Abstract
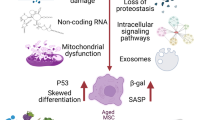
The aging phenomenon involves complex interrelated mechanisms at different levels of physiological processes, resulting in a decreased ability to maintain homeostasis of the organism due to organ dysfunctions. The chronic inflammation, which is progressing at the tissue level, operates as a driver of many age-related diseases. Nowadays, researchers consider cell aging (senescence) as one of the aging key hallmarks, manifested as qualitative and quantitative changes in the cellular composition and intercellular communication in tissues. This review highlights the modern concepts of aging at the cellular level. Particular attention is given to the mesenchymal stromal/stem cells (MSCs), which are involved in tissue homeostasis maintenance. Age-related MSC function alterations in tissue niches including immunomodulatory activity, hematopoiesis, and paracrine regulation are discussed. In addition, the approaches to MSC modification in vitro to attenuate the negative effects of aging are considered.
Similar content being viewed by others
REFERENCES
McHugh, D. and Gil, J., Senescence and aging: causes, consequences, and therapeutic avenues, J. Cell Biol., 2018, vol. 217, no. 1, p. 65.
Zhang, R., Chen, H.Z., and Liu, D.P., The four layers of aging, Cell Syst., 2015, vol. 1, no. 3, p. 180.
Moskalev, A.A., Proshkina, E.N., Belyi, A.A., and Solovyev, I.A., Genetics of aging and longevity, Russ. J. Genet.: Appl. Res., 2017, vol. 7, no. 4, p. 369.
López-Otín, C., Blasco, M.A., Partridge, L., et al., The hallmarks of aging, Cell, 2013, vol. 153, no. 6, p. 1194.
Muñoz-Espín, D., Cañamero, M., Maraver, A., et al., Programmed cell senescence during mammalian embryonic development, Cell, 2013, vol. 155, no. 5, p. 1104.
Muñoz-Espín, D. and Serrano, M., Cellular senescence: from physiology to pathology, Nat. Rev. Mol. Cell Biol., 2014, vol. 15, no. 7, p. 482.
van Deursen, J.M., The role of senescent cells in ageing, Nature, 2014, vol. 509, no. 7501, p. 439.
Farr, J.N., Xu, M., Weivoda, M.M., et al., Targeting cellular senescence prevents age-related bone loss in mice, Nat. Med., 2017, vol. 23, no. 9, p. 1072.
Gnani, D., Crippa, S., Della Volpe, L., et al., An early senescence state in aged mesenchymal stromal cells contributes to hematopoietic stem and progenitor cell clonogenic impairment through the activation of a proinflammatory program, Aging Cell, 2019, vol. 18, p. e12933.
Patil, P., Dong, Q., Wang, D., et al., Systemic clearance of p16INK4α-positive senescent cells mitigates age-associated intervertebral disc degeneration, Aging Cell, 2019, vol. 18, p. e12927.
Lewis McDougall, F.C., Ruchaya, P.J., Domenjo Vila, E., et al., Aged-senescent cells contribute to impaired heart regeneration, Aging Cell, 2019, vol. 18, p. e12931.
Hayflick, L. and Moorhead, P.S., The serial cultivation of human diploid cell strains, Exp. Cell Res., 1961, vol. 25, p. 585.
de Magalhães, J.P. and Passos, J.F., Stress, cell senescence and organismal ageing, Mech. Ageing Dev., 2018, vol. 170, p. 2.
Nakamura, A.J., Chiang, Y.J., Hathcock, K.S., et al., Both telomeric and non-telomeric DNA damage are determinants of mammalian cellular senescence, Epigenet. Chromatin, 2008, vol. 1, no. 1, p. 6.
Coppé, J.P., Patil, C.K., Rodier, F., et al., Senescence-associated secretory phenotypes reveal cell non-autonomous functions of oncogenic RAS and the p53 tumor suppressor, PLoS Biol., 2008, vol. 6, p. 2853.
Pole, A., Dimri, M., and Dimri, G.P., Oxidative stress, cellular senescence and ageing, AIMS Mol. Sci., 2016, vol. 3, no. 3, p. 300.
Sedelnikova, O.A., Redon, C.E., Dickey, J.S., et al., Role of oxidatively induced DNA lesions in human pathogenesis, Mutat. Res., 2010, vol. 704, p. 152.
von Zglinicki, T., Oxidative stress shortens telomeres, Trends Biochem. Sci., 2002, vol. 27, p. 339.
Campisi, J., Aging, cellular senescence, and cancer, Ann. Rev. Physiol., 2013, vol. 75, p. 685.
