of TSITIS, Russia (project no. AAAA-A19-119012490166-2).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflicts of interests. All applicable international, national and/or institutional principles for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Vays, V.B., Vangeli, I.M., Averina, O.A. et al. Ultrastructure of Hepatocytes from Laboratory Mice Fed a Standard Dry Laboratory Animal Diet. Biochemistry Moscow 85, 1082–1092 (2020). https://doi.org/10.1134/S0006297920090084
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297920090084