Abstract
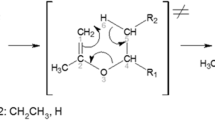
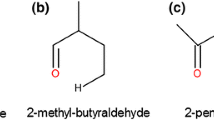
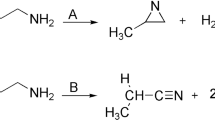
Quantum mechanical and Rice-Ramsperger-Kassel-Marcus (RRKM) calculations are carried out to study the thermal unimolecular decomposition of oxetane (1), 2-methyloxetane (2), and 2,2-dimethyloxetane (3) at the MPW1PW91/6-311 + G** level of theory. The results of the calculations reveal that decomposition reaction of compounds 1–3 yields formaldehyde and the corresponding substituted olefin. The predicted high-pressure-limit rate constants for the decomposition compounds 1–3 are represented as 6.61 × 1013exp(−32472/T), 9.33 × 1013exp(−29873/T), and 4.79 × 1013exp(−27055/T) s−1, respectively. The fall-off pressures for the decomposition of compounds 1–3 are found to be 9.42 × 10−2, 3.67 × 10−3, and 7.26 × 10−4 mm Hg, respectively. As the fall-off pressure of the decomposition process of compounds 1–3 are in the following order: P 1/2(1) > P 1/2(2) > P 1/2(3); therefore the decomposition rates are as follow: rate(1) < rate(2) < (3).
Similar content being viewed by others
References
F. O. Rice and M. T. Murphy, J. Am. Chem. Soc. 64, 896 (1942).
D. A. Bittker and W. D. Walters, J. Am. Chem. Soc. 77, 1429 (1955).
G. F. Cohoe and W. D. Walters, J. Phys. Chem. 71, 2326 (1967).
A. Barbot, Bull. Chim. France 2, 1438 (1935).
A. Bardot, Ann. Chim. (Paris) 11, 519 (1939).
K. A. Holbrook and R. A. Scott, J. Chem. Soc., Faraday Trans. I 71, 1849 (1975).
P. Hammonds and K. A. Holbrook, J. Chem. Soc., Faraday Trans. I 78, 2195 (1982).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuse- ria, M. A. Robb, J. R. Cheeseman, V. G. Zakrazewski, J. A. Montgomery, Jr. R. E. Startmann, J. C. Burant, S. Dapprich, J. M. Millam, A. D. Daniels, K. N. Kudin, M. C. Strain, O. Farkas, J. Tomasi, V. Barone, M. Cossi, R. Cammi, B. Mennucci, C. Adamo, S. Clif- ford, J. Ochterski, G. A. Petersson, P. Y. Ayala, Q. Cui, K. Morokuma, D. K. Malik, A. D. Rabuck, K. Raghavachar, J. B. Foresman, J. Cioslowski, J. V. Ortiz, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. Gomperts, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanyakkara, C. Gonzalez, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, J. L. Andres, M. Head-Gordon, E. S. Replogle, and J. A. Pople, Gaussian 98, Revision A.3 (Gaussian Inc., Pittsburgh, PA, USA, 1998).
Gauss View 03 (Gaussian Inc., Pittsburgh PA, 2003).
Serena Software (Box 3076, Bloomington, IN, USA).
J. J. P. Stewart, J. Computer-Aided Mol. Design 4, 1 (1990).
J. J. P. Stewart, QCPE 581 (Department of Chemistry, Indiana Univ., Bloomington, IN, USA, 1990).
M. J. S. Dewar, E. F. Heally, and J. J. P. Stewart, J. Chem. Soc., Faraday Trans. II 80, 227 (1984).
O. Ermer, Tetrahedron 31, 1849 (1975).
J. W. Mclever, Jr., Acc. Chem. Res. 7, 72 (1974).
A. Tahan, and A. Shiroudi, Chin. J. Struct. Chem. 30, 697 (2011).
W. J. Hehre, L. Random, P. V. R. Schleyer, and J. A. Pople, Ab initio Molecular Orbital Theory (Wiley, New York, 1986).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Shiroudi, A., Tahan, A. & Zahedi, E. The unimolecular thermal decomposition of oxetane and its methyl derivatives: An Ab initio and RRKM calculations. Russ. J. Phys. Chem. 86, 1245–1249 (2012). https://doi.org/10.1134/S0036024412060246
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024412060246