Ragnauth, C.D., Warren, D.T., Liu, Y., et al., Prelamin A acts to accelerate smooth muscle cell senescence and is a novel biomarker of human vascular aging, Circulation, 2010, vol. 121, p. 2200.
Scaffidi, P. and Misteli, T., Lamin A-dependent nuclear defects in human aging, Science, 2006, vol. 312, p. 1059.
Cao, K., Blair, C.D., Faddah, D.A., et al., Progerin and telomere dysfunction collaborate to trigger cellular senescence in normal human fibroblasts, J. Clin. Invest., 2011, vol. 121, no. 7, p. 2833.
Mitchell, S.J., Madrigal-Matute, J., Scheibye-Knudsen, M., et al., Effects of sex, strain, and energy intake on hallmarks of aging in mice, Cell Metab., 2016, vol. 23, no. 6, p. 1093.
Saxton, R.A. and Sabatini, D.M., mTOR signaling in growth, metabolism, and disease, Cell, 2017, vol. 168, no. 6, p. 960.
Herranz, N., Gallage, S., Mellone, M., et al., mTOR regulates MAPKAPK2 translation to control the senescence-associated secretory phenotype, Nat. Cell Biol., 2015, vol. 17, no. 9, p. 1205.
Laberge, R.-M., Sun, Y., Orjalo, A.V., et al., MTOR regulates the pro-tumorigenic senescence-associated secretory phenotype by promoting IL1A translation, Nat. Cell Biol., 2015, vol. 17, no. 8, p. 1049.
García-Prat, L., Martínez-Vicente, M., Perdiguero, E., et al., Autophagy maintains stemness by preventing senescence, Nature, 2016, vol. 529, no. 7584, p. 37.
Campisi, J. and d’Adda di Fagagna, F., Cellular senescence: when bad things happen to good cells, Nat. Rev. Mol. Cell Biol., 2007, vol. 8, no. 9, p. 729.
Salama, R., Sadaie, M., Hoare, M., and Narita, M., Cellular senescence and its effector programs, Genes Dev., 2014, vol. 28, no. 2, p. 99.
Imai, Y., Takahashi, A., Hanyu, A., et al., Crosstalk between the Rb pathway and AKT signaling forms a quiescence-senescence switch, Cell Rep., 2014, vol. 7, no. 1, p. 194.
Dimri, G.P., Lee, X., Basile, G., et al., A biomarker that identifies senescent human cells in culture and in aging skin in vivo, Proc. Natl. Acad. Sci. U.S.A., 1995, vol. 92, no. 20, p. 9363.
Watanabe, S., Kawamoto, S., Ohtani, N., and Hara, E., Impact of senescence-associated secretory phenotype and its potential as a therapeutic target for senescence-associated diseases, Cancer Sci., 2017, vol. 108, no. 4, p. 563.
Firsanov, D.V., Solovjeva, L.V., and Svetlova, M.P., H2AX phosphorylation at the sites of DNA double-strand breaks in cultivated mammalian cells and tissues, Clin. Epigenet., 2011, vol. 2, no. 2, p. 283.
Kuilman, T. and Peeper, D.S., Senescence-messaging secretome: SMS-ing cellular stress, Nat. Rev. Cancer, 2009, vol. 9, no. 2, p. 81.
Coppé, J.P., Desprez, P.Y., Krtolica, A., and Campisi, J., The senescence-associated secretory phenotype: the dark side of tumor suppression, Annu. Rev. Pathol., 2010, vol. 5, p. 99.
Coppé, J.P., Kauser, K., Campisi, J., and Beauséjour, C.M., Secretion of vascular endothelial growth factor by primary human fibroblasts at senescence, J. Biol. Chem., 2006, vol. 281, no. 40, p. 29568.
Elzi, D.J., Song, M., Hakala, K., et al., Wnt antagonist SFRP1 functions as a secreted mediator of senescence, Mol. Cell Biol., 2012, vol. 32, no. 21, p. 4388.
Nelson, G., Wordsworth, J., Wang, C., et al., A senescent cell bystander effect: senescence-induced senescence, Aging Cell, 2012, vol. 11, no. 2, p. 345.
Freund, A., Orjalo, A., Desprez, P.Y., and Campisi, J., Inflammatory networks during cellular senescence: causes and consequences, Trends Mol. Med., 2010, vol. 16, p. 238.
Campisi, J. and Robert, L., Cell senescence: role in aging and age-related diseases, Interdiscip. Top Gerontol., 2014, vol. 39, p. 45.
Doles, J., Storer, M., Cozzuto, L., et al., Age-associated inflammation inhibits epidermal stem cell function, Genes Dev., 2012, vol. 26, no. 19, p. 2144.
