Abstract
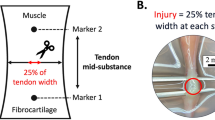
Previous studies have shown fetal tissues heal in a regenerative fashion without scar formation. The objective of this study is to compare the healing properties of adult and fetal tendons. Time-mated pregnant ewes at 80–85 days of gestation were utilized. A partial, midsubstance tenotomy was performed in the lateral extensor fetal tendons, and analogous tenotomies were created in the maternal limbs. One week after injury, the fetal and adult animals were sacrificed, and tendons were histologically and mechanically evaluated. Immunohistochemical staining for transforming growth factor beta isoform 1 (TGF-β1) was performed. Histologically, a gap with granulation tissue and inflammatory cells was visible in the site of wounding in the adult tendons. In the fetal tendons, no abnormalities were noted in the wound, with reconstitution of collagen architecture. TGF-β1 expression was low in fetal but upregulated in the adult wounds. No significant differences were found in the biomechanical properties between groups. We identified regenerative healing properties in injured fetal tendon, while adult tendon tissue healed reparatively with scar formation. Fetal tendons demonstrated a limited recovery of mechanical properties after injury that was no better than that of the adult tendons at seven days. A better understanding of the mechanisms of fetal healing may lead to novel therapeutic strategies in the clinical setting. © 2003 Biomedical Engineering Society.
PAC2003: 8719Rr, 8718La
Similar content being viewed by others
References
Abrahamsson, S. O., G. Lundborg, and L. S. Lohmander. Recombinant human insulin-like growth factor-I stimulates matrix synthesis and cell proliferation in rabbit flexor tendon. J. Orthop. Res. 9:495–502, 1991.
Adzick, N. S., and M. T. Longaker. Scarless fetal healing. Therapeutic implications. Ann. Surg. 215:3–7, 1992.
Banes, A. J., M. Tsuzaki, P. Hu, B. Brigman, T. Brown, L. Almekinders, W. T. Lawrence, and T. Fischer. PDGF–BB, IGF–I and mechanical load stimulate DNA synthesis in avian tendon fibroblasts. J. Biomech. 28:1505–1513, 1995.
Beanes, S. R., et al. Down-regulation of decorin, a transforming growth factor-beta modulator is associated with scarless fetal wound healing. J. Pediatr. Surg. 36:1666–1671, 2001.
Bidder, M., D. A. Towler, R. H. Gelberman, and M. I. Boyer. Expression of mRNA for vascular endothelial growth factor at the repair site of healing canine flexor tendon. J. Orthop. Res. 18:247–252, 2000.
Boyer, M. I., J. T. Watson, J. Lou, P. R. Manske, R. H. Gelberman, and S. R. Cai. Quantitative variation in vascular endothelial growth factor mRNA expression during early flexor tendon healing: An investigation in a canine model. J. Orthop. Res. 19:869–872, 2001.
Burrington, J. D. Wound healing in the fetal lamb. J. Pediatr. Surg. 6:523–528, 1971.
Chan, B. P., S. Fu, L. Qin, K. Lee, C. G. Rolf, and K. Chan. Effects of basic fibroblast growth factor (bFGF) on early stages of tendon healing: A rat patellar tendon model. Acta Orthop. Scand. 71:513–518, 2000.
Chang, J., D. Most, E. Stelnicki, J. V. Siebert, M. T. Longaker, K. Hui, and W. C. Lineaweaver. Gene expression of transforming growth factor beta-1 in rabbit zone II flexor tendon wound healing: Evidence for dual mechanisms of repair. Plast. Reconstr. Surg. 100:937–944, 1997.
Chang, J., D. Most, R. Thunder, B. Mehrara, M. T. Longaker, and W. C. Lineaweaver. Molecular studies in flexor tendon wound healing: The role of basic fibroblast growth factor gene expression. J. Hand Surg. [Am]23:1052–1058, 1998.
Chang, J., R. Thunder, D. Most, M. T. Longaker, and W. C. Lineaweaver. Studies in flexor tendon wound healing: Neutralizing antibody to TGF-beta 1 increases postoperative range of motion. Plast. Reconstr. Surg. 105:148–155, 2000.
Coleman, C., T. L. Tuan, S. Buckley, K. D. Anderson, and D. Warburton. Contractility, transforming growth factor-beta, and plasmin in fetal skin fibroblasts: Role in scarless wound healing. Pediatr. Res. 43:403–409, 1998.
Cowin, A. J., T. M. Holmes, P. Brosnan, and M. W. Ferguson. Expression of TGF-beta and its receptors in murine fetal and adult dermal wounds. Eur. J. Dermatol. 11:424–431, 2001.
Derwin, K. A., L. J. Soslowsky, W. D. Green, and S. H. Elder. A new optical system for the determination of deformations and strains: Calibration characteristics and experimental results. J. Biomech. 27:1277–1285, 1994.
Jaibaji, M. Advances in the biology of zone II flexor tendon healing and adhesion formation. Ann. Plast. Surg. 45:83–92, 2000.
Julia, M. V., A. Albert, L. Morales, D. Miro, M. A. Sancho, and X. Garcia. Wound healing in the fetal period: The resistance of the scar to rupture. J. Pediatr. Surg. 28:1458–1462, 1993.
Levinson, H., Z. Peled, W. Liu, M. T. Longaker, G. M. Allison, and H. P. Ehrlich. Fetal rat amniotic fluid: Transforming growth factor beta and fibroblast collagen lattice contraction. J. Surg. Res. 100:205–210, 2001.
