Abstract
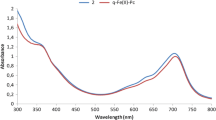
Suspended droplet three-phase liquid microextraction is a relatively new sample preparation technique for analysis of drugs. In the current study, tramadol hydrochloride was determined using tetrathiocyanato cobalt(II), (NH4)2[Co(SCN)4], as the complexing agent. The maximum of absorption for the formed ternary complex between tramadol hydrochloride and tetrathiocyanato cobalt(II) was observed at 625 nm. In this method, the target compound was extracted from the aqueous phase (5 mL containing 5 μg/mL of sample, pH 11) into an organic phase and then back-extracted into a directly suspended droplet as acceptor phase (3 μL of 1.74 × 10–9 M tetrathiocyanato cobalt(II), pH 2.8). 1-Octanol was used as organic phase. Under the optimized conditions: extraction time 90 s, back-extraction time 8 min and stirring rate 750 rpm, the linear range, the relative standard deviation (RSD, n = 5) and the limit of detection for tramadol hydrochloride were found 0.5–8 μg/mL, 4.9% and 8 ng/mL (n = 5), respectively. Also, the tap water and surface water samples were successfully analyzed using the proposed method and the relative recoveries obtained from the spiked water samples were 98.2 and 97.6%, respectively.
Similar content being viewed by others
References
Frankus, E., Friderichs, E., Kim, S.M., and Osterloh, G., Arzneim. Forsch., 1978, vol. 28, p. 114.
Aysel, K. and Yucel, K., II Farmaco, 2005, vol. 60, nos. 2, p. 163.
Kartinasari, W.F., Palupi, T., and Indrayanto, G., J. Liq. Chromatogr. Relat. Technol., 2005, vol. 27, nos. 4, p. 737.
Zaghloul, I.Y. and Radwan, M.A., J. Liq. Chromatogr. Relat. Technol., 1997, vol. 20, nos. 5, p. 779.
Zhong, Y.L., Yaowu Fenxi Zazhi, 1997, vol. 17, p. 279.
Krzek, J. and Stare, M., Biomed. Chromatogr., 2004, vol. 18, nos. 8, p. 589.
Pospisilova, M., Polasek, M., and Jokl, V., J. Pharm. Biomed. Anal., 1998, vol. 18, nos. 4–5, p. 777.
Ganjali, M.R., Memari, Z., Faridbod, F., Dinarvand, R., and Norouzi, P., Electroanalysis, 2008, vol. 20, nos. 24, p. 2663.
Wu, Y.J., Zhang, J.H., Zhao, K.L., and Zhang, Z.P., Fenxi Shiyanshi, 2005, vol. 24, p. 49.
Huang, C.L., Xiu, R., Liu, Z.Y., Li, Y.C., and Meng, C.J., Fenxi Kexue Xuebao, 2005, vol. 21, p. 48.
Huang, C.L., Xiu, R., Dong, Q.X., Chen, X.Z., and You, H.Q., Fenxi Ceshi Xuebao, 2005, vol. 24, p. 35.
Wang, M., Long, Y.L., Li, D.H., Zhou, W.B., Zhao, G.Z., and Zhang, Z., Fenxi Huaxue Fenxi, 1997, vol. 25, p. 448.
Hopkala, H., Misztal, G., and Wieczorek, A., Die Pharmazie, 1998, vol. 53, nos. 12, p. 869.
Yue, J.L., Yu, Q.H., Li, D.H., Zhao, G.Z., Shi, W., and An, L.R., Fenxi Huaxue, 2000, vol. 28, p. 84.
Puig, D. and Barcelo, D., TrAC, Trends Anal. Chem., 1996, vol. 15, nos. 8, p. 362.
Patsias, J. and Papadopoulou-Mourkidou, E., J. Chromatogr. A, 2000, vol. 904, nos. 2, p. 171.
Sarafras-Yazdia, A., Mofazzeli, F., and Es’haghi, Z., J. Chromatogr. A, 2009, vol. 1216, nos. 26, p. 5086.
Masrournia, M., Eshaghi, Z., and Mostafa, A., Am. Anal. Chem., 2012, vol. 2, nos. 2, p. 235.
Sarafraz-Yazdi, A., Mofazzeli, F., and Es’haghi, Z., Chromatographia, 2008, vol. 67, nos. 1, p. 49.
Krylov, V.A., Krylov, A.V., and Mosyagin, P.V., J. Anal. Chem., 2011, vol. 66, nos. 4, p. 331.
Liu, J., Jiang, G., Chi, Y., Cai, Y., Zhou, Q., and Hu, J., Anal. Chem., 2003, vol. 75, nos. 21, p. 5870.
Ayad, M.M., Khalil, H.M., El-Henawee, M., and Hosny, M., Sci. Pharm., 1991, vol. 67, p. 241.
Zhao, L., Zhu, L., and Lee, H.K., J. Chromatogr. A, 2002, vol. 963, nos. 1–2, p. 335.
Hou, L. and Lee, H.K., Anal. Chem., 2003, vol. 75, nos. 11, p. 2784.
Sarafraz-Yazdi, A. and Es’haghi, Z., J. Chromatogr. A, 2005, vol. 1082, nos. 2, p. 136.
Jonsson, J.A. and Mathiasson, L., J. Sep. Sci., 2001, vol. 24, nos. 7, p. 495.
Abdellatef, H.E., Spectrochim. Acta, Part A, 2007, vol. 66, nos. 4–5, p. 1248.
Ayad, M.M., Shalaby, A.A., Abdellatef, H.E., and Hosny, M.M., J. Pharm. Biomed. Anal., 2002, vol. 28, nos. 2, p. 311.
El-Walily, A.M., Belal, S.F., and Bakry, R.S., J. Pharm. Biomed. Anal., 1996, vol. 14, nos. 5, p. 561.
Saram, A., Mervat, M.H., Hisham, E., and Abdellatef, L.M.N., Chem. Ind. Chem. Eng., 2011, vol. 17, p. 269.
Lewis, R.J., Sax’s Dangerous Properties of Industrial Materials, New York: Van Nostrand Reinhold, 1992. 8th ed.
Liu, H. and Dasgupta, P.K., Anal. Chem., 1996, vol. 68, nos. 11, p. 1817.
Howard, P.H. and Meylan, W.M., Handbook of Physical Properties of Organic Chemicals, Boca Raton: CRC Press, 1997.
Jeannot, M.A. and Cantwell, F.F., Anal. Chem., 1997, vol. 69, nos. 2, p. 235.
Boyd-Boland, A.A. and Pawliszyn, J.B., J. Chromatogr. A, 1995, vol. 704, nos. 1, p. 163.
Lord, H.L. and Pawliszyn, J., Anal. Chem., 1997, vol. 69, nos. 19, p. 3899.
Eshaghi, Z. and Azmoodeh, R., Arab. J. Chem., 2010, vol. 3, nos. 1, p. 21.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kazemi, M.S. Determination of tramadol hydrochloride in water samples by directly suspended droplet three-phase liquid phase microextraction with tetrathiocyanato cobalt(II) as the complexing agent using UV-spectroscopy. J Anal Chem 70, 1330–1335 (2015). https://doi.org/10.1134/S1061934815110064
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934815110064