Turinetto, V., Vitale, E., and Giachino, C., Senescence in human mesenchymal stem cells: functional changes and implications in stem cell-based therapy, Int. J. Mol. Sci., 2016, vol. 7, no. 7, p. E1164.
Li, Y., Wu, Q., Wang, Y., et al., Senescence of mesenchymal stem cells, Int. J. Mol. Med., 2017, vol. 39, no. 4, p. 775.
Payushina, O.V., Localization and functions of mesenchymal stromal cells in vivo, Biol. Bull. Rev., 2016, vol. 6, no. 1, p. 1.
Lunyak, V.V., Amaro-Ortiz, A., and Gaur, M., Mesenchymal stem cells secretory responses: senescence messaging secretome and immunomodulation perspective, Front. Genet., 2017, vol. 8, p. 220.
Ullah, I., Subbarao, R.B., and Rho, G.J., Human mesenchymal stem cells—current trends and future prospective, Biosci. Rep., 2015, vol. 35, no. 2, p. e00191.
Dominici, M., Le Blanc, K., Mueller, I., et al., Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement, Cytotherapy, 2006, vol. 8, no. 4, p. 315.
Hoogduijn, M.J., Betjes, M.G., and Baan, C.C., Mesenchymal stromal cells for organ transplantation: different sources and unique characteristics? Curr. Opin. Organ Transplant., 2014, vol. 19, no. 1, p. 41.
Mattar, P. and Bieback, K., Comparing the immunomodulatory properties of bone marrow, adipose tissue, and birth-associated tissue mesenchymal stromal cells, Front. Immunol., 2015, vol. 6, p. 560.
McLeod, C.M. and Mauck, R.L., On the origin and impact of mesenchymal stem cell heterogeneity: new insights and emerging tools for single cell analysis, Eur. Cell Mater., 2017, vol. 34, p. 217.
Andreeva, E.R. and Buravkova, L.B., Paracrine activity of multipotent mesenchymal stromal cells and its modulation in hypoxia, Hum. Physiol., 2013, vol. 39, no. 3, p. 315.
Richardson, S.M., Kalamegam, G., Pushparaj, P.N., et al., Mesenchymal stem cells in regenerative medicine: focus on articular cartilage and intervertebral disc regeneration, Methods, 2016, vol. 99, p. 69.
Rubina, K.A., Kalinina, N.I., Efimenko, A.Yu., et al., Mechanism of stimulation of angiogenesis in ischemic myocardium with the help of adipose tissue stromal cells, Kardiologiya, 2010, no. 50, p. 51.
Kalinina, N.I., Sysoeva, V.Yu., Rubina, K.A., et al., Mesenchymal stem cells in tissue growth and repair, Acta Nat., 2011, vol. 3, no. 4, p. 32.
Zuk, P.A., The adipose-derived stem cell: looking back and looking ahead, Mol. Biol. Cell, 2010, vol. 21, p. 1783.
Natesan, S., Zhang, G., Baer, D.G., et al., A bilayer construct controls adipose-derived stem cell differentiation into endothelial cells and pericytes without growth factor stimulation, Tissue Eng., Part A, 2011, vol. 17, nos. 7–8, p. 941.
Gnecchi, M., Danieli, P., Malpasso, G., and Ciuffreda, M.C., Paracrine mechanisms of mesenchymal stem cells in tissue repair, Methods Mol. Biol., 2016, vol. 1416, p. 123.
Hodgkinson, C.P., Bareja, A., Gomez, J.A., and Dzau, V.J., Emerging concepts in paracrine mechanisms in regenerative cardiovascular medicine and biology, Circ. Res., 2016, vol. 118, no. 1, p. 95.
Gornostaeva, A., Andreeva, E., and Buravkova, L., Factors governing the immunosuppressive effects of multipotent mesenchymal stromal cells in vitro, Cytotechnology, 2016, vol. 68, no. 4, p. 565.
Gu, Y., Li, T., Ding, Y., et al., Changes in mesenchymal stem cells following long-term culture in vitro, Mol. Med. Rep., 2016, vol. 13, no. 6, p. 5207.
Legzdina, D., Romanauska, A., Nikulshin, S., et al., Characterization of senescence of culture-expanded human adipose-derived mesenchymal stem cells, Int. J. Stem Cells, 2016, vol. 9, no. 1, p. 124.
Ratushnyy, A., Lobanova, M., and Buravkova, L.B., Expansion of adipose tissue-derived stromal cells at “physiologic” hypoxia attenuates replicative senescence, Cell Biochem. Funct., 2017, vol. 35, no. 4, p. 232.