Lin, K. Y., J. C. Posnick, M. M. al-Qattan, J. Vajsar, and L. E. Becker. Fetal nerve healing: An experimental study. Plast. Reconstr. Surg. 93:1323–1333, 1994.
Lin, R. Y., K. M. Sullivan, P. A. Argenta, M. Meuli, H. P. Lorenz, and N. S. Adzick. Exogenous transforming growth factor-beta amplifies its own expression and induces scar formation in a model of human fetal skin repair. Ann. Surg. 222:146–154, 1995.
Lorenz, H. P. TGF-beta isoform and receptor expression during scarless wound repair. J. Craniofac Surg. 12:387–388, 2001.
Lorenz, H. P., R. Y. Lin, M. T. Longaker, D. J. Whitby, and N. S. Adzick. The fetal fibroblast: The effector cell of scarless fetal skin repair. Plast. Reconstr. Surg. 96:1251–1259, 1260–1261, 1995.
Martin, P., M. C. Dickson, F. A. Millan, and R. J. Akhurst. Rapid induction and clearance of TGF beta 1 is an early response to wounding in the mouse embryo. Dev. Genet. 14:225–238, 1993.
Nakamura, Y., S. Hirano, K. Suzuki, K. Seki, T. Sagara, and T. Nishida. Signaling mechanism of TGF-beta1–induced collagen contraction mediated by bovine trabecular meshwork cells. Invest. Ophthalmol. Visual Sci. 43:3465–3472, 2002.
Namba, R. S., M. Meuli, K. M. Sullivan, A. X. Le, and N. S. Adzick. Spontaneous repair of superficial defects in articular cartilage in a fetal lamb model. J. Bone Jt. Surg., Am. Vol. 80:4–10, 1998.
Peled, Z. M., S. J. Rhee, M. Hsu, J. Chang, T. M. Krummel, and M. T. Longaker. The ontogeny of scarless healing II: EGF and PDGF-B gene expression in fetal rat skin and fibroblasts as a function of gestational age. Ann. Plast. Surg. 47:417–424, 2001.
Robbins, J. R., S. P. Evanko, and K. G. Vogel. Mechanical loading and TGF-beta regulate proteoglycan synthesis in tendon. Arch. Biochem. Biophys. 342:203–211, 1997.
Rowlatt, U. Intrauterine wound healing in a 20 week human fetus. Virchows Arch. A: Pathol. Anat. Histol. 381:353–361, 1979.
Shah, M., D. M. Foreman, and M. W. Ferguson. Control of scarring in adult wounds by neutralising antibody to transforming growth factor beta. Lancet339:213–214, 1992.
Shah, M., D. M. Foreman, and M. W. Ferguson. Neutralisation of TGF-beta 1 and TGF-beta 2 or exogenous addition of TGF-beta 3 to cutaneous rat wounds reduces scarring. J. Cell. Sci. 108:985–1002, 1995.
Slate, R. K., J. C. Posnick, M. D. Wells, J. A. Goldstein, F. W. Keeley, and P. S. Thorner. Fetal tibial bone healing in utero: The effects of miniplate fixation. Plast. Reconstr. Surg. 92:874–883, 1993.
Soslowsky, L. J., S. Thomopoulos, S. Tun, C. L. Flanagan, C. C. Keefer, J. Mastaw, and J. E. Carpenter. Neer Award 1999. Overuse activity injures the supraspinatus tendon in an animal model: A histologic and biomechanical study. J. Shoulder Elbow Surg. 9:79–84, 2000.
Strickland, J. W. Flexor Tendons—Acute Injuries; edited by W. C. Pederson. Philadelphia: Churchill Livingstone, 1999, pp. 1851–1897.
Thomopoulos, S., G. R. Williams, and L. J. Soslowsky. Tendon to bone healing: Changes in structural, compositional, and biomechanical properties due to post-operative activity level. In 48th Annual Meeting, Orthopaedic Research Society, Dallas, TX, 2002.
Vaughan, M. B., E. W. Howard, and J. J. Tomasek. Transforming growth factor-beta 1 promotes the morphological and functional differentiation of the myofibroblast. Exp. Cell Res. 257:180–189, 2000.
Weinzweig, J., K. E. Panter, M. Pantaloni, A. Spangenberger, J. S. Harper, F. Lui, D. Gardner, T. L. Wierenga, and L. E. Edstrom. The fetal cleft palate: I. Characterization of a congential model. Plast. Reconstr. Surg. 103:419–428, 1999.
Weinzweig, J., K. E. Panter, M. Pantaloni, A. Spangenberger, J. S. Harper, F. Lui, L. F. James, and L. E. Edstrom. The fetal cleft palate: II. Scarless healing after in utero repair of a congential model. Plast. Reconstr. Surg. 104:1356–1364, 1999.
Whitby, D. J., H. F. McMullen, J. J. Sung, L. I. Gold, J. W. Siebert, and M. T. Longaker. Localization of TGF-beta isoforms in adult and fetal mouse lip wounds. Surg. Forum45:651–653, 1994.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Beredjiklian, P.K., Favata, M., Cartmell, J.S. et al. Regenerative Versus Reparative Healing in Tendon: A Study of Biomechanical and Histological Properties in Fetal Sheep. Annals of Biomedical Engineering 31, 1143–1152 (2003). https://doi.org/10.1114/1.1616931
Issue Date:
DOI: https://doi.org/10.1114/1.1616931