Kim, M., Kim, C., Choi, Y.S., et al., Age-related alterations in mesenchymal stem cells related to shift in differentiation from osteogenic to adipogenic potential: implication to age-associated bone diseases and defects, Mech. Ageing Dev., 2012, vol. 133, no. 5, p. 215.
Despars, G., Carbonneau, C.L., Bardeau, P., et al., Loss of the osteogenic differentiation potential during senescence is limited to bone progenitor cells and is dependent on p53, PLoS One, 2013, vol. 8, no. 8, p. e73206.
Wagner, W., Horn, P., Castoldi, M., et al., Replicative senescence of mesenchymal stem cells: a continuous and organized process, PLoS One, 2008, vol. 3, no. 5, p. e2213.
Digirolamo, C.M., Stokes, D., Colter, D., et al., Propagation and senescence of human marrow stromal cells in culture: a simple colony-forming assay identifies samples with the greatest potential to propagate and differentiate, Br. J. Haematol., 1999, vol. 107, no. 2, p. 275.
Cheng, H., Qiu, L., Ma, J., et al., Replicative senescence of human bone marrow and umbilical cord derived mesenchymal stem cells and their differentiation to adipocytes and osteoblasts, Mol. Biol. Rep., 2011, vol. 38, no. 8, p. 5161.
Stechschulte, L.A. and Lecka-Czernik, B., Reciprocal regulation of PPARγ and RUNX2 activities in marrow mesenchymal stem cells: fine balance between p38 MAPK and protein phosphatase 5, Curr. Mol. Biol. Rep., 2017, vol. 3, no. 2, p. 107.
Lee, Y.H., Lee, H.Y., Kim, T.G., et al., PPARγ maintains homeostasis through autophagy regulation in dental pulp, J. Dent. Res., 2015, vol. 94, no. 5, p. 729.
Sepúlveda, J.C., Tomé, M., Fernández, M.E., et al., Cell senescence abrogates the therapeutic potential of human mesenchymal stem cells in the lethal endotoxemia model, Stem Cells, 2014, vol. 32, no. 7, p. 1865.
Baggiolini, M. and Clark-Lewis, I., Interleukin-8, a chemotactic and inflammatory cytokine, FEBS Lett., 1992, vol. 307, p. 97.
Li, A., Dubey, S., Varney, M.L., et al., IL-8 directly enhanced endothelial cell survival, proliferation, and matrix metalloproteinases production and regulated angiogenesis, J. Immunol., 2003, vol. 170, p. 3369.
Blankenberg, S., Rupprecht, H.J., Bickel, C., et al., Circulating cell adhesion molecules and death in patients with coronary artery disease, Circulation, 2001, vol. 104, p. 1336.
Tanuma, N., Sakuma, H., Sasaki, A., and Matsumoto, Y., Chemokine expression by astrocytes plays a role in microglia/macrophage activation and subsequent neurodegeneration in secondary progressive multiple sclerosis, Acta Neuropathol., 2006, vol. 112, p. 195.
Spoettl, T., Hausmann, M., Herlyn, M., et al., Monocyte chemoattractant protein-1 (MCP-1) inhibits the intestinal-like differentiation of monocytes, Clin. Exp. Immunol., 2006, vol. 145, p. 190.
Li, Y., Xu, X., Wang, L., et al., Senescent mesenchymal stem cells promote colorectal cancer cells growth via galectin-3 expression, Cell Biosci., 2015, vol. 5, p. 21.
Di, G.H., Liu, Y., Lu, Y., et al., IL-6 secreted from senescent mesenchymal stem cells promotes proliferation and migration of breast cancer cells, PLoS One, 2014, vol. 9, no. 11, p. e113572.
Skolekova, S., Matuskova, M., Bohac, M., et al., Cisplatin-induced mesenchymal stromal cells-mediated mechanism contributing to decreased antitumor effect in breast cancer cells, Cell Commun. Signaling, 2016, vol. 14, p. 4.
Minieri, V., Saviozzi, S., Gambarotta, G., et al., A new paradigm in cardiac regeneration: The mesenchymal stem cell secretome, Stem Cells Int., 2015, vol. 2015, p. 765846.
Özcan, S., Alessio, N., Acar, M.B., et al., Unbiased analysis of senescence associated secretory phenotype (SASP) to identify common components following different genotoxic stresses, Aging (N.Y.), 2016, vol. 8, no. 7, p. 1316.
van Niel, G., D’Angelo, G., and Raposo, G., Shedding light on the cell biology of extracellular vesicles, Nat. Rev. Mol. Cell Biol., 2018, vol. 19, no. 4, p. 213.
Tkach, M. and Théry, C., Communication by extracellular vesicles: where we are and where we need to go, Cell, 2016, vol. 164, no. 6, p. 1226.
Liu, S., Mahairaki, V., Bai, H., et al., Highly purified human extracellular vesicles produced by stem cells alleviate aging cellular phenotypes of senescent human cells, Stem Cells, 2019, vol. 37, no. 6, p. 779.
Geiger, H., de Haan, G., and Florian, M.C., The ageing hematopoietic stem cell compartment, Nat. Rev. Immunol., 2013, vol. 13, no. 5, p. 376.
Kfoury, Y. and Scadden, D.T., Mesenchymal cell contributions to the stem cell niche, Cell Stem Cell, 2015, vol. 16, no. 3, p. 239.
Adams, G.B., Martin, R.P., Alley, I.R., et al., Therapeutic targeting of a stem cell niche, Nat. Biotechnol., 2007, vol. 25, no. 2, p. 238.
Mendez-Ferrer, S., Michurina, T.V., Ferraro, F., et al., Mesenchymal and hematopoietic stem cells form a unique bone marrow niche, Nature, 2010, vol. 466, no. 7308, p. 829.
Mendelson, A. and Frenette, P.S., Hematopoietic stem cell niche maintenance during homeostasis and regeneration, Nat. Med., 2014, vol. 20, no. 8, p. 833.
Haas, S., Hansson, J., Klimmeck, D., et al., Inflammation-induced emergency megakaryopoiesis driven by hematopoietic stem cell-like megakaryocyte progenitors, Cell Stem Cell, 2015, vol. 17, no. 4, p. 422.
Pietras, E.M., Mirantes-Barbeito, C., Fong, S., et al., Chronic interleukin-1 exposure drives hematopoietic stem cells towards precocious myeloid differentiation at the expense of self-renewal, Nat. Cell Biol., 2016, vol. 18, no. 6, p. 607.
Bernardo, M.E. and Locatelli, F., Mesenchymal stromal cells in hematopoietic stem cell transplantation, Methods Mol. Biol., 2016, vol. 1416, p. 3.
Takeuchi, M., Takeuchi, K., Kohara, A., et al., Chromosomal instability in human mesenchymal stem cells immortalized with human papilloma virus E6, E7 and hTERT genes, In Vitro Cell Dev. Biol. Anim., 2007, vol. 43, nos. 3–4, p. 129.
Wei, F., Qu, C., Song, T., et al., Vitamin C treatment promotes mesenchymal stem cell sheet formation and tissue regeneration by elevating telomerase activity, J. Cell Physiol., 2012, vol. 227, no. 9, p. 3216.
Lin, T.M., Tsai, J.L., Lin, S.D., et al., Accelerated growth and prolonged lifespan of adipose tissue-derived human mesenchymal stem cells in a medium using reduced calcium and antioxidants, Stem Cells Dev., 2005, vol. 14, no. 1, p. 92.
Choi, J.R., Pingguan-Murphy, B., Wan Abas, W.A., et al., In situ normoxia enhances survival and proliferation rate of human adipose tissue-derived stromal cells without increasing the risk of tumourigenesis, PLoS One, 2015, vol. 10, no. 1, p. e0115034.
Buravkova, L.B., Andreeva, E.R., Gogvadze, V., and Zhivotovsky, B., Mesenchymal stem cells and hypoxia: where are we? Mitochondrion, 2014, vol. 19, part A, p. 105.
Gharibi, B., Farzadi, S., Ghuman, M., and Hughes, F.J., Inhibition of Akt/mTOR attenuates age-related changes in mesenchymal stem cells, Stem Cells, 2014, vol. 32, no. 8, p. 2256.
Okada, M., Kim, H.W., Matsuura, K., et al., Abrogation of age-induced microRNA-195 rejuvenates the senescent mesenchymal stem cells by reactivating telomerase, Stem Cells, 2016, vol. 34, no. 1, p. 148.
Gharibi, B. and Hughes, F.J., Effects of medium supplements on proliferation, differentiation potential and in vitro expansion of mesenchymal stem cells, Stem Cells Transl. Med., 2012, vol. 1, no. 11, p. 771.
Funding
The study was supported by the Russian Foundation for Basic Research (project no. 19-015-00150) and the scholarship of the President of the Russian Federation (no. SP-960.2019.4).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Translated by M. Batrukova
Rights and permissions
About this article
Cite this article
Ratushnyy, A.Y., Buravkova, L.B. Cell Senescence and Mesenchymal Stromal Cells. Hum Physiol 46, 85–93 (2020). https://doi.org/10.1134/S0362119720010132
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0362119720010132